Revolutionizing Cosmetic Active Production via Continuous Flow Hydrogenation Technology
The landscape of fine chemical manufacturing is undergoing a paradigm shift, moving decisively from traditional batch processing to advanced continuous flow technologies. A prime example of this evolution is found in the production of Hydroxypropyl Tetrahydropyrane Triol, widely known in the cosmetics industry as Pro-Xylane or Vitronectin (CAS 439685-79-7). This high-value molecule is critical for anti-aging formulations due to its ability to induce glycosaminoglycan biosynthesis. Recently, patent CN114835666A disclosed a groundbreaking continuous synthesis method that addresses long-standing inefficiencies in the supply chain. By leveraging microchannel reactors and fixed-bed catalytic hydrogenation, this technology promises to redefine the standards for purity and operational safety. For R&D directors and procurement specialists, understanding this transition is vital for securing a reliable cosmetic active ingredient supplier capable of meeting the rigorous demands of modern dermatological applications.
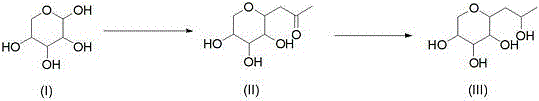
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of Hydroxypropyl Tetrahydropyrane Triol has been plagued by significant technical bottlenecks inherent to batch processing. The traditional route typically involves a condensation reaction between D-xylose and acetylacetone under alkaline conditions, followed by a reduction step using sodium borohydride. This legacy approach suffers from excessively long reaction times, often exceeding 13 hours for the complete two-step sequence, which severely limits throughput capacity. Moreover, the prolonged exposure of intermediates to high temperatures and alkaline environments leads to extensive degradation and the formation of deeply colored by-products. These impurities necessitate aggressive post-treatment protocols, including the heavy use of activated carbon, which unfortunately adsorbs the desired product along with the impurities, thereby depressing overall yield. Additionally, the reliance on sodium borohydride introduces boron-containing residues that are notoriously difficult to separate from polyhydroxy compounds, complicating purification and jeopardizing the stringent purity specifications required for cosmetic actives.
The Novel Approach
In stark contrast, the methodology outlined in patent CN114835666A introduces a fully continuous synthesis strategy that fundamentally alters the reaction dynamics. By utilizing a microchannel reactor for the initial condensation step, the residence time is slashed from hours to merely 30 to 120 seconds. This rapid processing prevents the thermal degradation responsible for product discoloration, resulting in a significantly lighter intermediate stream. Furthermore, the novel approach replaces the stoichiometric sodium borohydride reduction with a catalytic hydrogenation step performed in a fixed-bed reactor. This shift not only eliminates the introduction of boron impurities but also enhances the atom economy of the process. The continuous nature of the system allows for precise control over temperature and pressure, ensuring consistent product quality and facilitating seamless automation. This represents a massive leap forward in cost reduction in fine chemical manufacturing, transforming a labor-intensive batch operation into a streamlined, high-efficiency production line.
Mechanistic Insights into Microchannel Condensation and Fixed-Bed Hydrogenation
The core innovation of this synthesis lies in the superior heat and mass transfer characteristics of the microchannel reactor employed in the first step. In traditional batch vessels, heat removal during the exothermic condensation of D-xylose and acetylacetone is often inefficient, leading to localized hot spots that promote side reactions and polymerization. The microchannel architecture ensures near-instantaneous heat exchange, maintaining the reaction mixture precisely within the optimal 50-100°C window. This thermal precision is crucial for preserving the stereochemical integrity of the sugar derivative and minimizing the formation of chromophores. Following the rapid condensation, the stream is immediately neutralized with hydrochloric acid, quenching the alkalinity before it can degrade the sensitive intermediate. This immediate stabilization is a key factor in achieving the high-purity profiles observed in the final product, demonstrating why this method is superior for producing high-purity OLED material precursors or cosmetic actives where color is a critical quality attribute.
The second mechanistic breakthrough occurs in the fixed-bed hydrogenation unit, where the ketone functionality of the intermediate is reduced to the corresponding alcohol. Unlike batch hydrogenation, which poses safety risks due to the large headspace of hydrogen gas, the fixed-bed system operates with a continuous flow of hydrogen and liquid substrate over a supported catalyst, such as Ruthenium on Carbon. The elevated pressure (1-6 MPa) and temperature (80-150°C) within the confined catalyst bed drive the reduction to completion with high selectivity. Crucially, this heterogeneous catalysis avoids the generation of inorganic salt by-products associated with hydride reductions. The absence of boron species simplifies the downstream workup immensely, as there is no need for complex chelation or extensive washing steps to remove metal contaminants. This mechanistic elegance ensures that the final Hydroxypropyl Tetrahydropyrane Triol meets the rigorous purity standards demanded by global regulatory bodies for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Hydroxypropyl Tetrahydropyrane Triol Efficiently
Implementing this continuous synthesis route requires a strategic integration of fluid dynamics and catalytic engineering. The process begins with the precise metering of D-xylose and alkali solutions into the microreactor, where mixing efficiency dictates the initial yield. Operators must maintain strict control over flow rates to ensure the residence time remains within the narrow 30-120 second window to prevent over-reaction. Following neutralization, the stream enters the hydrogenation phase, where catalyst loading and hydrogen pressure become the critical variables. Detailed standard operating procedures regarding pump calibration, pressure relief systems, and catalyst activation are essential for safe operation. For a comprehensive breakdown of the specific parameters and equipment setup required to replicate this high-yield process, please refer to the standardized synthesis guide below.
- Prepare aqueous D-xylose and alkali solutions, then introduce them with acetylacetone into a microchannel reactor at 50-100°C for rapid condensation.
- Neutralize the intermediate stream with hydrochloric acid to adjust pH before entering the hydrogenation stage.
- Pass the neutralized stream through a fixed-bed reactor with a supported metal catalyst under hydrogen pressure to achieve final reduction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this continuous manufacturing protocol offers tangible economic and logistical benefits that extend far beyond simple yield improvements. The elimination of batch bottlenecks means that production capacity can be scaled linearly by running the system for longer durations or numbering up reactors, rather than building massive new vessels. This flexibility significantly reduces lead time for high-purity cosmetic actives, allowing suppliers to respond more agilely to market fluctuations. Furthermore, the inherent safety of the continuous hydrogenation process lowers insurance premiums and reduces the regulatory burden associated with handling hazardous chemicals in large quantities. The reduction in purification complexity translates directly into lower operational expenditures, making this a highly attractive option for cost-sensitive supply chains looking to optimize their sourcing strategies without compromising on quality.
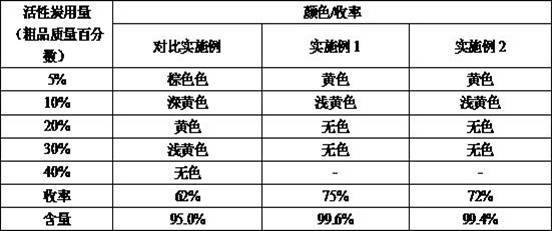
- Cost Reduction in Manufacturing: The most immediate financial impact stems from the drastic reduction in raw material waste and purification consumables. In the traditional batch process, the dark color of the intermediate necessitates the use of substantial amounts of activated carbon for decolorization, which inevitably adsorbs and traps a portion of the valuable product, lowering the effective yield. The continuous method produces a much lighter intermediate, requiring significantly less activated carbon. As demonstrated in the comparative data, this reduction in adsorbent usage not only lowers material costs but also recovers product that would otherwise be lost, effectively boosting the overall mass balance and reducing the cost per kilogram of the final API.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the long cycle times of batch manufacturing, where a single failed run can result in days of lost production. The continuous nature of this new method mitigates this risk by establishing a steady-state production environment where deviations are detected and corrected in real-time. The robustness of the fixed-bed catalyst system ensures consistent output quality over extended periods, reducing the frequency of batch rejections. This reliability is paramount for partners seeking a reliable cosmetic active ingredient supplier who can guarantee uninterrupted delivery schedules for large-scale formulation projects.
- Scalability and Environmental Compliance: From an environmental perspective, the process aligns perfectly with green chemistry principles by minimizing solvent usage and eliminating toxic boron waste streams. The absence of boron by-products simplifies wastewater treatment, reducing the load on effluent processing facilities and ensuring compliance with increasingly strict environmental regulations. Moreover, the compact footprint of microchannel and fixed-bed systems allows for significant capacity expansion within existing facility footprints, avoiding the capital expenditure associated with constructing new large-scale batch plants. This scalability ensures that the supply chain can grow in tandem with market demand for anti-aging ingredients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this continuous synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on how this method outperforms legacy techniques. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their own production lines or for procurement teams assessing supplier capabilities.
Q: How does the continuous flow method improve product purity compared to batch synthesis?
A: The continuous method utilizes a microchannel reactor which drastically reduces residence time in alkaline conditions, preventing the formation of colored by-products. Furthermore, it replaces sodium borohydride reduction with catalytic hydrogenation, eliminating difficult-to-remove boron impurities.
Q: What are the safety advantages of this new synthesis route?
A: Traditional batch hydrogenation involves large volumes of flammable solvents and hydrogen gas in a single vessel, posing significant explosion risks. The patented continuous fixed-bed system operates with minimal hold-up volume and precise pressure control, inherently safer for industrial scale-up.
Q: Does this process reduce waste generation during purification?
A: Yes, because the intermediate produced in the microchannel reactor has a much lighter color than batch intermediates, significantly less activated carbon is required for decolorization. This reduces solid waste and minimizes product loss due to adsorption.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hydroxypropyl Tetrahydropyrane Triol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of advanced continuous flow technologies is the key to unlocking the next generation of high-performance cosmetic and pharmaceutical ingredients. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN114835666A are fully realized in practical, industrial settings. We operate stringent purity specifications and maintain rigorous QC labs equipped to detect trace impurities, guaranteeing that every batch of Hydroxypropyl Tetrahydropyrane Triol meets the exacting standards required for premium skincare formulations. Our commitment to technological excellence ensures that our clients receive materials that are not only pure but also produced via sustainable and safe manufacturing practices.
We invite global partners to collaborate with us to leverage these cutting-edge synthesis routes for your product pipelines. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our advanced manufacturing capabilities can drive value and efficiency in your supply chain.
