Revolutionizing Fluvastatin Production: Advanced Enzymatic Catalysis for Commercial Scale-Up
Revolutionizing Fluvastatin Production: Advanced Enzymatic Catalysis for Commercial Scale-Up
The pharmaceutical industry is witnessing a paradigm shift towards biocatalytic processes, driven by the urgent need for greener, safer, and more selective synthetic routes. A prime example of this technological evolution is detailed in patent CN112322676A, which discloses a highly efficient method for preparing fluvastatin intermediates through enzyme catalysis. This innovation addresses critical bottlenecks in the traditional synthesis of fluvastatin diol, a key precursor for HMG-CoA reductase inhibitors used in treating hypercholesterolemia. By leveraging immobilized carbonyl reductase, the disclosed method achieves exceptional chiral purity and reaction efficiency, offering a compelling alternative to conventional chemical reduction strategies that often suffer from poor selectivity and hazardous operating conditions.
For R&D directors and process chemists, the implications of this patent are profound. It demonstrates how biological catalysts can be engineered and immobilized to withstand industrial processing requirements while delivering stereochemical precision that is difficult to achieve with small-molecule catalysts. The transition from harsh chemical reagents to mild enzymatic systems not only aligns with global sustainability goals but also simplifies downstream purification, thereby enhancing the overall economic viability of producing high-value pharmaceutical intermediates. As we delve deeper into the technical specifics, it becomes clear that this approach represents a significant leap forward in the manufacturing of complex statin drugs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fluvastatin and its intermediates has relied heavily on classical organic chemistry techniques that involve aggressive reagents and extreme conditions. Traditional routes often utilize sodium borohydride or lithium aluminum hydride for the reduction of ketone precursors, reactions that are inherently dangerous due to the pyrophoric nature of these reducing agents. Furthermore, chemical reduction frequently lacks the necessary stereoselectivity, resulting in racemic mixtures or products with low enantiomeric excess that require costly and time-consuming resolution steps. The background art cited in the patent highlights processes involving multiple steps, high energy consumption, and the generation of significant hazardous waste, all of which contribute to inflated production costs and supply chain vulnerabilities.
Beyond safety concerns, the chemical approach struggles with impurity profiles. The use of strong bases and reactive metals can lead to side reactions, degrading the sensitive indole and fluorophenyl moieties present in the fluvastatin structure. This necessitates rigorous purification protocols, often involving column chromatography or repeated recrystallizations, which drastically reduce overall yield. For procurement managers, these inefficiencies translate into higher raw material costs and longer lead times. The inability to consistently produce high-purity intermediates in a single pass creates a bottleneck that limits the scalability of the entire manufacturing operation, making it difficult to meet the surging global demand for cardiovascular medications.
The Novel Approach
In stark contrast, the novel enzymatic method described in the patent utilizes a specialized carbonyl reductase to convert fluvastatin ketone (Formula I) directly into fluvastatin diol (Formula II) with remarkable precision. This biocatalytic transformation occurs under mild alkaline conditions, typically between 25°C and 35°C, eliminating the thermal stress associated with traditional synthesis. The core of this innovation lies in the use of an immobilized enzyme system, where the carbonyl reductase is anchored onto a diatomaceous earth carrier and embedded with sodium alginate. This immobilization strategy not only stabilizes the enzyme but also facilitates its recovery and reuse, a critical factor for industrial application.
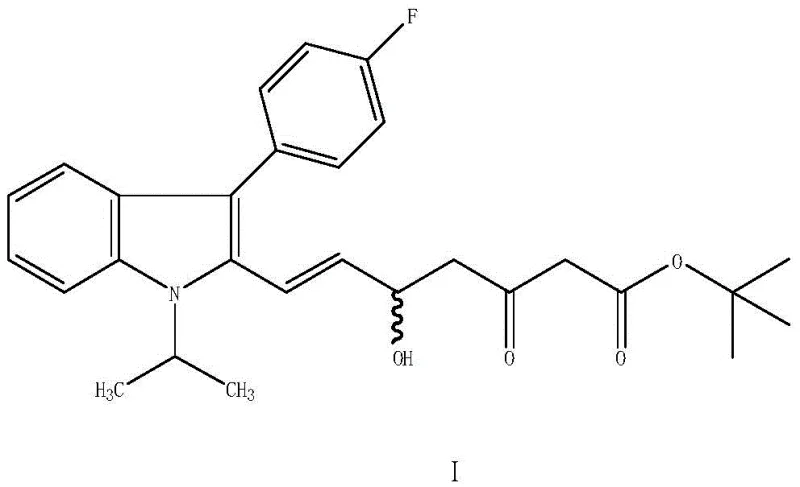
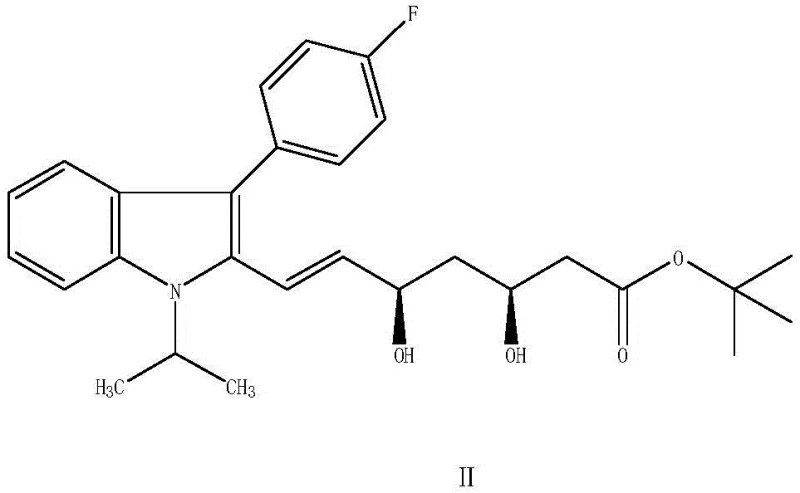
The reaction employs isopropanol as a hydrogen donor, which serves a dual purpose: it provides the necessary hydride equivalents for the reduction and participates in a cofactor regeneration cycle. This elegant design ensures that the expensive NADH cofactor is continuously recycled in situ, minimizing the need for stoichiometric amounts of external reducing agents. The result is a process that is not only safer and more environmentally friendly but also economically superior. By avoiding the use of heavy metals and hazardous chemicals, the novel approach simplifies waste treatment and reduces the regulatory burden, making it an attractive option for manufacturers seeking to optimize their production footprint while maintaining the highest standards of product quality.
Mechanistic Insights into Immobilized Carbonyl Reductase Catalysis
The success of this synthetic route hinges on the sophisticated mechanism of the immobilized carbonyl reductase. At the molecular level, the enzyme facilitates the stereospecific transfer of a hydride ion from the reduced nicotinamide adenine dinucleotide (NADH) cofactor to the carbonyl carbon of the fluvastatin ketone substrate. This transfer is highly regioselective and stereoselective, ensuring that the hydroxyl group is introduced with the correct configuration to form the biologically active (3R, 5S)-dihydroxy derivative. The enzyme's active site is precisely shaped to accommodate the bulky indole and fluorophenyl groups of the substrate, excluding other potential orientations that would lead to unwanted stereoisomers. This intrinsic selectivity is what allows the process to achieve enantiomeric excess (ee) values exceeding 99.5%, a benchmark that is exceptionally difficult to reach with chemical catalysts.
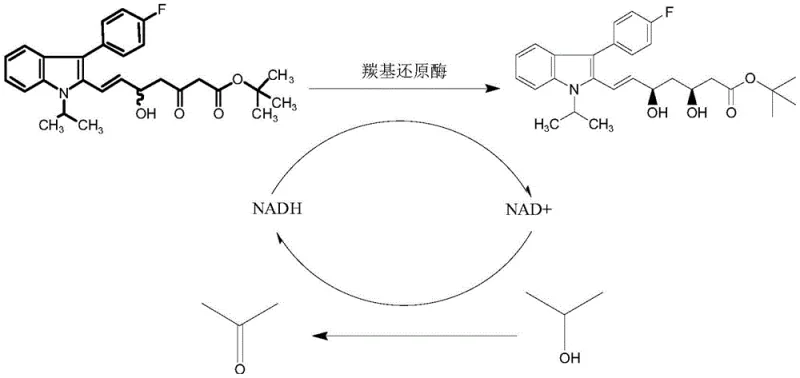
Crucially, the mechanism includes an integrated cofactor regeneration loop. As NADH is oxidized to NAD+ during the reduction of the ketone, the enzyme simultaneously catalyzes the oxidation of the hydrogen donor, isopropanol, back into acetone. This reaction regenerates NADH from NAD+, allowing the catalytic cycle to continue with only a catalytic amount of the cofactor. The immobilization of the enzyme on diatomaceous earth further enhances this mechanism by preventing enzyme aggregation and maintaining a high surface area for substrate interaction. The sodium alginate embedding acts as a protective matrix, shielding the enzyme from shear forces and solvent-induced denaturation. This structural integrity ensures that the catalyst remains active over multiple cycles, providing a robust and reliable platform for continuous or batch processing in a commercial setting.
How to Synthesize Fluvastatin Diol Efficiently
Implementing this enzymatic route requires careful attention to the preparation of the biocatalyst and the optimization of reaction parameters. The process begins with the immobilization of the carbonyl reductase, followed by the setup of the reaction vessel with the appropriate solvent system and hydrogen donor. The mild conditions allow for straightforward equipment requirements, avoiding the need for specialized high-pressure or cryogenic reactors. Detailed standard operating procedures regarding enzyme loading, solvent ratios, and crystallization protocols are essential for reproducibility. For a comprehensive guide on executing this synthesis with maximum efficiency and yield, please refer to the standardized protocol below.
- Prepare the immobilized carbonyl reductase by adsorbing the enzyme onto diatomaceous earth and embedding it with sodium alginate in an ether solvent, followed by filtration and freeze-drying.
- Conduct the enzymatic reaction by mixing fluvastatin ketone, a hydrogen donor like isopropanol, and a weak base such as triethanolamine in an ether solvent, then adding the immobilized enzyme and NAD+ cofactor.
- Perform post-reaction workup by filtering to recover the enzyme, removing the solvent under reduced pressure, and crystallizing the product from an acetonitrile-water mixture to obtain high-purity fluvastatin diol.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this enzymatic technology offers transformative benefits for procurement and supply chain operations. The shift away from hazardous chemical reagents significantly lowers the barrier to entry for manufacturing facilities, as it reduces the need for expensive safety infrastructure and specialized waste disposal services. The ability to recycle the immobilized enzyme multiple times directly impacts the cost of goods sold (COGS), as the catalyst constitutes a major portion of the variable costs in biocatalytic processes. Furthermore, the high selectivity of the reaction minimizes the formation of byproducts, which means less raw material is wasted on generating impurities that must later be removed. This efficiency translates into substantial cost savings and a more predictable supply of high-quality intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive and dangerous reducing agents like sodium borohydride, combined with the recyclability of the immobilized enzyme, leads to a drastic reduction in raw material expenses. The process operates at ambient temperatures, significantly lowering energy consumption compared to thermal chemical reactions. Additionally, the high purity of the crude product reduces the load on downstream purification units, saving on solvent usage and processing time, which collectively drives down the overall manufacturing cost.
- Enhanced Supply Chain Reliability: By simplifying the synthesis route and reducing the number of unit operations, the risk of production delays is minimized. The robustness of the immobilized enzyme ensures consistent batch-to-batch quality, reducing the likelihood of failed batches that can disrupt supply schedules. The use of common, commercially available solvents like MTBE and isopropanol further secures the supply chain against raw material shortages, ensuring a steady flow of intermediates to meet market demand.
- Scalability and Environmental Compliance: The mild reaction conditions and aqueous-compatible workup make this process highly scalable from pilot plant to commercial production without significant re-engineering. The reduction in hazardous waste generation aligns with increasingly stringent environmental regulations, mitigating the risk of compliance-related shutdowns. This green chemistry approach not only improves the corporate sustainability profile but also future-proofs the manufacturing asset against evolving regulatory landscapes.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this enzymatic process is vital for stakeholders evaluating its adoption. Common inquiries often revolve around the stability of the enzyme, the specific conditions required for optimal performance, and the quality of the final product. The following answers address these critical aspects based on the data provided in the patent documentation, offering clarity on how this technology performs in practical applications.
Q: What are the advantages of using immobilized carbonyl reductase over chemical reduction for fluvastatin?
A: The enzymatic method offers superior chiral selectivity with ee values exceeding 99.5%, eliminating the need for complex separation of stereoisomers. Additionally, it operates under mild conditions (25-35°C) without hazardous reducing agents like sodium borohydride, significantly improving process safety and environmental compliance.
Q: How does the immobilization process enhance the catalyst's performance?
A: Immobilizing the carbonyl reductase on diatomaceous earth with sodium alginate embedding prevents enzyme leaching and denaturation. This structural stability allows the catalyst to be recovered via simple filtration and reused multiple times while maintaining high catalytic activity and conversion rates.
Q: What solvents are compatible with this enzymatic synthesis route?
A: The process utilizes ether solvents such as methyl tert-butyl ether (MTBE), isopropyl ether, or tetrahydrofuran (THF). These solvents effectively dissolve both the substrate and product while facilitating easy separation from the immobilized enzyme and allowing for efficient solvent recovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluvastatin Diol Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of enzymatic catalysis in reshaping the landscape of pharmaceutical intermediate production. Our team of expert process chemists and engineers possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are uniquely positioned to adapt the innovative methods described in patent CN112322676A to meet your specific volume requirements, ensuring that you receive a supply of fluvastatin diol that meets stringent purity specifications. Our state-of-the-art rigorous QC labs are equipped to verify the high enantiomeric excess and chemical purity guaranteed by this advanced biocatalytic route.
We invite you to collaborate with us to leverage this cutting-edge technology for your supply chain. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your current production metrics. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our commitment to innovation can drive efficiency and reliability in your fluvastatin manufacturing operations.
