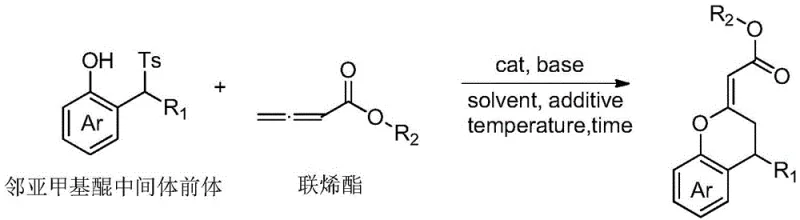
Advanced Catalytic Synthesis of Chiral Chromene Derivatives for Commercial Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing complex chiral scaffolds, particularly those with proven biological significance. Patent CN108129440B introduces a groundbreaking approach for the synthesis of chiral chromene derivatives, utilizing a micromolecular catalyst derived from cinchona alkaloids to catalyze the asymmetric nucleophilic addition and cyclization of dienyl esters on stable o-methylenequinone intermediates. This technology represents a significant leap forward in asymmetric organocatalysis, offering a pathway to optically active chromene derivatives that possess potential pharmacological activities without the reliance on heavy metal catalysts. The method is characterized by its operational simplicity, mild reaction conditions, and the ability to achieve exceptional enantiomeric excess values, making it a highly attractive candidate for the commercial production of high-purity pharmaceutical intermediates. By leveraging this novel catalytic system, manufacturers can access a versatile library of chiral building blocks essential for drug discovery and development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of chiral chromene skeletons has relied heavily on transition metal catalysis or enzymatic processes, which often present substantial challenges for large-scale industrial application. Metal-catalyzed routes frequently require stringent exclusion of air and moisture, expensive ligands, and complex downstream processing to remove trace metal residues that are strictly regulated in pharmaceutical products. Furthermore, many conventional methods suffer from limited substrate scope, requiring specific protecting groups or harsh reaction conditions that can compromise the integrity of sensitive functional groups within the molecule. These limitations not only inflate the overall cost of goods but also introduce significant supply chain risks due to the reliance on scarce precious metals and specialized reagents. Consequently, there is a critical industry demand for alternative synthetic strategies that can deliver high stereocontrol while simplifying the manufacturing workflow and reducing environmental impact.
The Novel Approach
The innovative methodology described in the patent data overcomes these historical barriers by employing a small molecule organocatalyst derived from naturally abundant cinchona alkaloids. This approach facilitates the asymmetric nucleophilic addition and cyclization reaction under remarkably mild conditions, typically ranging from minus 20 degrees Celsius to room temperature, thereby preserving the stability of diverse functional groups. The use of a stable o-methylenequinone intermediate precursor allows for a highly controlled reaction environment that minimizes side reactions and byproduct formation. Unlike metal-based systems, this organocatalytic route does not necessitate rigorous inert atmosphere techniques for every step, significantly lowering the barrier to entry for commercial scale-up. The result is a streamlined process that delivers chiral chromene derivatives with outstanding optical purity and yield, providing a sustainable and economically viable solution for the production of complex API intermediates.
Mechanistic Insights into Cinchona Alkaloid-Catalyzed Cyclization
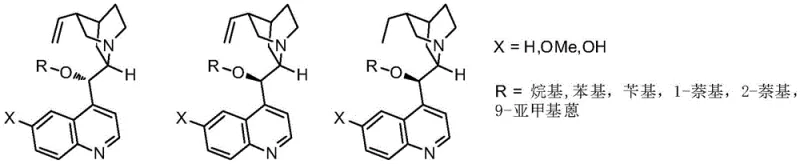
The core of this technological advancement lies in the unique activation mode provided by the cinchona alkaloid-derived catalyst, which orchestrates the stereochemical outcome of the reaction through a well-defined transition state. The catalyst functions by activating the dienyl ester through hydrogen bonding or ion-pairing interactions, while simultaneously stabilizing the o-methylenequinone intermediate to facilitate the nucleophilic attack. This dual activation strategy ensures that the addition occurs with high facial selectivity, leading to the formation of the desired enantiomer with minimal racemization. The structural diversity of the catalyst library, as illustrated in the patent figures, allows for fine-tuning of the steric and electronic properties to match specific substrate requirements. This level of mechanistic control is crucial for R&D teams aiming to optimize impurity profiles and ensure consistent batch-to-batch reproducibility in a GMP manufacturing setting.
Furthermore, the reaction mechanism inherently supports excellent impurity control, which is a paramount concern for regulatory compliance in the pharmaceutical sector. By avoiding transition metals, the process eliminates the risk of metal leaching, a common issue that often requires additional scavenging steps and validation protocols. The mild basic conditions employed, utilizing common inorganic bases like potassium carbonate or sodium carbonate, prevent the degradation of acid-sensitive moieties that might be present in advanced intermediates. The reaction proceeds through a concerted addition-cyclization sequence that efficiently constructs the benzopyran skeleton in a single operation, reducing the number of isolation steps and potential points of failure. This efficiency translates directly into higher overall throughput and reduced waste generation, aligning with the principles of green chemistry and sustainable manufacturing practices.
How to Synthesize Chiral Chromene Derivatives Efficiently
To implement this synthesis effectively, the process begins with the preparation of the reaction vessel under standard laboratory conditions, where the cinchona alkaloid-derived catalyst is introduced at a loading of 1 to 30 mol percent relative to the substrate. The o-methylenequinone intermediate precursor and the dienyl ester are then added in a molar ratio ranging from 2:1 to 5:1, dissolved in an appropriate organic solvent such as chlorobenzene or dichloromethane. An inorganic base, such as potassium carbonate, is added to facilitate the reaction, along with optional additives like molecular sieves to manage water content. The mixture is stirred at temperatures between minus 20 degrees Celsius and room temperature for a duration of 6 to 72 hours, allowing the asymmetric transformation to reach completion. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction system by dissolving the o-methylenequinone intermediate precursor and dienyl ester in an organic solvent such as chlorobenzene.
- Add the cinchona alkaloid-derived small molecule catalyst and an inorganic base like potassium carbonate to the mixture under nitrogen protection.
- Stir the reaction at temperatures ranging from -20°C to room temperature for 6 to 72 hours, then purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this synthesis route offers compelling advantages that directly address the pain points of cost volatility and supply chain fragility associated with traditional metal-catalyzed processes. The elimination of expensive transition metal catalysts and specialized ligands results in a drastic reduction in raw material costs, while the use of commodity chemicals for bases and solvents ensures stable pricing and ready availability. The simplicity of the workup procedure, which often involves direct solvent removal and column chromatography without complex metal scavenging, significantly shortens the production cycle time. This efficiency allows for faster turnaround on custom synthesis requests and improves the overall agility of the supply chain in responding to market demands for key pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The removal of precious metal catalysts eliminates the need for costly recovery systems and extensive testing for residual metals, leading to substantial operational savings. Additionally, the high yield and selectivity of the reaction minimize the loss of valuable starting materials, further optimizing the cost structure. The ability to use simpler catalysts that are either commercially available or easily synthesized reduces dependency on specialized suppliers, creating a more resilient and cost-effective procurement strategy for long-term production campaigns.
- Enhanced Supply Chain Reliability: By relying on robust organocatalysts and common reagents, the manufacturing process becomes less susceptible to disruptions caused by the scarcity of rare earth metals or complex ligands. The mild reaction conditions reduce the energy consumption and equipment stress, extending the lifespan of manufacturing assets and ensuring consistent output. This reliability is critical for maintaining continuous supply to downstream partners, reducing the risk of stockouts and ensuring that project timelines for drug development are met without delay.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively from milligram to multi-kilogram scales without loss of performance. The absence of heavy metals simplifies waste treatment and disposal, ensuring compliance with increasingly stringent environmental regulations. This eco-friendly profile not only reduces disposal costs but also enhances the corporate sustainability image, which is becoming a key factor in supplier selection criteria for major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the feasibility and advantages of adopting this method for industrial applications. Understanding these details is essential for technical teams evaluating the route for potential integration into their existing manufacturing platforms.
Q: What are the primary advantages of this cinchona alkaloid catalyzed method over traditional metal catalysis?
A: This method eliminates the need for expensive and toxic transition metal catalysts, significantly reducing downstream purification costs and environmental hazards while maintaining high enantioselectivity up to 99% ee.
Q: Is the catalyst used in this synthesis commercially available or easy to prepare?
A: The catalyst is derived from readily available cinchona alkaloids and can be either purchased directly or synthesized through a simple, scalable procedure involving quinine derivatives and alkyl chlorides.
Q: What represents the typical yield and purity profile for these chiral chromene derivatives?
A: The patented process consistently delivers high yields often exceeding 90% with exceptional enantiomeric excess values reaching up to 99%, ensuring suitability for stringent pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Chromene Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating innovative academic research into reliable commercial reality. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent CN108129440B can be seamlessly transferred to your supply chain. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of chiral chromene derivatives meets the highest standards required for pharmaceutical applications. Our commitment to quality and consistency makes us the ideal partner for organizations seeking to secure a stable supply of high-value intermediates.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this organocatalytic method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability to support your drug development goals with precision and reliability.
