Scalable Synthesis of Telapristone Acetate Intermediates via Novel Bromination Strategy
The pharmaceutical industry continuously seeks robust and scalable pathways for synthesizing complex steroid intermediates, particularly for selective progesterone receptor modulators like Telapristone Acetate (CDB-4124). A pivotal advancement in this domain is detailed in patent CN103772468A, which discloses a highly efficient preparation method for the critical intermediate 3,3,20,20-bis(ethylenedioxy)-17α,21-dihydroxy-19-norpregna-5(10),9(11)-diene, also known as Compound 1. This molecule serves as the foundational scaffold for producing Telapristone Acetate, a potent therapeutic agent indicated for treating uterine fibroids, endometriosis, and breast cancer. The disclosed technology represents a significant leap forward in process chemistry, offering a streamlined 4-step synthesis that addresses long-standing challenges regarding safety, operational complexity, and yield optimization inherent in earlier generations of steroid manufacturing protocols.
For R&D directors and process chemists evaluating supply chain resilience, the shift away from hazardous reagents towards benign, high-yielding transformations is paramount. The methodology outlined in this patent not only ensures the availability of high-purity pharmaceutical intermediates but also aligns with modern green chemistry principles by minimizing toxic waste streams. By leveraging readily available starting materials and avoiding extreme reaction conditions, this route establishes a new benchmark for the commercial scale-up of complex steroid derivatives, ensuring a reliable supply of high-purity pharmaceutical intermediates for downstream API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those disclosed in WO9741145, have historically relied on precarious chemical transformations that pose significant risks to both personnel and production continuity. As illustrated in the reaction scheme below, the conventional Route 1 necessitates the use of highly toxic cyanide reagents to introduce the critical C17 side chain functionality. This reliance on cyanides creates severe occupational health hazards and imposes stringent environmental compliance burdens on manufacturing facilities, requiring specialized waste treatment infrastructure to neutralize lethal byproducts before discharge.
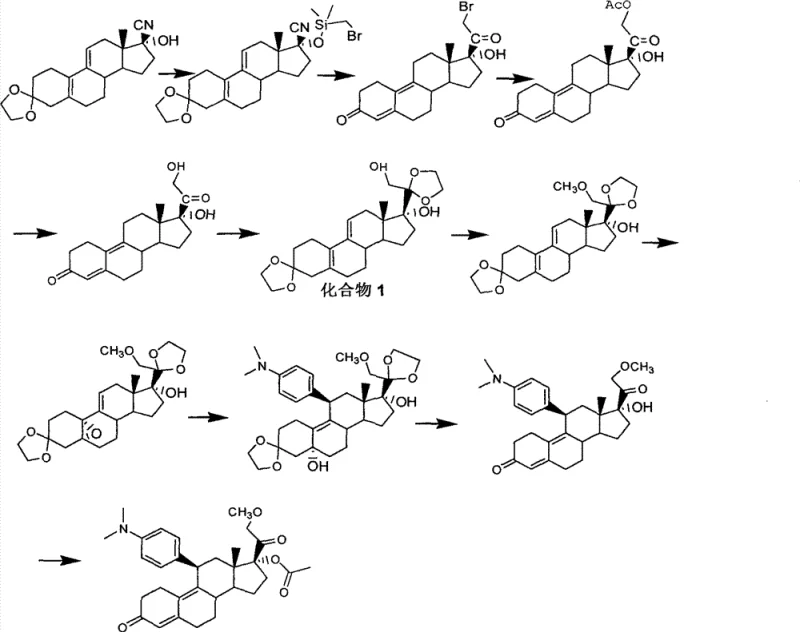
Furthermore, the operational parameters of these legacy processes are notoriously difficult to manage on an industrial scale. The initial steps often demand cryogenic conditions, specifically maintaining reaction temperatures as low as -78°C, which incurs substantial energy costs and requires specialized refrigeration equipment that limits batch sizes. Additionally, the purification of intermediates in these older routes frequently relies on column chromatography, a technique that is impractical for multi-kilogram or ton-scale production due to solvent consumption and throughput limitations. These factors collectively hinder cost reduction in API manufacturing and create bottlenecks in the supply chain.
The Novel Approach
In stark contrast, the innovative strategy presented in patent CN103772468A circumvents these obstacles through a clever redesign of the synthetic sequence. The new Route 2 initiates from Compound 5, bypassing the need for cyanide addition entirely by utilizing a bromination-hydrolysis sequence to construct the necessary oxygenated side chain. This approach operates under mild conditions, with the initial bromination occurring at approximately 0°C, a temperature easily achievable with standard industrial cooling water rather than expensive cryogenic fluids. The subsequent steps proceed at ambient temperature or under reflux, drastically simplifying the thermal management requirements for the reactor systems.
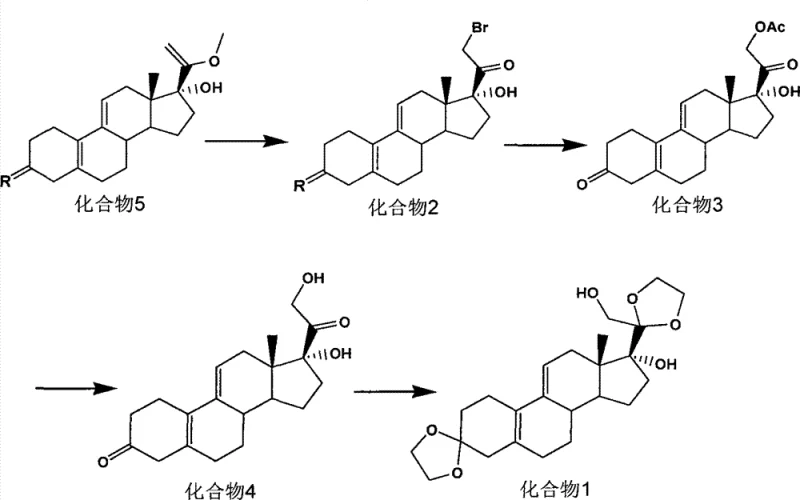
Moreover, the workup procedures in this novel approach are designed for scalability, utilizing standard extraction and crystallization techniques rather than chromatographic purification. This transition from batch-intensive purification to continuous or semi-continuous processing significantly enhances the throughput capacity of the manufacturing line. By eliminating the most hazardous and energy-intensive steps, this method not only improves the safety profile of the facility but also delivers a total molar yield exceeding 50%, proving that safety and efficiency can be achieved simultaneously in the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into the Bromination and Substitution Cascade
The core of this synthetic breakthrough lies in the precise functionalization of the steroid D-ring. The process begins with the bromination of Compound 5 (17α-hydroxy-20-methoxy-19-norpregna-5(10),9(11),20-triene-3-one or its protected variants) using elemental bromine in a dichloromethane and pyridine system. This electrophilic addition targets the C20-C21 double bond or facilitates alpha-bromination depending on the specific tautomer, ultimately yielding the 21-bromo intermediate, Compound 2. The use of pyridine acts as an acid scavenger, preventing acid-catalyzed degradation of the sensitive diene system while promoting the formation of the bromo-ketone motif essential for the subsequent nucleophilic displacement.
Following the bromination, the process employs a dual-function substitution and hydrolysis step using potassium acetate and potassium iodide in acetone. The iodide ion acts as a catalyst to facilitate the displacement of the bromide by the acetate nucleophile, forming the 21-acetoxy species found in Compound 3. Crucially, the reaction conditions are tuned to simultaneously hydrolyze the 3-position ketone protection if present, or simply install the acetate group with high regioselectivity. This cascade effectively installs the C21 oxygen functionality with the correct stereochemistry, setting the stage for the final deprotection and ketalization steps that yield the target diol Compound 1 with high fidelity and minimal epimerization.
How to Synthesize Telapristone Acetate Intermediate Efficiently
The execution of this synthesis requires careful control of stoichiometry and temperature to maximize the yield of each transformation. Starting from Compound 5, the bromination must be monitored closely via TLC to prevent over-bromination, followed by a quench into dilute hydrochloric acid to remove pyridine salts. The subsequent acetate substitution is driven to completion under reflux, ensuring full conversion of the bromo-intermediate. Finally, the global deprotection and ketalization using ethylene glycol and triethyl orthoformate in the presence of p-toluenesulfonic acid locks the 3 and 20 positions as ethylene ketals, stabilizing the molecule for storage and further derivatization. Detailed standardized synthesis steps are provided in the guide below.
- Perform bromination of Compound 5 at 0°C using bromine and pyridine in dichloromethane to yield Compound 2.
- Execute substitution and hydrolysis of Compound 2 using potassium acetate and potassium iodide to form Compound 3.
- Hydrolyze Compound 3 under reflux in potassium bicarbonate methanol solution to obtain Compound 4.
- Complete the synthesis by reacting Compound 4 with ethylene glycol and triethyl orthoformate at room temperature to yield Compound 1.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates directly into enhanced operational stability and risk mitigation. The elimination of cyanide reagents removes a major regulatory hurdle, simplifying the permitting process for new manufacturing lines and reducing the liability associated with handling Schedule 1 chemicals. This shift allows for a more flexible sourcing strategy for raw materials, as the starting materials for this route are more commoditized and less subject to the strict controls imposed on cyanide salts, thereby enhancing supply chain reliability and reducing lead time for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the removal of extreme thermal requirements. By operating at 0°C instead of -78°C, the facility eliminates the need for specialized cryogenic cooling loops, resulting in substantial energy savings and lower capital expenditure on refrigeration equipment. Furthermore, the avoidance of column chromatography in favor of crystallization and extraction significantly reduces solvent consumption and waste disposal costs, driving down the overall cost of goods sold (COGS) for the intermediate.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent batch-to-batch quality, minimizing the risk of production failures that can disrupt downstream API synthesis. The use of stable intermediates like Compound 2 and Compound 3 allows for potential stockpiling strategies, decoupling the production of the final intermediate from immediate demand fluctuations. This flexibility is crucial for maintaining continuity of supply for critical medications like Telapristone Acetate, ensuring that patient access is never compromised by manufacturing bottlenecks.
- Scalability and Environmental Compliance: The process is inherently designed for scale, utilizing common organic solvents like dichloromethane, acetone, and methanol which are easily recovered and recycled in standard distillation units. The absence of heavy metal catalysts or toxic cyanides simplifies the wastewater treatment profile, allowing the facility to meet stringent environmental discharge standards with greater ease. This environmental compatibility future-proofs the manufacturing asset against tightening global regulations on chemical emissions and hazardous waste generation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of the technology's readiness for industrial deployment. Understanding these nuances is essential for stakeholders evaluating the feasibility of integrating this intermediate into their existing production portfolios.
Q: What are the safety advantages of the new synthesis route for Telapristone Acetate intermediates?
A: The novel route described in patent CN103772468A completely eliminates the use of highly toxic cyanide reagents required in previous methods, significantly improving occupational health safety and reducing environmental hazards associated with waste disposal.
Q: How does the new process improve scalability compared to conventional methods?
A: Unlike conventional routes requiring cryogenic conditions at -78°C and complex column chromatography purification, this method operates at mild temperatures ranging from 0°C to reflux, facilitating easier thermal control and direct crystallization or extraction for large-scale production.
Q: What is the overall yield efficiency of this 4-step synthetic pathway?
A: The optimized 4-step sequence starting from Compound 5 achieves a total molar yield exceeding 50%, demonstrating high efficiency and material conservation suitable for cost-effective commercial manufacturing of steroid intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Telapristone Acetate Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable supply of high-quality steroid intermediates for the global pharmaceutical market. Our technical team has extensively analyzed the pathway described in CN103772468A and possesses the expertise to implement this 4-step synthesis with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from pilot plant validation to full-scale manufacturing without compromising on quality or timeline.
We invite you to collaborate with us to leverage this advanced synthetic route for your Telapristone Acetate projects. Our rigorous QC labs and stringent purity specifications guarantee that every batch of intermediate meets the highest international standards. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis, specific COA data, and route feasibility assessments tailored to your specific volume requirements and quality targets.
