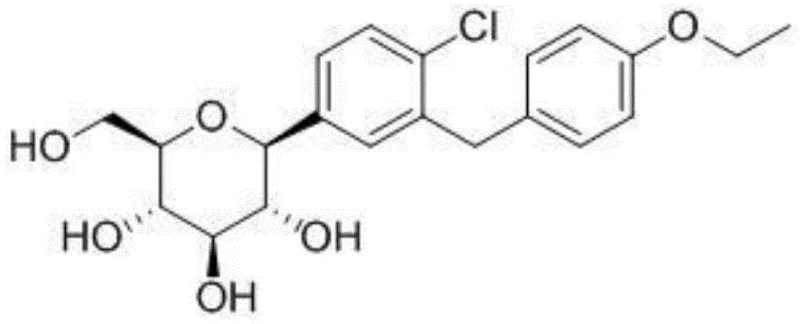
Advanced Synthetic Route for 5-Bromo-2-chloro-4'-ethoxy Diphenylmethane via Protected Intermediates
Introduction to Next-Generation Dapagliflozin Intermediate Synthesis
The global demand for SGLT2 inhibitors, particularly dapagliflozin, has necessitated the development of robust, high-yielding synthetic routes for their key intermediates. Patent CN110922305A, published in March 2020, introduces a transformative methodology for synthesizing 5-bromo-2-chloro-4'-ethoxy diphenylmethane, a critical building block in the dapagliflozin value chain. This innovation addresses long-standing challenges in regioselectivity and impurity control that have plagued earlier generations of synthetic chemistry. By leveraging a strategic protecting group approach, the disclosed method ensures that the final intermediate meets the stringent purity specifications required for modern pharmaceutical manufacturing. For R&D directors and procurement specialists alike, understanding this technological shift is vital for securing a reliable supply of high-quality API precursors.
The significance of this patent lies in its ability to bypass the formation of structural isomers that are notoriously difficult to separate. In the context of complex small molecule synthesis, the presence of even trace amounts of ortho-substituted impurities can compromise the safety profile of the final drug substance. The inventors have ingeniously utilized a nitro-functionalized benzyl ether moiety to direct the electrophilic aromatic substitution exclusively to the desired para position. This level of control not only enhances the chemical purity of the intermediate but also streamlines the entire production workflow, reducing the need for resource-intensive recrystallization or chromatographic steps. As the industry moves towards greener and more efficient processes, such methodological advancements represent the gold standard for pharmaceutical intermediate production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
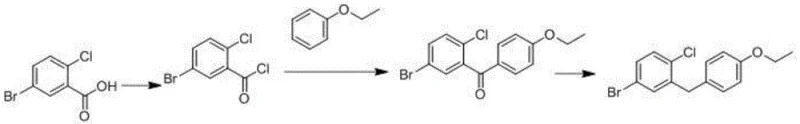
Historically, the synthesis of 5-bromo-2-chloro-4'-ethoxy diphenylmethane relied on the direct Friedel-Crafts acylation of phenetole (ethoxybenzene) with 5-bromo-2-chlorobenzoyl chloride. While conceptually straightforward, this traditional pathway suffers from a fundamental flaw in regioselectivity. The ethoxy group is an ortho/para-directing activator, meaning the acylation reaction inevitably produces a mixture of the desired para-isomer and the unwanted ortho-isomer, specifically 4-bromo-1-chloro-2'-ethoxy diphenylmethane. Separating these two isomers is chemically arduous due to their similar physical properties, often requiring multiple recrystallizations or preparative HPLC, which drastically reduces overall yield and increases manufacturing costs.
Furthermore, the persistence of these ortho-impurities poses a significant risk to the quality of the final Active Pharmaceutical Ingredient (API). If carried through subsequent glycosylation steps, these impurities can lead to the formation of structurally related by-products in dapagliflozin itself, complicating regulatory approval and batch release. The inability to completely suppress the formation of the ortho-side product in conventional routes creates a bottleneck in the supply chain, forcing manufacturers to accept lower yields or invest heavily in purification infrastructure. This inefficiency highlights the urgent need for a synthetic strategy that inherently prevents the formation of these problematic isomers rather than attempting to remove them post-reaction.
The Novel Approach
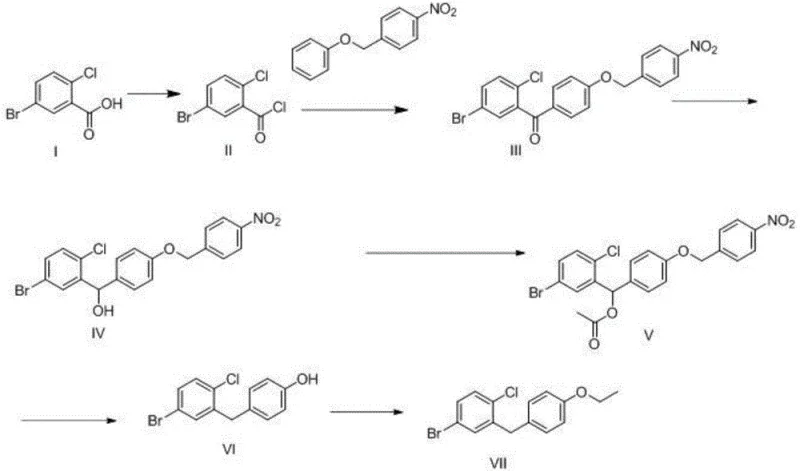
The innovative route disclosed in CN110922305A circumvents these issues by employing a masked phenol strategy. Instead of using phenetole directly, the synthesis utilizes 1-nitro-4-(phenoxymethyl)benzene as the nucleophilic partner. The introduction of the p-nitrobenzyl group serves a dual purpose: it acts as a robust protecting group for the phenol and exerts a directing influence that favors para-substitution while suppressing ortho-attack. The acylation of this protected substrate with 5-bromo-2-chlorobenzoyl chloride proceeds with high regioselectivity, yielding the ketone intermediate almost exclusively in the desired configuration. This fundamental change in the synthetic logic eliminates the root cause of the impurity problem.
Following the acylation, the process employs a streamlined sequence of reduction, acetylation, and hydrogenation to unveil the final structure. A key feature of this novel approach is the efficiency of the hydrogenation step, which simultaneously reduces the benzylic carbonyl functionality to a methylene bridge and cleaves the p-nitrobenzyl protecting group to regenerate the free phenol. This telescoped transformation reduces the number of unit operations and minimizes solvent waste. Finally, a simple ethylation step installs the required ethoxy group. The result is a process that delivers the target intermediate with exceptional purity and yield, offering a commercially superior alternative to legacy methods for any organization seeking cost reduction in API manufacturing.
Mechanistic Insights into Regioselective Acylation and Hydrogenolysis
The core of this technological advancement lies in the electronic and steric modulation of the aromatic ring during the Friedel-Crafts acylation. In the conventional route, the ethoxy group activates the ring strongly, leading to kinetic mixtures. In the patented method, the substrate 1-nitro-4-(phenoxymethyl)benzene presents a phenoxy ring that is less activated than phenetole, yet still sufficiently nucleophilic for acylation in the presence of a Lewis acid like aluminum trichloride (AlCl3). The bulky p-nitrobenzyl ether moiety at the oxygen atom likely imposes steric constraints that disfavor attack at the ortho positions adjacent to the oxygen. Furthermore, the electron-withdrawing nature of the distant nitro group (transmitted through the methylene bridge) may subtly tune the reactivity of the ring, preventing over-reaction or polysubstitution. This precise control ensures that the electrophilic attack by the 5-bromo-2-chlorobenzoyl cation occurs predominantly at the para position.
The subsequent transformation from the ketone to the final diphenylmethane involves a sophisticated cascade of redox reactions. Initially, sodium borohydride reduces the ketone to a secondary alcohol, which is immediately protected as an acetate using acetyl chloride. This acetylation is crucial as it converts the hydroxyl group into a better leaving group for the subsequent hydrogenolysis. When subjected to hydrogen gas over a palladium on carbon (Pd/C) catalyst, the molecule undergoes hydrogenolysis at two distinct sites. First, the benzylic C-O bond of the acetate is cleaved and reduced to a C-H bond, forming the methylene bridge characteristic of the diphenylmethane scaffold. Concurrently, the benzylic C-O bond connecting the phenol to the p-nitrobenzyl group is cleaved. This dual reactivity is highly advantageous, as it achieves two synthetic goals—reduction and deprotection—in a single operational step, thereby enhancing the overall atom economy and process efficiency.
From an impurity control perspective, this mechanism is self-cleansing. Any minor amounts of ortho-isomer that might theoretically form during the initial acylation would possess a different steric environment, potentially affecting the rate of the subsequent reduction or hydrogenolysis steps, allowing for easier separation at the alcohol or acetate stage if necessary. However, the primary benefit is the prevention of the ortho-isomer at the source. The final ethylation step using iodoethane and potassium carbonate is a standard Williamson ether synthesis, which proceeds cleanly on the phenolic substrate. The robustness of this final step ensures that the high purity achieved in the earlier stages is maintained in the final product, delivering a material that is ready for the demanding glycosylation reactions required to synthesize dapagliflozin.
How to Synthesize 5-Bromo-2-chloro-4'-ethoxy Diphenylmethane Efficiently
The execution of this synthetic route requires careful attention to temperature control and reagent stoichiometry to maximize the benefits of the novel chemistry. The process begins with the activation of the benzoic acid, followed by the critical acylation step which sets the stereochemical and regiochemical trajectory of the synthesis. Operators must ensure that the Lewis acid catalyst is properly activated and that the addition of the aromatic substrate is controlled to manage the exotherm. The subsequent reduction and hydrogenation steps require an inert atmosphere to prevent catalyst poisoning and ensure safety. The following guide outlines the standardized operational parameters derived from the patent examples, providing a roadmap for laboratory scale-up and pilot plant implementation.
- Convert 5-bromo-2-chlorobenzoic acid to its acid chloride using thionyl chloride at 50°C, followed by Friedel-Crafts acylation with 1-nitro-4-(phenoxymethyl)benzene.
- Reduce the resulting ketone with sodium borohydride, acetylate the alcohol, and perform catalytic hydrogenation to simultaneously reduce the benzylic position and deprotect the phenol.
- Complete the synthesis by ethylating the phenolic hydroxyl group using iodoethane and potassium carbonate to yield the final diphenylmethane intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical elegance. The primary advantage is the drastic simplification of the purification train. By eliminating the formation of the stubborn ortho-isomer, manufacturers can avoid the costly and time-consuming chromatographic separations or repeated recrystallizations associated with older methods. This directly translates to shorter cycle times and higher throughput, allowing suppliers to respond more agilely to market demand fluctuations. The reliability of supply is significantly enhanced because the process is less prone to batch failures caused by out-of-specification impurity profiles, ensuring a consistent flow of material to downstream API producers.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by yield optimization and waste reduction. With reported yields reaching as high as 96% in the final ethylation step and overall high efficiency in the preceding steps, the consumption of raw materials per kilogram of product is minimized. Furthermore, the elimination of expensive purification resins or large volumes of solvents for chromatography lowers the variable cost of goods sold (COGS). The use of commodity chemicals like thionyl chloride, aluminum trichloride, and sodium borohydride ensures that raw material costs remain stable and predictable, shielding the supply chain from volatility associated with exotic reagents.
- Enhanced Supply Chain Reliability: The robustness of the chemistry contributes to a more resilient supply chain. The reaction conditions are mild, typically ranging from 0°C to 50°C, which reduces the energy load on manufacturing facilities and minimizes the risk of thermal runaways or safety incidents. The intermediates generated, such as the acetylated alcohol, are stable and can be isolated if necessary, providing flexibility in production scheduling. This stability allows for the strategic stocking of key intermediates, buffering the supply chain against disruptions in the availability of starting materials like 5-bromo-2-chlorobenzoic acid.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, the process is well-suited for commercial scale-up of complex pharmaceutical intermediates. The hydrogenation step, while requiring pressure equipment, is a standard unit operation in fine chemical plants and generates minimal waste compared to stoichiometric reduction methods. The avoidance of heavy metal catalysts in the acylation step (using AlCl3 instead) simplifies wastewater treatment and reduces the burden of heavy metal clearance testing in the final API. This alignment with green chemistry principles facilitates regulatory compliance and supports the sustainability goals of modern pharmaceutical enterprises.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. These insights are derived directly from the experimental data and claims presented in the patent literature, offering clarity on the practical aspects of adopting this route. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer and for commercial teams negotiating supply agreements.
Q: How does this new synthetic route improve purity compared to conventional methods?
A: Conventional methods acylate phenetole directly, which generates difficult-to-remove ortho-substituted impurities. This patented route uses a p-nitrobenzyl protecting group strategy that sterically and electronically directs acylation to the para position, effectively eliminating the ortho-isomer and simplifying downstream purification.
Q: What are the critical reaction conditions for the hydrogenation step?
A: The process utilizes palladium on carbon (Pd/C) as a catalyst under a hydrogen atmosphere. This single step is highly efficient as it accomplishes both the reduction of the benzylic acetate/alcohol to a methylene bridge and the cleavage of the p-nitrobenzyl ether protecting group to reveal the free phenol.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process operates under mild conditions (0-50°C) and uses standard reagents like thionyl chloride, aluminum trichloride, and sodium borohydride. The high yields reported (up to 96% in the final step) and the avoidance of complex chromatographic purifications make it highly scalable for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Bromo-2-chloro-4'-ethoxy Diphenylmethane Supplier
At NINGBO INNO PHARMCHEM, we recognize that the quality of the final drug product is inextricably linked to the purity of its intermediates. Our technical team has thoroughly analyzed the methodology described in CN110922305A and has integrated these advanced principles into our own manufacturing platforms. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. Our facilities are equipped with rigorous QC labs capable of detecting trace impurities at ppm levels, guaranteeing that every batch of 5-bromo-2-chloro-4'-ethoxy diphenylmethane meets stringent purity specifications required by global regulatory bodies.
We invite pharmaceutical partners to collaborate with us to leverage this superior synthetic route for their dapagliflozin programs. By choosing NINGBO INNO PHARMCHEM, you gain access to a supply chain that prioritizes both quality and cost-efficiency. We encourage you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our implementation of this patented technology can optimize your supply chain and reduce your overall manufacturing costs.
