Advanced Stereospecific Synthesis of S-Pro-Xylane for High-End Cosmetic Manufacturing
Advanced Stereospecific Synthesis of S-Pro-Xylane for High-End Cosmetic Manufacturing
The landscape of cosmetic active ingredient manufacturing is undergoing a significant transformation driven by the demand for higher purity and stereospecific efficacy. Patent CN115819387A introduces a groundbreaking synthesis method for stereospecific hydroxypropyl tetrahydropyrane triol, widely known in the industry as S-Pro-Xylane or S-Boscalid. This innovation addresses the critical limitations of prior art by utilizing a highly efficient metal-catalyzed hydrogen transfer reaction to achieve specific S-configuration selectivity. Unlike traditional methods that often yield racemic mixtures requiring difficult separation, this novel approach leverages chiral Ruthenium catalysts to directly construct the desired stereochemistry with exceptional precision. For R&D directors and procurement specialists, this represents a pivotal shift towards safer, more scalable, and economically viable production of high-value anti-aging actives. The technology ensures that the final product meets the stringent purity specifications required by top-tier global cosmetic brands while maintaining a robust safety profile throughout the manufacturing process.
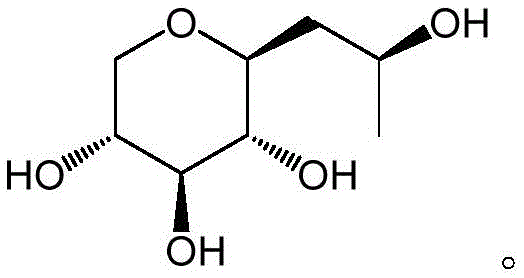
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of hydroxypropyl tetrahydropyrane triol has been plagued by significant technical and safety challenges that hinder large-scale commercial viability. Traditional routes often rely on the use of harsh stoichiometric reducing agents, such as lithium aluminum hydride or sodium borohydride, which pose severe safety hazards including fire and explosion risks in industrial settings. Furthermore, many existing processes fail to control stereochemistry effectively, resulting in racemic mixtures that necessitate complex and yield-depleting resolution steps to isolate the biologically active S-enantiomer. Previous patents, such as those involving acylation protection strategies, require multiple synthetic steps including triester formation and recrystallization, which drastically increase production costs and waste generation. The inability to simultaneously achieve high yield and high stereoselectivity has long been a bottleneck, forcing manufacturers to compromise on either purity or economic efficiency. These legacy methods also often involve toxic solvents and difficult post-treatment procedures that complicate regulatory compliance and environmental sustainability efforts.
The Novel Approach
The methodology disclosed in CN115819387A offers a transformative solution by replacing dangerous reduction protocols with a catalytic hydrogen transfer system. This innovative route begins with a benign Knoevenagel condensation in an aqueous phase, followed by a sophisticated asymmetric reduction using isopropanol as both solvent and hydrogen donor. By eliminating the need for external high-pressure hydrogen gas or hazardous hydride reagents, the process inherently enhances operational safety and simplifies reactor design requirements. The use of specific chiral Ruthenium catalysts allows for the direct conversion of the beta-xyloside intermediate into the S-configuration product with yields consistently exceeding 90%. This streamlined two-step sequence not only reduces the overall processing time but also minimizes the generation of chemical waste, aligning perfectly with modern green chemistry principles. For supply chain managers, this translates to a more reliable production schedule with fewer interruptions caused by safety incidents or complex purification bottlenecks.
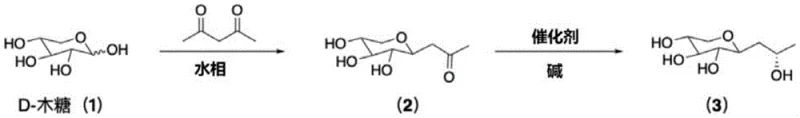
Mechanistic Insights into Ru-Catalyzed Asymmetric Transfer Hydrogenation
The core of this technological breakthrough lies in the precise application of Noyori-type asymmetric transfer hydrogenation, a mechanism that offers unparalleled control over stereochemical outcomes. In this system, the chiral Ruthenium catalyst acts as a molecular machine that selectively transfers hydride ions from the isopropanol solvent to the ketone carbonyl group of the intermediate. The chirality of the ligand environment surrounding the Ruthenium center dictates the facial selectivity of the hydride attack, ensuring that only the S-configuration is formed with high enantiomeric excess. This catalytic cycle is highly efficient because the catalyst is regenerated in situ, allowing for low catalyst loading while maintaining high turnover numbers. The reaction proceeds under mild alkaline conditions, which prevents the degradation of the sensitive sugar moiety that often occurs under acidic or strongly oxidative environments. Understanding this mechanism is crucial for R&D teams aiming to optimize reaction parameters such as temperature and base concentration to maximize throughput without compromising stereoselectivity.
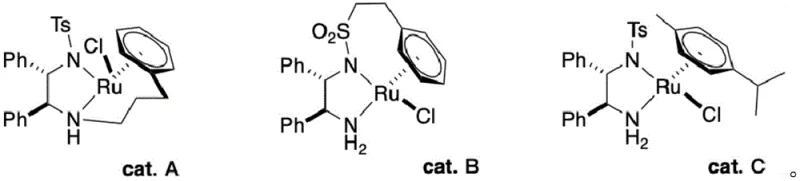
Impurity control in this synthesis is intrinsically linked to the high specificity of the catalytic system, which minimizes the formation of side products common in non-catalytic reductions. Traditional chemical reducers often lack discrimination, leading to over-reduction or the reduction of other functional groups, whereas the Ru-catalyst targets only the specific ketone functionality. The mild reaction conditions further suppress thermal degradation pathways that could generate colored impurities or polymeric byproducts, resulting in a crude product of exceptional quality. Post-reaction workup is simplified to pH adjustment and solvent evaporation, avoiding the complex extraction sequences needed to remove metal salts from stoichiometric reagents. This high level of purity from the crude stage significantly reduces the burden on downstream purification units, ensuring that the final API or cosmetic active meets rigorous international standards. For quality assurance teams, this mechanistic robustness provides a consistent impurity profile that is easier to validate and monitor across different production batches.
How to Synthesize S-Pro-Xylane Efficiently
The practical implementation of this synthesis route is designed for seamless integration into existing fine chemical manufacturing facilities with minimal retrofitting. The process initiates with the condensation of D-xylose and acetylacetone in water, a step that is exothermic but easily managed with standard cooling systems. Following the isolation of the beta-xyloside intermediate, the critical hydrogen transfer step is conducted in isopropanol, leveraging the solvent's dual role to drive the equilibrium forward. Detailed operational parameters regarding temperature ramps, catalyst activation, and crystallization protocols are essential for reproducing the high yields reported in the patent examples. Operators must adhere to strict nitrogen inerting procedures to protect the sensitive catalyst species from oxidation, ensuring long catalyst life and consistent performance. The following guide outlines the standardized workflow derived from the patent's exemplary embodiments to assist technical teams in process validation.
- Perform Knoevenagel condensation between D-xylose and acetylacetone in an aqueous alkaline phase to generate beta-acetonyl xyloside.
- Conduct asymmetric transfer hydrogenation of the intermediate using isopropanol as the hydrogen donor and a chiral Ruthenium catalyst under basic conditions.
- Isolate the final S-configuration product through pH adjustment, solvent removal, and recrystallization from methanol.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented synthesis route delivers substantial strategic advantages that directly impact the bottom line and supply chain resilience for cosmetic ingredient manufacturers. The elimination of hazardous reducing agents removes the need for specialized storage and handling infrastructure, thereby lowering capital expenditure and insurance costs associated with dangerous goods. By achieving high yields in fewer steps, the process drastically reduces raw material consumption and waste disposal fees, contributing to a significantly lower cost of goods sold. The reliance on readily available starting materials like D-xylose and acetylacetone ensures a stable supply chain that is not vulnerable to the geopolitical fluctuations often seen with exotic reagents. Furthermore, the simplicity of the workup procedure shortens the overall production cycle time, allowing facilities to increase throughput and respond more agilely to market demand spikes. These factors combined create a compelling value proposition for procurement managers seeking to optimize margins without sacrificing product quality.
- Cost Reduction in Manufacturing: The transition from stoichiometric reduction to catalytic hydrogen transfer fundamentally alters the cost structure by removing expensive reagents and simplifying waste treatment. Since the catalyst is used in small amounts and the solvent acts as the reductant, the variable cost per kilogram of product is significantly optimized compared to traditional hydride-based methods. Additionally, the high stereoselectivity eliminates the financial loss associated with discarding the unwanted R-enantiomer, effectively doubling the useful output from the same amount of raw material input. This efficiency gain allows manufacturers to offer more competitive pricing to downstream formulators while maintaining healthy profit margins. The reduction in processing steps also lowers utility consumption, including energy for heating and cooling, further enhancing the economic viability of the process.
- Enhanced Supply Chain Reliability: Utilizing a robust catalytic system with commercially available ligands and metals mitigates the risk of supply disruptions caused by single-source reagent dependencies. The process operates under mild conditions that are less demanding on equipment, reducing the frequency of maintenance downtime and extending the lifespan of reactor vessels. Because the reaction is tolerant to minor variations in feedstock quality, procurement teams have greater flexibility in sourcing raw materials without compromising the final product specification. This resilience is critical for maintaining continuous supply to major cosmetic brands that require just-in-time delivery schedules. The scalability of the water-based first step also means that production can be easily ramped up to meet seasonal demand surges without requiring massive new infrastructure investments.
- Scalability and Environmental Compliance: The green chemistry attributes of this method, particularly the use of water and isopropanol, align perfectly with increasingly stringent environmental regulations and corporate sustainability goals. The absence of heavy metal waste streams typical of stoichiometric reductions simplifies effluent treatment and reduces the environmental footprint of the manufacturing site. This eco-friendly profile enhances the marketability of the final ingredient to conscious consumers and brands committed to clean beauty standards. Scaling this process from pilot to commercial tonnage is straightforward due to the homogeneous nature of the reaction and the absence of gas-liquid mass transfer limitations found in high-pressure hydrogenation. Consequently, manufacturers can confidently commit to long-term supply agreements knowing that the technology is proven to perform reliably at industrial scales.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this stereospecific synthesis technology. These insights are derived directly from the patent data to provide clarity on process capabilities and product specifications. Understanding these details is essential for technical buyers evaluating the feasibility of adopting this new manufacturing route. The answers highlight the specific advantages in terms of safety, purity, and operational efficiency that distinguish this method from legacy technologies.
Q: Why is the S-configuration of Pro-Xylane preferred over the racemic mixture?
A: The S-configuration exhibits superior bioactivity in stimulating glycosaminoglycan (GAG) synthesis and promoting collagen production compared to the racemate or R-configuration, making it the gold standard for anti-aging formulations.
Q: What are the safety advantages of this hydrogen transfer method?
A: This method utilizes isopropanol as a safe hydrogen donor instead of hazardous stoichiometric reducing agents like lithium aluminum hydride, significantly eliminating explosion risks and simplifying industrial safety protocols.
Q: How does the new catalytic route improve yield compared to traditional methods?
A: By employing specific chiral Ruthenium complexes, the process achieves yields consistently over 90% with high stereoselectivity, avoiding the low yields and complex purification steps associated with older acylation-reduction pathways.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable S-Pro-Xylane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to maintain competitiveness in the global cosmetic ingredients market. Our team of expert chemists has thoroughly analyzed the technology disclosed in CN115819387A and is fully prepared to implement this state-of-the-art catalytic process for our clients. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab to plant is seamless and efficient. Our facility is equipped with stringent purity specifications and rigorous QC labs capable of verifying the high enantiomeric excess and chemical purity required for premium anti-aging applications. We are committed to delivering S-Pro-Xylane that not only meets but exceeds the performance expectations of leading international brands.
We invite procurement directors and R&D leaders to contact our technical procurement team to discuss how this innovative synthesis method can benefit your specific supply chain needs. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this catalytic route for your projects. We encourage you to request specific COA data and route feasibility assessments to validate the superior quality and reliability of our manufacturing capabilities. Let us collaborate to bring the next generation of high-performance cosmetic actives to market faster and more efficiently.
