Advanced Synthesis Of TBCC: Technical Upgrades For Commercial Scale Production
The global demand for efficient low-temperature bleaching agents in the textile and detergent industries has driven significant innovation in the synthesis of oxygen bleaching activators. A pivotal advancement in this sector is detailed in patent CN102863383A, which outlines a novel preparation method for N-[4-(triethylaminomethyl)benzoyl]caprolactam chloride, commonly known as TBCC. This compound represents a critical fine chemical intermediate that enables effective bleaching at reduced temperatures, thereby preserving fiber integrity and reducing energy consumption during industrial washing processes. The technical breakthrough described in this patent addresses long-standing inefficiencies in traditional manufacturing routes by substituting expensive and hazardous precursors with cost-effective, widely available commodity chemicals. By leveraging a streamlined four-step synthetic pathway, this method not only enhances the economic viability of TBCC production but also aligns with modern environmental standards by minimizing toxic byproduct generation. For R&D directors and procurement specialists, understanding the nuances of this synthesis is essential for securing a reliable supply chain of high-performance textile auxiliaries.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of TBCC and similar cationic bleach activators has been constrained by reliance on sophisticated and costly starting materials that complicate the supply chain. Prior art methods, such as those described in patents WO95/29160 and US5686015, typically utilize 4-chloromethylbenzoyl chloride as a key precursor, a reagent that commands a high market price due to its complex preparation requirements. Furthermore, these legacy processes often necessitate the use of gaseous ethyl halides for the final quaternization step, introducing severe operational hazards and requiring specialized pressure-rated equipment to manage gas handling safely. The evolution of hydrogen chloride gas as a byproduct during these reactions poses additional challenges for waste management and corrosion control, increasing the overall capital expenditure for manufacturing facilities. Consequently, the combination of high raw material costs, complex engineering requirements, and safety risks has resulted in elevated market prices for TBCC, limiting its widespread adoption in cost-sensitive applications like mass-market detergents.
The Novel Approach
In stark contrast to these cumbersome legacy protocols, the methodology disclosed in patent CN102863383A introduces a paradigm shift by utilizing 4-methylbenzoic acid, caprolactam, and triethylamine as the foundational building blocks. This strategic selection of raw materials leverages the abundant availability and low cost of 4-methylbenzoic acid, effectively decoupling the production cost from the volatility of specialty acid chloride markets. The process replaces the hazardous gaseous alkylation step with a liquid-phase quaternization using triethylamine, which can be conducted under standard atmospheric reflux conditions, drastically simplifying the reactor design and operational protocol. By transforming the synthesis into a series of manageable liquid-solid reactions, the new approach mitigates the risks associated with toxic gas release and allows for more precise control over reaction parameters. This transition not only lowers the barrier to entry for manufacturers but also ensures a more stable and predictable production timeline, which is crucial for maintaining continuity in the supply of high-purity textile chemical intermediates.
Mechanistic Insights into Multi-Step Functionalization and Quaternization
The core of this innovative synthesis lies in a meticulously orchestrated sequence of functional group transformations that build molecular complexity with high fidelity. The process initiates with the radical bromination of 4-methylbenzoic acid using N-bromosuccinimide (NBS) and benzoyl peroxide (BPO) as an initiator, selectively functionalizing the benzylic position to yield 4-bromomethylbenzoic acid. This intermediate subsequently undergoes hydrolysis under acidic reflux conditions to convert the bromide into a hydroxymethyl group, generating 4-hydroxymethylbenzoic acid with excellent conversion rates. The resulting alcohol is then activated via chlorination using thionyl chloride, forming the reactive 4-chloromethylbenzoyl chloride species in situ, which immediately reacts with caprolactam in the presence of a base to form the amide linkage. This step-wise construction ensures that each functional group is introduced with high regioselectivity, minimizing the formation of structural isomers that could compromise the bleaching efficacy of the final product.
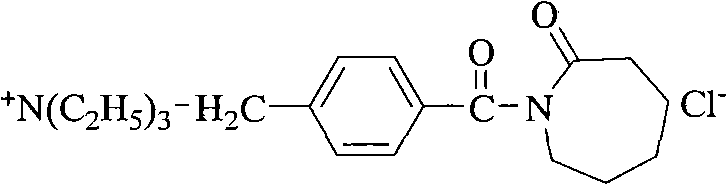
Following the formation of the amide backbone, the final critical transformation involves the quaternization of the benzylic chloride with triethylamine to generate the cationic TBCC structure. This nucleophilic substitution reaction proceeds efficiently in polar aprotic solvents like acetonitrile, driving the formation of the quaternary ammonium salt which is essential for the molecule's solubility and activity in aqueous bleaching baths. From an impurity control perspective, the ability to isolate and purify intermediates such as 4-hydroxymethylbenzoic acid and the chloromethylbenzoyl caprolactam precursor allows manufacturers to strip away side products before they propagate through the synthesis. This modular approach to purification ensures that the final TBCC product meets stringent purity specifications required for sensitive textile applications, where residual impurities could lead to fabric discoloration or reduced wash performance. The mechanistic robustness of this route provides a solid foundation for scaling up production while maintaining consistent quality attributes batch after batch.
How to Synthesize TBCC Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and temperature control across the four distinct reaction stages to maximize overall yield. The process begins with the bromination step where precise molar ratios of NBS and catalyst are maintained to prevent over-bromination, followed by a controlled hydrolysis to ensure complete conversion to the alcohol. Subsequent activation with thionyl chloride must be managed to avoid excess reagent carryover, which could interfere with the delicate acylation with caprolactam. For a comprehensive breakdown of the specific reaction conditions, solvent volumes, and workup procedures required to achieve the reported yields, please refer to the standardized synthesis guide below.
- Bromination of 4-methylbenzoic acid using NBS and BPO to form 4-bromomethylbenzoic acid.
- Hydrolysis of the bromo-intermediate to generate 4-hydroxymethylbenzoic acid.
- Chlorination with thionyl chloride followed by acylation with caprolactam.
- Final quaternization reaction with triethylamine to yield TBCC.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthesis route presents a compelling value proposition centered on cost stability and operational simplicity. By shifting the raw material base from expensive specialty chlorides to commodity organic acids, manufacturers can significantly insulate their production costs from the price volatility often seen in the fine chemical sector. The elimination of gaseous reagents not only reduces the need for complex safety infrastructure but also streamlines the logistics of raw material intake, as all inputs can be handled as liquids or solids using standard drum or tank storage systems. This simplification of the input supply chain enhances reliability, ensuring that production schedules are less likely to be disrupted by the availability issues that frequently plague hazardous gas suppliers. Furthermore, the environmentally benign nature of the process reduces the burden on waste treatment facilities, potentially lowering regulatory compliance costs and facilitating smoother permitting for capacity expansions.
- Cost Reduction in Manufacturing: The substitution of high-cost 4-chloromethylbenzoyl chloride with inexpensive 4-methylbenzoic acid fundamentally alters the cost structure of TBCC production. This raw material swap removes a significant cost driver, allowing for substantial margin improvement or more competitive pricing strategies in the marketplace. Additionally, the use of standard reflux conditions eliminates the energy penalties associated with high-pressure gas reactions, further contributing to lower utility expenses per kilogram of product. The cumulative effect of these efficiencies is a manufacturing process that is inherently more economical, providing a buffer against market fluctuations and enabling long-term price stability for downstream customers.
- Enhanced Supply Chain Reliability: Sourcing 4-methylbenzoic acid, caprolactam, and triethylamine is significantly less risky than procuring specialized acid chlorides or gaseous alkylating agents, as these are high-volume commodity chemicals produced by multiple global vendors. This diversification of the supplier base mitigates the risk of single-source bottlenecks, ensuring that production lines can remain operational even if one vendor faces disruptions. The liquid-phase nature of the entire synthesis also simplifies transportation and storage logistics, removing the need for specialized pressurized transport containers and reducing the administrative burden of handling hazardous materials. Consequently, the supply chain becomes more resilient and responsive to changes in demand, supporting just-in-time delivery models for major detergent and textile manufacturers.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing unit operations such as filtration, reflux, and recrystallization that are standard in multi-ton chemical plants. The absence of toxic gas emissions simplifies the environmental permitting process and reduces the capital investment required for scrubbing and abatement systems. This alignment with green chemistry principles not only future-proofs the manufacturing asset against tightening environmental regulations but also enhances the brand reputation of the end-product as a sustainable textile solution. The ability to scale from pilot batches to commercial tonnage without fundamental process changes ensures a smooth technology transfer and rapid time-to-market for new production capacity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of TBCC synthesized via this advanced route. These insights are derived directly from the experimental data and process descriptions found in the underlying patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this intermediate into their existing formulations or supply networks.
Q: What are the primary advantages of the new TBCC synthesis route over conventional methods?
A: The novel route utilizes inexpensive and widely available raw materials like 4-methylbenzoic acid instead of costly 4-chloromethylbenzoyl chloride. It eliminates the need for handling gaseous ethyl halides, significantly improving operational safety and reducing equipment complexity.
Q: How does this process impact the environmental profile of bleaching activator manufacturing?
A: The process is designed to be environmentally friendly by avoiding the release of harmful hydrogen chloride gas associated with older methods. The use of standard reflux conditions and common solvents simplifies waste treatment and aligns with stricter green chemistry regulations.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the methodology relies on mature, conventional reaction steps such as bromination, hydrolysis, and acylation which are easily scalable. The total yield reaching up to 63% demonstrates robust efficiency suitable for commercial manufacturing volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable TBCC Supplier
As the textile and detergent industries continue to evolve towards more sustainable and efficient processing methods, the demand for high-performance activators like TBCC is poised for growth. NINGBO INNO PHARMCHEM stands ready to support this transition by leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of this synthesis, ensuring stringent purity specifications and rigorous QC labs verify every batch against international standards. We understand that consistency is key for formulators, and our commitment to quality assurance guarantees that the TBCC supplied meets the exact performance profiles required for low-temperature bleaching applications.
We invite potential partners to engage with our technical team to discuss how this optimized synthesis route can benefit your specific product portfolio. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this supply source. We encourage you to contact our technical procurement team today to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring a seamless integration of this critical intermediate into your global supply chain.
