Advanced Organofluorine-Mediated Synthesis of 2,3-Unsaturated Glycosides for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing complex carbohydrate architectures, particularly 2,3-unsaturated glycosides, which serve as pivotal chiral precursors for bioactive molecules such as heparin, rutin, and various antiviral agents. A groundbreaking approach detailed in patent CN114736250A introduces a novel synthetic pathway mediated by organofluorine solvents, addressing long-standing challenges in glycosidic bond formation. This technology leverages a synergistic system comprising specific perfluorinated solvents and trace amounts of cation exchange resin to achieve exceptional yields and stereoselectivity. For R&D directors and procurement specialists, this represents a significant shift away from traditional heavy metal catalysis towards a more sustainable, cost-effective, and scalable manufacturing paradigm. The method effectively resolves the inherent contradiction in oxoglycosidic bond synthesis where acidic conditions typically favor leaving group departure but simultaneously suppress nucleophilicity, offering a streamlined solution for producing high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,3-unsaturated glycosides via Ferrier rearrangement has relied heavily on two categories of catalysts, both presenting substantial drawbacks for large-scale commercial production. The first category involves heavy metal catalysts such as Scandium triflate, Titanium tetrachloride, or Gold chloride, which not only drive up raw material costs but also introduce severe risks of heavy metal residue in the final active pharmaceutical ingredients (APIs), necessitating expensive and complex purification steps to meet regulatory standards. The second category utilizes strong Lewis acids like Boron trifluoride etherate or Trimethylsilyl triflate, which are notoriously sensitive to moisture and oxygen, requiring stringent anhydrous conditions that complicate reactor operations and increase safety hazards. Furthermore, the strong acidity required for these traditional methods often limits the substrate scope, degrading acid-sensitive functional groups and leading to inconsistent yields. This fundamental chemical contradiction—where the acidity needed to expel the C-3 substituent inadvertently reduces the nucleophilicity of the attacking alcohol—has long plagued the efficiency of oxoglycosidation reactions, creating a bottleneck for reliable API intermediate supplier networks.
The Novel Approach
The innovative methodology disclosed in the patent circumvents these historical limitations by employing a unique organofluorine-mediated system that decouples the acidity requirement from nucleophile suppression. By utilizing perfluorohexane (PFH) as both solvent and mediator alongside a sulfonic acid cation exchange resin, the reaction achieves a delicate balance where the C-F…H interactions enhance the nucleophilicity of the alcohol oxygen while the resin facilitates the departure of the C-3 acetoxy group. This dual-action mechanism allows the reaction to proceed efficiently at moderate temperatures (80-120°C) without the need for stoichiometric amounts of harsh Lewis acids or toxic heavy metals. As illustrated in the general reaction scheme below, this approach consistently delivers products with exclusive alpha-stereoselectivity and yields exceeding 90%, providing a robust platform for cost reduction in pharmaceutical intermediate manufacturing.

Mechanistic Insights into Organofluorine-Mediated Catalysis
The core innovation of this technology lies in the specific molecular interactions facilitated by the organofluorine environment, which fundamentally alters the reaction kinetics compared to conventional polar solvents. Spectroscopic evidence, including NMR analysis, confirms that the fluorine atoms in the solvent engage in weak hydrogen bonding interactions (C-F…H) with the hydroxyl protons of the alcohol nucleophile. This interaction effectively increases the electron density on the oxygen atom, thereby boosting its nucleophilicity even in the presence of the acidic resin required to activate the glycal donor. Simultaneously, the proton transfer mechanism mediated by the solid resin promotes the formation of the allylic cation at the C-1 position by assisting the departure of the C-3 substituent. This synergistic effect resolves the traditional paradox of glycosylation, allowing for high-efficiency bond formation under mild conditions. The result is a highly controlled reaction environment that prevents side reactions and ensures the formation of a single stereoisomer, which is critical for the biological activity of downstream drug candidates.
Furthermore, the versatility of this catalytic system is demonstrated by its broad substrate tolerance, accommodating a wide range of alcohol nucleophiles including primary, secondary, cyclic, and even unsaturated alcohols without compromising yield or selectivity. The data indicates that whether the nucleophile is ethanol, cyclohexanol, or propargyl alcohol, the reaction maintains high efficiency and exclusive alpha-selectivity, suggesting a universal applicability for synthesizing diverse glycoside libraries. This broad scope is essential for medicinal chemists exploring structure-activity relationships (SAR) who require rapid access to varied analogues. The ability to handle diverse functional groups without protection-deprotection sequences further simplifies the synthetic route, reducing the overall step count and improving the atom economy of the process. Such mechanistic robustness ensures that the commercial scale-up of complex pharmaceutical intermediates remains feasible and predictable.
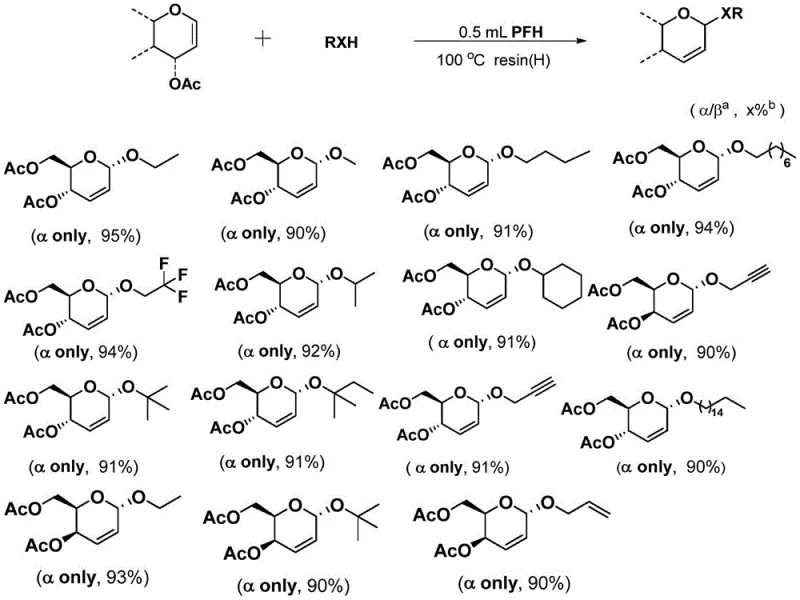
How to Synthesize 2,3-Unsaturated Glycosides Efficiently
The operational simplicity of this organofluorine-mediated protocol makes it highly attractive for translation from laboratory discovery to pilot plant production. The standard procedure involves mixing the triacetylated glucal donor and the desired alcohol acceptor in perfluorohexane, followed by the addition of a catalytic amount of cation exchange resin. The mixture is then heated under inert atmosphere, typically reaching completion within 6 to 12 hours depending on the specific substrate. Upon completion, the workup is remarkably straightforward: the reaction mixture is cooled, and a common organic solvent like ethyl acetate is added to induce phase separation. The product resides in the upper organic layer, while the fluorinated solvent and resin remain in the lower phase or can be easily separated, allowing for immediate recovery and reuse. Detailed standardized synthesis steps for implementing this green chemistry protocol are provided in the guide below.
- Combine triacetylated glucal and the alcohol nucleophile in perfluorohexane (PFH) solvent under inert gas protection.
- Add a trace amount of sulfonic acid cation exchange resin (0.6 wt%) to the reaction mixture.
- Heat the mixture to 100°C for 6 hours, then cool, extract with ethyl acetate, and separate the organic phase to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this organofluorine-mediated technology offers tangible benefits in terms of cost structure, supply reliability, and environmental compliance. By eliminating the dependence on scarce and expensive heavy metal catalysts, manufacturers can significantly reduce raw material expenditures and avoid the volatility associated with precious metal markets. Moreover, the removal of heavy metals from the process stream drastically simplifies downstream purification, removing the need for specialized scavenging resins or extensive chromatography, which translates directly into reduced processing time and lower utility costs. The simplified workup procedure, relying on simple phase separation rather than complex aqueous quenches, minimizes wastewater generation and reduces the burden on waste treatment facilities, aligning with increasingly strict global environmental regulations.
- Cost Reduction in Manufacturing: The elimination of stoichiometric heavy metal catalysts and harsh Lewis acids removes a major cost driver from the bill of materials, while the simplified purification process reduces solvent consumption and labor hours. The ability to operate without stringent anhydrous conditions further lowers infrastructure costs by reducing the need for specialized drying equipment and inert gas consumption. Additionally, the high atom economy and minimal byproduct formation mean that less raw material is wasted, maximizing the output per batch and improving the overall return on investment for production campaigns.
- Enhanced Supply Chain Reliability: The robustness of the cation exchange resin and the stability of the organofluorine solvent ensure consistent batch-to-batch quality, reducing the risk of production delays caused by catalyst deactivation or sensitivity to ambient conditions. The broad substrate scope allows for flexible sourcing of alcohol starting materials, mitigating supply chain bottlenecks associated with specialized reagents. Furthermore, the high yield and selectivity reduce the need for reprocessing off-spec material, ensuring a steady and predictable flow of high-purity intermediates to downstream formulation teams.
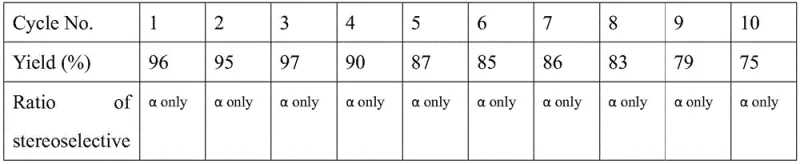
- Scalability and Environmental Compliance: The heterogeneous nature of the resin catalyst and the distinct phase behavior of the fluorinated solvent make this process inherently scalable, facilitating easy transition from gram-scale optimization to multi-ton commercial production. The recyclability of the solvent and catalyst system, demonstrated to maintain efficiency over multiple cycles, significantly reduces the volume of hazardous waste generated per kilogram of product. This green chemistry profile not only lowers disposal costs but also enhances the corporate sustainability metrics, which are increasingly important for securing contracts with major multinational pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this organofluorine-mediated synthesis technology. These insights are derived directly from the experimental data and comparative studies presented in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this method for their specific product pipelines.
Q: How does the organofluorine solvent improve stereoselectivity in glycosylation?
A: The organofluorine solvent creates a unique C-F…H interaction environment that enhances the nucleophilicity of the alcohol oxygen while the resin facilitates the leaving group departure, resulting in exclusive alpha-stereoselectivity.
Q: Can the catalytic system be recycled for industrial production?
A: Yes, the patent demonstrates that the organofluorine solvent and resin system can be recycled for over 10 cycles with maintained high yields and stereoselectivity, significantly reducing waste.
Q: What are the advantages over traditional heavy metal catalysts?
A: This method eliminates the need for expensive and toxic heavy metals like Scandium or Gold, avoiding metal residue issues in pharmaceutical intermediates and simplifying purification processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Unsaturated Glycosides Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this organofluorine-mediated technology for the production of high-value carbohydrate derivatives. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless and efficient. Our state-of-the-art facilities are equipped to handle fluorinated solvents safely and efficiently, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest industry standards. We are committed to leveraging this advanced synthetic route to deliver superior quality intermediates that accelerate your drug development timelines.
We invite you to collaborate with us to explore how this innovative chemistry can optimize your specific manufacturing needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your current production challenges, demonstrating exactly how this metal-free approach can improve your margins. Please contact our technical procurement team today to request specific COA data for our glycoside portfolio and to discuss route feasibility assessments for your next project, ensuring a secure and competitive supply chain for your critical pharmaceutical ingredients.
