Advanced Synthesis of 5α-Androst-7-Methyl Intermediates for High-Purity Steroid Manufacturing
Introduction to High-Efficiency Steroid Intermediate Synthesis
The pharmaceutical industry constantly seeks robust pathways for synthesizing complex steroidal scaffolds, particularly those functionalized at the C7 position, which are critical precursors for active pharmaceutical ingredients like Tibolone. Patent CN100478350C introduces a groundbreaking methodology for the preparation of 5α-androst-7-methyl-3-hydroxy-17-one and its subsequent reduction to 5α-androst-7-methyl-3,17-diol. This technology addresses long-standing challenges in steroid chemistry by replacing low-yielding conjugate addition methods with a high-efficiency sequence involving catalytic hydrogenation and Wittig olefination. For R&D directors and procurement specialists, this patent represents a significant leap forward in process reliability, offering a route that consistently delivers high purity and superior yields compared to historical benchmarks. The ability to access these 7-methylated intermediates with such precision opens new avenues for cost-effective manufacturing of hormonal therapies and cardiovascular treatments.
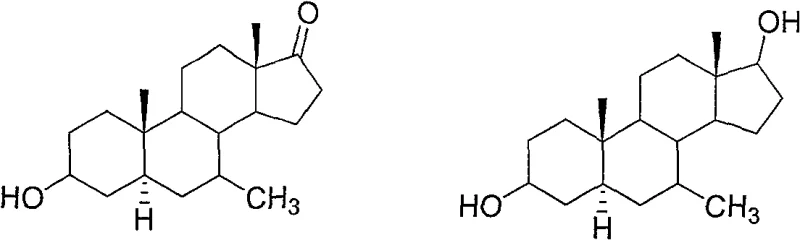
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, introducing a methyl group at the C7 position of the steroid nucleus has been a formidable synthetic challenge characterized by poor atom economy and low conversion rates. Prior art, such as the work by Grunwell and Ni, relied on CuCl-catalyzed 1,6-conjugate addition of methyl Grignard reagents to androst-4,6-dien-3-ones. While conceptually sound, this approach suffered from catastrophic yield losses, often capping at merely 20% efficiency, which renders it commercially unviable for large-scale production. Furthermore, alternative methods reported by Zheng involving triethylsilane reductions of 7-keto intermediates managed only marginal improvements, with total yields hovering around 40%. These conventional routes not only waste valuable starting materials but also generate complex impurity profiles that require extensive and costly purification steps, thereby inflating the final cost of goods and extending lead times for API manufacturers.
The Novel Approach
In stark contrast, the methodology disclosed in CN100478350C employs a sophisticated four-step sequence that dramatically enhances overall throughput. By utilizing an androst-5-ene-3-acetoxy-7-one-17-ethylene ketal starting material, the process leverages a strategic Wittig reaction to install the C7 functionality as an exocyclic methylene group, which is subsequently reduced to the desired methyl group. This indirect approach bypasses the steric and electronic hurdles associated with direct alkylation at the crowded C7 position. The result is a step-wise yield profile where individual transformations consistently exceed 90% efficiency, culminating in a final product yield that is multiples higher than traditional Grignard-based protocols. This novel pathway not only simplifies the operational workflow but also ensures a much cleaner reaction profile, facilitating easier downstream processing and higher final purity.
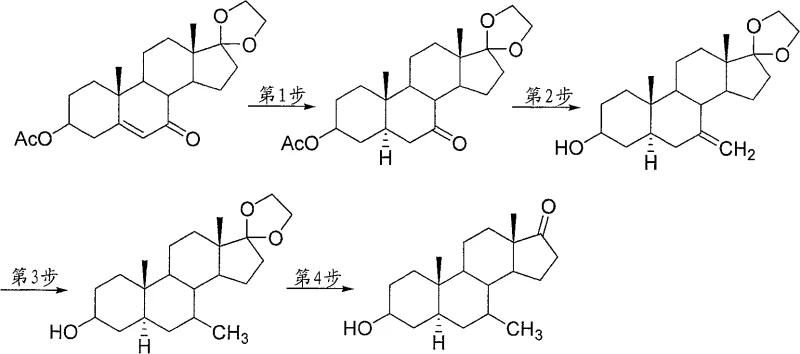
Mechanistic Insights into Catalytic Hydrogenation and Wittig Olefination
The core of this technological advancement lies in the precise orchestration of catalytic hydrogenation and carbon-carbon bond-forming reactions. The process initiates with the catalytic hydrogenation of the C5-C6 double bond using 5% Pd/C in ethyl acetate. This step is critical not only for saturating the A/B ring junction but also for establishing the requisite 5α-stereochemistry, which is essential for the biological activity of the downstream derivatives. The use of palladium on carbon ensures high selectivity, minimizing over-reduction or isomerization side reactions. Following this, the Wittig reaction utilizes methylenetriphenylphosphorane, generated in situ from methyltriphenylphosphonium bromide and n-butyllithium, to attack the C7 ketone. This transformation effectively converts the carbonyl oxygen into an exocyclic double bond (=CH2), setting the stage for the final methylation. The subsequent hydrogenation of this exocyclic alkene is highly efficient, converting the methylene group into a stable methyl substituent without affecting other sensitive functionalities like the C3-hydroxyl or the C17-ketal protection group.
Impurity control is inherently built into this mechanistic design. By protecting the C17 ketone as an ethylene ketal throughout the C7 functionalization steps, the process prevents unwanted side reactions at the D-ring, which is a common pitfall in steroid synthesis. The deprotection step, utilizing p-toluenesulfonic acid in acetone, is mild yet effective, cleanly revealing the C17 ketone without epimerization or degradation of the newly installed 7-methyl group. This orthogonal protection strategy ensures that the final impurity profile is dominated by easily removable byproducts rather than structurally similar steroid analogs. For quality control teams, this means that meeting stringent pharmacopeial standards becomes a manageable task rather than a bottleneck, ensuring that the high-purity pharmaceutical intermediate reaches the market with minimal risk of regulatory rejection.
How to Synthesize 5α-Androst-7-Methyl-3-Hydroxy-17-One Efficiently
Implementing this synthesis requires careful attention to reaction conditions, particularly during the Wittig olefination and the final deprotection stages. The protocol outlined in the patent provides a robust framework that balances reactivity with selectivity, making it accessible for both pilot-scale and commercial manufacturing environments. Operators must ensure anhydrous conditions during the generation of the phosphorus ylide to maximize the conversion of the C7 ketone. Furthermore, the recycling of the Pd/C catalyst in the hydrogenation steps offers an immediate opportunity for cost optimization and waste reduction. For a detailed breakdown of the specific reagent quantities, temperature controls, and workup procedures required to replicate these high yields, please refer to the standardized operating procedure below.
- Perform catalytic hydrogenation of the starting androst-5-ene derivative using Pd/C to saturate the C5-C6 double bond with 94.5% yield.
- Execute a Wittig reaction using methylenetriphenylphosphorane to introduce the exocyclic methylene group at the C7 position.
- Conduct a second catalytic hydrogenation to reduce the C7-methylene group to a C7-methyl group, followed by acid-catalyzed deprotection of the C17-ketal.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this patented synthesis route offers transformative benefits that extend far beyond simple yield improvements. The shift from low-efficiency Grignard additions to this high-yielding catalytic sequence fundamentally alters the cost structure of producing 7-methyl steroids. By drastically reducing the amount of starting material required to produce a kilogram of API intermediate, manufacturers can achieve substantial cost savings on raw material procurement. Additionally, the use of heterogeneous catalysts like Pd/C, which can be recovered and reused, eliminates the need for expensive stoichiometric metal reagents and the associated heavy metal waste disposal costs. This aligns perfectly with modern green chemistry initiatives and reduces the environmental compliance burden on production facilities.
- Cost Reduction in Manufacturing: The elimination of low-yielding steps directly correlates to a lower cost per unit of output. Since the process avoids the use of cryogenic conditions and exotic reagents, the operational expenditure (OpEx) related to energy consumption and specialized equipment is significantly minimized. The high selectivity of the reactions reduces the load on purification units such as chromatography columns, allowing for faster batch turnover and lower solvent consumption. These factors combine to create a leaner, more cost-effective manufacturing process that enhances profit margins for reliable pharmaceutical intermediate suppliers.
- Enhanced Supply Chain Reliability: The robustness of this chemical route ensures consistent batch-to-batch quality, which is paramount for maintaining uninterrupted supply lines to downstream API manufacturers. The starting materials, such as androst-5-ene derivatives, are commercially available and stable, reducing the risk of supply disruptions caused by scarce precursors. Furthermore, the simplified workflow reduces the total processing time, enabling manufacturers to respond more agilely to fluctuations in market demand. This reliability is crucial for securing long-term contracts with major pharmaceutical companies that prioritize supply continuity.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard unit operations like filtration, extraction, and distillation that are easily transferable from the laboratory to multi-ton reactors. The avoidance of toxic heavy metal catalysts (like copper in older methods) and the ability to recycle palladium catalysts significantly reduce the hazardous waste footprint. This facilitates smoother regulatory approvals and lowers the cost of waste treatment, making the commercial scale-up of complex steroid intermediates both economically and environmentally sustainable.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing production portfolios.
Q: What is the primary advantage of this synthesis route over traditional Grignard methods?
A: Unlike traditional CuCl-catalyzed Grignard additions which suffer from low yields (around 20%), this patented route utilizes a Wittig strategy combined with catalytic hydrogenation to achieve step yields exceeding 90%, significantly improving overall process efficiency.
Q: How is stereochemical control at the 5α-position achieved?
A: Stereocontrol is managed during the initial catalytic hydrogenation step using Pd/C, which selectively reduces the C5-C6 double bond to favor the thermodynamically stable 5α-configuration, ensuring the correct backbone for downstream biological activity.
Q: Can this process be scaled for commercial production of Tibolone precursors?
A: Yes, the process avoids cryogenic conditions and uses standard heterogeneous catalysts (Pd/C) and common solvents (Ethyl Acetate, THF), making it highly suitable for scale-up from kilogram to multi-ton commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5α-Androst-7-Methyl-3-Hydroxy-17-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent to plant is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify every batch. By leveraging the advanced synthesis techniques described in CN100478350C, we can offer our partners a competitive edge through superior product quality and consistent availability.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains for steroid-based therapeutics. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your project goals and accelerate your time to market.
