Advanced Manufacturing of Allylestrenol: A Scalable 5-Step Synthetic Route for Global Hormone Supply Chains
Advanced Manufacturing of Allylestrenol: A Scalable 5-Step Synthetic Route for Global Hormone Supply Chains
The global demand for high-purity hormonal therapeutics necessitates robust, scalable, and economically viable synthetic pathways. Patent CN102225960A discloses a highly efficient preparation method for Allylestrenol (CAS 432-60-0), a critical oral progestogen used extensively in the management of threatened abortion and hormone-deficient miscarriages. This technical insight report analyzes the patented 5-step synthesis starting from Androst-4-ene-3,17-dione, highlighting its superiority over legacy fermentation and semi-synthetic routes. By leveraging selective protection strategies and a streamlined Birch reduction, this methodology delivers exceptional yield and purity, addressing the critical needs of R&D directors and supply chain managers for reliable pharmaceutical intermediates. The process eliminates the bottlenecks associated with biological variability, offering a chemically defined pathway that ensures batch-to-batch consistency essential for regulatory compliance in the hormonal sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of Allylestrenol has relied on complex routes that pose significant challenges for industrial scale-up and cost containment. For instance, prior art such as GB841411 describes a pathway involving fermentation, etherification, and reduction to obtain 17-hydroxy-estrand-3-methyl ether, followed by elimination and oxidation steps. This multi-stage approach is inherently flawed due to its excessive length and the rigorous production requirements associated with biological fermentation steps, which often introduce variability in raw material quality. Furthermore, the post-processing in these traditional routes is notoriously difficult, requiring extensive purification to remove biological impurities and side products, which drastically increases manufacturing costs and lead times. Another reported route in the Indian Journal of Chemistry, while operationally simpler, suffers from critically low yields at key intermediate stages, specifically intermediate 5, rendering the process economically unviable for large-scale hormone manufacturing. These limitations underscore the urgent need for a more direct, high-yielding chemical synthesis that bypasses the inefficiencies of older technologies.
The Novel Approach
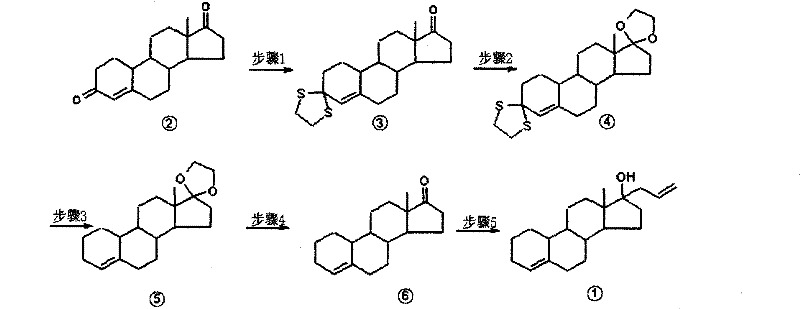
The methodology outlined in patent CN102225960A represents a paradigm shift in steroid synthesis, utilizing a concise 5-step sequence that maximizes atom economy and operational simplicity. Starting from the readily available Androst-4-ene-3,17-dione, the process employs a strategic orthogonal protection scheme: first protecting the C3 ketone as a dithioketal, followed by C17 ketalization. This specific order of operations is crucial for controlling regioselectivity and preventing unwanted side reactions during the subsequent reduction phase. The core innovation lies in the application of a Birch reduction to simultaneously eliminate the C3 thioketal and establish the desired ring unsaturation, a transformation that traditionally requires multiple discrete steps. By integrating these transformations, the novel approach significantly shortens the synthetic timeline, reduces solvent consumption, and simplifies the isolation of intermediates through straightforward crystallization and filtration techniques. This streamlined workflow not only enhances the overall throughput but also drastically lowers the environmental footprint, aligning with modern green chemistry principles in fine chemical intermediates production.
Mechanistic Insights into Selective Protection and Birch Reduction
The success of this synthetic route hinges on the precise control of functional group reactivity through selective protection. In the initial step, the C3 ketone of the starting dione is converted into a dithioketal using ethanedithiol catalyzed by boron trifluoride etherate at controlled temperatures between -5°C and 10°C. This transformation is highly exothermic and requires careful thermal management to prevent polymerization or degradation, yet it proceeds with remarkable efficiency, achieving yields up to 99%. The resulting dithioketal serves as a robust protecting group that is stable under the acidic conditions required for the subsequent C17 protection but labile enough to be removed during the dissolving metal reduction. Following this, the C17 ketone is masked as an ethylene ketal using ethylene glycol and p-toluenesulfonic acid. This step is equally critical, as it prevents nucleophilic attack at the C17 position during the Grignard reaction later in the sequence, ensuring that the allyl group is introduced exclusively at the intended site after deprotection. The high selectivity of these protection steps, with yields reaching 97% for the ketal formation, establishes a high-purity foundation for the downstream chemistry, minimizing the burden on purification units.
The mechanistic centerpiece of this route is the Birch reduction, performed using lithium metal in liquid ammonia at cryogenic temperatures ranging from -40°C to -50°C. In this reductive environment, solvated electrons facilitate the cleavage of the carbon-sulfur bonds in the C3 dithioketal, effectively removing the sulfur protecting groups while concurrently reducing the conjugated system of the steroid nucleus. The use of lithium in liquid ammonia provides a potent reducing environment that is uniquely capable of effecting this desulfurization without compromising the integrity of the sensitive steroid skeleton. Following the reduction, the reaction is quenched with absolute ethanol, and the ammonia is evaporated, leaving behind the desulfurized intermediate. Subsequent acidic hydrolysis removes the C17 ketal protection, regenerating the ketone functionality necessary for the final carbon-carbon bond formation. This sequence demonstrates a sophisticated understanding of steroid reactivity, allowing for the construction of the Allylestrenol framework with minimal byproduct formation and superior impurity profiles compared to non-selective reduction methods.
How to Synthesize Allylestrenol Efficiently
The synthesis of Allylestrenol via this patented route offers a clear roadmap for process chemists aiming to establish a robust manufacturing line. The protocol leverages standard unit operations such as low-temperature dosing, cryogenic reduction, and aqueous workups, making it highly transferable from pilot plant to commercial scale. The detailed procedure involves precise stoichiometric control, particularly in the Grignard step where allylmagnesium chloride is reacted with the deprotected ketone intermediate. Maintaining the reaction temperature between 0°C and 5°C during the addition of the Grignard reagent is paramount to controlling exotherms and preventing over-addition or decomposition of the organometallic species.
- Protect the C3 ketone of Androst-4-ene-3,17-dione using ethanedithiol and boron trifluoride etherate to form the dithioketal intermediate.
- Protect the C17 ketone using ethylene glycol and p-toluenesulfonic acid to form the dihydric alcohol ketal.
- Perform Birch reduction using lithium metal in liquid ammonia to eliminate the C3 thioketal and modify the A-ring unsaturation.
- Hydrolyze the C17 protecting group under acidic conditions to restore the C17 ketone functionality.
- React the resulting ketone with allylmagnesium chloride (Grignard reagent) to introduce the allyl group at C17, yielding Allylestrenol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route translates into tangible strategic advantages regarding cost stability and supply security. The elimination of fermentation steps removes the dependency on biological raw materials, which are often subject to seasonal fluctuations and biological contamination risks, thereby ensuring a more predictable and continuous supply of the active pharmaceutical ingredient. Furthermore, the high chemical yields observed in the initial protection steps mean that less starting material is wasted, directly correlating to a reduction in the cost of goods sold (COGS). The simplified purification protocols, which rely on crystallization and filtration rather than complex chromatography, significantly reduce the consumption of expensive solvents and silica gel, leading to substantial cost reduction in pharmaceutical intermediates manufacturing. Additionally, the shorter synthetic sequence reduces the overall cycle time per batch, allowing manufacturers to respond more agilely to market demand spikes without the need for excessive inventory buffering.
- Cost Reduction in Manufacturing: The process achieves exceptional yields in the early stages, with the dithioketal protection step reaching 99% and the ketal protection step reaching 97%. These high conversion rates minimize the loss of valuable steroid starting materials, which are often the most expensive component of the bill of materials. By maximizing the throughput of each reaction vessel, the facility can produce more output per unit of time and energy, driving down the fixed cost allocation per kilogram of product. Moreover, the avoidance of transition metal catalysts, which often require expensive removal steps to meet residual metal specifications, further streamlines the downstream processing costs.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as ethanedithiol, ethylene glycol, lithium, and allyl chloride ensures that the supply chain is not vulnerable to the shortages of specialized reagents. These raw materials are widely available from multiple global suppliers, mitigating the risk of single-source dependency. The robustness of the chemical steps, characterized by wide operating windows and tolerance to minor variations, ensures consistent production schedules. This reliability is critical for maintaining the continuity of supply for downstream drug manufacturers who operate on just-in-time inventory models and cannot afford disruptions in their API supply lines.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing reaction conditions that are easily replicated in large-scale stainless steel reactors. The workup procedures involve aqueous washes and phase separations, which are standard operations in any GMP facility, facilitating easy technology transfer. From an environmental perspective, the reduction in step count and the elimination of heavy metal catalysts result in a lower E-factor (mass of waste per mass of product). The ability to recover and recycle solvents like tetrahydrofuran and dichloromethane further enhances the sustainability profile of the process, helping manufacturers meet increasingly stringent environmental regulations and corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Allylestrenol using this specific methodology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the key advantages of this Allylestrenol synthesis route compared to traditional fermentation methods?
A: This chemical synthesis route offers significantly higher selectivity and yield, particularly in the initial protection steps where yields reach 99% and 97%. Unlike fermentation routes which can suffer from biological variability and complex downstream processing, this method utilizes straightforward crystallization and filtration, ensuring consistent quality and easier industrial scale-up.
Q: How does the Birch reduction step impact the purity profile of the intermediate?
A: The Birch reduction step effectively eliminates the C3 thioketal protecting group while simultaneously establishing the correct unsaturation pattern in the A-ring. This dual functionality reduces the total number of synthetic steps required, minimizing the accumulation of impurities that often occur in longer multi-step sequences, thereby enhancing the overall purity of the final API intermediate.
Q: Is this manufacturing process suitable for large-scale commercial production?
A: Yes, the process is explicitly designed for industrial applicability. It avoids expensive transition metal catalysts and utilizes common reagents like lithium, ammonia, and standard Grignard reagents. The workup procedures involve simple aqueous washes and recrystallization, which are highly adaptable to multi-tonnage reactor systems, ensuring reliable supply continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Allylestrenol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality hormonal intermediates in the global healthcare landscape. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in patent CN102225960A can be executed with precision and consistency. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the identity and potency of every batch. Our commitment to quality assurance means that our Allylestrenol meets the demanding requirements of international pharmacopoeias, providing our partners with the confidence they need to advance their drug development programs.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to optimize their supply chains. By leveraging our expertise in steroid chemistry, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Together, we can ensure a stable, cost-effective, and high-quality supply of Allylestrenol, supporting the delivery of vital therapies to patients worldwide.
