Advanced Manufacturing of TSL-1502M Intermediate for High-Purity PARP Inhibitor Production
Introduction to Next-Generation PARP Inhibitor Intermediates
The landscape of oncology drug development has been significantly reshaped by the advent of Poly (ADP-ribose) polymerase (PARP) inhibitors, which have demonstrated profound efficacy in treating advanced solid tumors, particularly those with BRCA1/2 gene defects. Central to the commercial viability of these life-saving therapeutics is the robust and scalable synthesis of their key intermediates. Patent CN112010850A discloses a breakthrough preparation method for TSL-1502M, a critical intermediate used in the manufacture of the potent PARP inhibitor TSL-1502. This patent outlines a novel reductive amination strategy that addresses longstanding challenges in yield and purity, offering a streamlined pathway for industrial production. By leveraging sodium cyanoborohydride as a selective reducing agent, the disclosed process achieves exceptional product quality while maintaining operational simplicity, making it an ideal candidate for reliable pharmaceutical intermediate supplier networks aiming to secure the global supply chain for next-generation cancer therapies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex heterocyclic amines like those found in the TSL-1502 scaffold has relied on alkylation strategies that often suffer from poor regioselectivity and harsh reaction conditions. Traditional methods frequently require strong bases or elevated temperatures, which can lead to the degradation of sensitive functional groups and the formation of difficult-to-remove impurities. Furthermore, earlier synthetic routes described in prior art, such as those found in related patent families, often involve multi-step sequences with low overall yields, necessitating extensive purification protocols that drive up manufacturing costs and extend lead times. These inefficiencies create significant bottlenecks for cost reduction in API manufacturing, as the accumulation of byproducts complicates the isolation of the active pharmaceutical ingredient and increases the burden on waste management systems.
The Novel Approach
In stark contrast to these legacy methods, the invention detailed in CN112010850A introduces a highly efficient reductive amination protocol that transforms the precursor compound of Formula I directly into the target intermediate TSL-1502M. This innovative route utilizes propionaldehyde and sodium cyanoborohydride under mild acidic conditions, effectively bypassing the need for aggressive reagents. The process is characterized by its operational simplicity and high atom economy, allowing for the direct installation of the propyl group onto the pyrrolidine ring with remarkable precision. As illustrated in the reaction scheme below, this approach not only simplifies the synthetic tree but also inherently suppresses the formation of side products, thereby enhancing the overall process robustness.
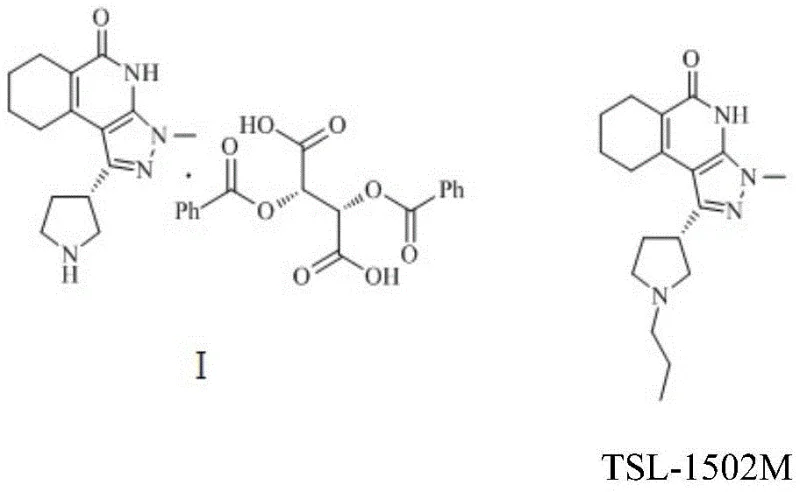
The strategic implementation of this novel chemistry enables manufacturers to achieve molar yields exceeding 70% with product purities greater than 99.5%, setting a new benchmark for efficiency in the production of high-purity pharmaceutical intermediates. By eliminating the need for complex protection-deprotection sequences often associated with amine functionalization, this method significantly reduces the number of unit operations required, directly translating to lower capital expenditure and improved throughput for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Sodium Cyanoborohydride-Catalyzed Reductive Amination
The success of this synthesis hinges on the unique chemoselectivity of sodium cyanoborohydride (NaBH3CN), a reagent that plays a pivotal role in controlling the reaction trajectory. The mechanism proceeds through the initial condensation of the secondary amine in the precursor with propionaldehyde to form an iminium ion intermediate. Unlike standard borohydrides, the electron-withdrawing cyano group in NaBH3CN reduces the nucleophilicity of the hydride species, rendering it stable in acidic media and selective for iminium ions over ketones or aldehydes. This selectivity is paramount, as it prevents the unwanted reduction of the carbonyl group present in the isoquinolinone core of the molecule, ensuring that the structural integrity of the pharmacophore is preserved throughout the transformation.
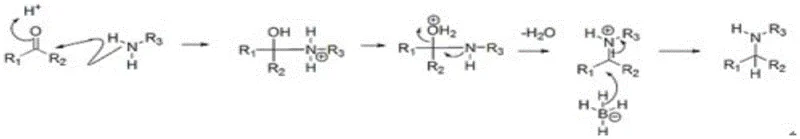
Furthermore, the reaction conditions are meticulously optimized to manage the exothermic nature of the imine formation and subsequent reduction. The patent specifies a strict temperature window of 10-20°C during the addition of reagents, a critical parameter that mitigates the risk of thermal runaway which could otherwise lead to the decomposition of the reducing agent or the formation of polymeric impurities. The quenching process is equally sophisticated; the addition of hydrochloric acid not only neutralizes the reaction mixture but also facilitates the decomposition of excess cyanoborohydride into volatile and water-soluble byproducts like hydrogen cyanide and boric acid. These byproducts are efficiently removed during the subsequent concentration and extraction steps, ensuring that the final product is free from toxic metal residues or persistent inorganic contaminants, a key requirement for meeting stringent purity specifications in drug substance manufacturing.
How to Synthesize TSL-1502M Efficiently
The practical execution of this synthesis requires precise adherence to the specified stoichiometry and thermal profiles to maximize yield and minimize impurity generation. The process begins with the dissolution of the chiral precursor salt in ethanol, followed by the controlled addition of the reducing agent and aldehyde. The subsequent workup involves a series of pH adjustments and liquid-liquid extractions designed to separate the basic product from acidic impurities and unreacted starting materials. For a comprehensive understanding of the standardized operating procedures required to replicate this high-yielding process, please refer to the detailed technical guide below.
- Dissolve the precursor compound (Formula I) in ethanol and cool the mixture to 5-15°C before adding sodium cyanoborohydride.
- Dropwise add propionaldehyde while maintaining the reaction temperature between 10-20°C to prevent exothermic runaway and impurity formation.
- Quench the reaction with hydrochloric acid, adjust pH, and perform a multi-stage extraction process using dichloromethane and sodium bicarbonate to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this reductive amination technology offers transformative benefits for procurement and supply chain stakeholders seeking to optimize their sourcing strategies. The streamlined nature of the process eliminates several unit operations typically required in traditional alkylation routes, resulting in a drastically simplified production workflow that enhances overall equipment effectiveness. This simplification directly contributes to substantial cost savings by reducing solvent consumption, energy usage, and labor hours associated with complex purification steps. Moreover, the use of readily available and inexpensive reagents like propionaldehyde and sodium cyanoborohydride ensures a stable and resilient supply chain, mitigating the risks associated with sourcing exotic or volatile raw materials.
- Cost Reduction in Manufacturing: The elimination of harsh reagents and the reduction in purification complexity lead to significant operational cost efficiencies. By avoiding the use of expensive catalysts or protecting groups, the process lowers the direct material costs while simultaneously reducing the environmental footprint associated with waste disposal. The high selectivity of the reaction minimizes the loss of valuable chiral intermediates, ensuring that the maximum amount of starting material is converted into the desired product, which is a critical factor in improving the overall cost-effectiveness of the manufacturing campaign.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures consistent batch-to-batch quality, which is essential for maintaining uninterrupted supply to downstream API manufacturers. The mild reaction conditions reduce the likelihood of equipment corrosion or failure, thereby extending the lifespan of manufacturing assets and reducing maintenance downtime. Additionally, the scalability of the process from laboratory to pilot and commercial scales has been validated, providing confidence that supply volumes can be ramped up rapidly to meet fluctuating market demands without compromising on product quality or delivery timelines.
- Scalability and Environmental Compliance: The process design inherently supports green chemistry principles by utilizing ethanol as a primary solvent and generating minimal hazardous waste. The efficient removal of cyanide species during the quenching and workup phases ensures compliance with strict environmental regulations regarding toxic effluent discharge. This alignment with sustainability goals not only reduces regulatory risk but also enhances the corporate social responsibility profile of the supply chain, making it an attractive option for multinational corporations committed to ethical and eco-friendly sourcing practices.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this patented technology, we have compiled a set of frequently asked questions based on the specific details outlined in the patent documentation. These insights address common concerns regarding reaction safety, impurity profiles, and scalability, providing a clear roadmap for technical teams evaluating this process for potential integration into their manufacturing portfolios. Understanding these nuances is vital for making informed decisions about process adoption and supplier qualification.
Q: Why is sodium cyanoborohydride preferred for this synthesis?
A: Sodium cyanoborohydride offers superior chemoselectivity, reducing the imine intermediate without affecting other carbonyl groups, which minimizes side reactions and simplifies purification.
Q: What are the critical temperature controls during the reaction?
A: Maintaining the temperature between 10-20°C during the addition of reagents is crucial to prevent the formation of impurities caused by exothermic spikes.
Q: How is the final purity of TSL-1502M ensured?
A: High purity (>99.5%) is achieved through a rigorous workup involving pH adjustment, multiple dichloromethane extractions, and washing with sodium bicarbonate to remove acidic impurities and unreacted aldehydes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable TSL-1502M Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a dependable supply of high-quality intermediates for the development of life-saving oncology drugs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical clients. We are committed to delivering TSL-1502M and related PARP inhibitor intermediates with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify every batch against the highest industry standards.
We invite you to collaborate with us to leverage this advanced synthetic technology for your upcoming projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline constraints. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in process chemistry can drive value and efficiency in your supply chain.
