Advanced Manufacturing of Sertaconazole Nitrate: A Safer, Scalable Route for Global Supply Chains
Advanced Manufacturing of Sertaconazole Nitrate: A Safer, Scalable Route for Global Supply Chains
The pharmaceutical industry constantly seeks robust manufacturing pathways that balance high purity with operational safety, particularly for potent antifungal agents like Sertaconazole Nitrate. Patent CN1974567B introduces a transformative preparation method that fundamentally alters the synthetic landscape for this critical active pharmaceutical ingredient (API). By shifting away from hazardous hydride bases and carcinogenic solvents, this technology offers a streamlined approach that utilizes common alkaline conditions in alcohol-organic solvent mixtures. This innovation not only simplifies the operational workflow but also addresses critical environmental and safety concerns inherent in legacy processes. For global procurement teams and R&D directors, understanding this shift is vital for securing a reliable sertaconazole nitrate supplier capable of meeting stringent regulatory and volume demands without compromising on safety standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Sertaconazole Nitrate, as described in earlier patents such as EP151477 and US5135943, relied heavily on aggressive and dangerous reagents that posed significant logistical and safety challenges. The traditional route necessitated the use of sodium hydride (NaH) as a base, a substance known for its pyrophoric nature, requiring strictly anhydrous and oxygen-free environments under inert gas protection. Furthermore, these processes utilized hexamethylphosphoric triamide (HMPA) as a solvent, a chemical flagged for its carcinogenic properties and high cost, making recovery and waste disposal economically and environmentally burdensome. The downstream processing was equally cumbersome, often requiring silica gel column chromatography to isolate the free base before salt formation, a step that drastically reduces throughput and increases solvent consumption. These factors collectively rendered conventional methods unsuitable for modern, large-scale industrial applications where safety and efficiency are paramount.
The Novel Approach
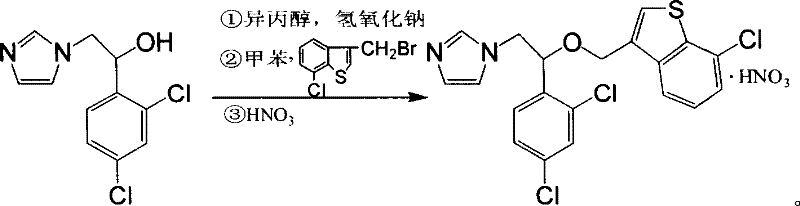
In stark contrast, the methodology disclosed in CN1974567B presents a remarkably温和 (mild) and operationally simple alternative that bypasses the pitfalls of its predecessors. This novel approach employs sodium hydroxide or potassium hydroxide in a mixed solvent system of isopropanol and an organic solvent like toluene, eliminating the need for hazardous sodium hydride and toxic HMPA entirely. The reaction proceeds through a reflux stage followed by a room temperature coupling, which significantly reduces energy consumption and thermal stress on the reactants. Crucially, the process allows for direct salt formation with nitric acid after a simple aqueous workup, completely removing the need for tedious silica gel purification steps. This transition represents a major leap forward in cost reduction in pharmaceutical intermediates manufacturing, enabling producers to achieve high-purity outputs with a fraction of the operational complexity and risk associated with older technologies.
Mechanistic Insights into Alkali-Catalyzed Etherification
The core of this improved synthesis lies in the efficient generation of the nucleophilic alkoxide species under controlled alkaline conditions. In the initial step, 1-(2,4-dichlorophenyl)-2-(1-imidazolyl)ethanol is treated with sodium hydroxide in isopropanol under reflux. This step effectively deprotonates the hydroxyl group to form the corresponding alkoxide, which serves as a potent nucleophile for the subsequent substitution reaction. Unlike the violent and uncontrollable deprotonation seen with sodium hydride, the use of alkali hydroxides in protic solvents provides a buffered environment that minimizes side reactions and degradation of the sensitive imidazole ring. Following the formation of the alkoxide, the introduction of 3-bromomethyl-7-chlorobenzo[b]thiophene in toluene facilitates a smooth nucleophilic substitution (SN2 mechanism) at the benzylic position. This biphasic or mixed-solvent system ensures excellent solubility for both the ionic intermediate and the organic halide, driving the reaction to completion with high selectivity.
Furthermore, the mechanistic pathway inherently supports superior impurity control, a critical factor for R&D directors focused on quality. The mild reaction conditions prevent the formation of colored polymeric byproducts that typically plague high-temperature phase transfer catalysis methods, which often yield reddish-brown crude products difficult to decolorize. By maintaining the reaction at room temperature during the coupling phase, the process preserves the structural integrity of the chlorinated aromatic systems, preventing dehalogenation or other thermal decomposition pathways. The final salt formation with nitric acid is conducted in a way that promotes the crystallization of the nitrate salt directly from the organic phase, effectively excluding non-basic impurities. This intrinsic purity advantage means that the commercial scale-up of complex azole derivatives can be achieved with fewer recrystallization cycles, thereby improving overall yield and reducing the environmental footprint of the manufacturing process.
How to Synthesize Sertaconazole Nitrate Efficiently
Implementing this synthesis route requires precise control over stoichiometry and phase separation to maximize the benefits of the patented method. The process begins with the activation of the imidazole alcohol component, followed by the strategic addition of the thiophene electrophile, ensuring that the reaction kinetics favor the desired ether linkage. Operators must adhere to the specified reflux times to ensure complete deprotonation before cooling the mixture for the coupling step, which is allowed to proceed overnight to ensure full conversion without thermal input. The detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and workup procedures derived from the patent examples, are outlined below to guide technical teams in replicating this high-efficiency protocol.
- Reflux 1-(2,4-dichlorophenyl)-2-(1-imidazolyl)ethanol with sodium hydroxide in isopropanol for 3-5 hours to generate the alkoxide intermediate.
- Cool the mixture to room temperature and add a toluene solution of 3-bromomethyl-7-chlorobenzo[b]thiophene, stirring for 12-15 hours.
- Separate the organic layer, wash with water, and treat with nitric acid to precipitate the final Sertaconazole Nitrate salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route translates into tangible strategic advantages that extend far beyond simple chemical transformation. The elimination of specialized, hazardous reagents like sodium hydride removes a significant bottleneck in raw material sourcing, as sodium hydroxide is a ubiquitous commodity chemical available globally with stable pricing. This shift drastically simplifies the logistics of raw material storage and handling, as there is no longer a need for specialized inert atmosphere equipment or explosion-proof facilities dedicated to hydride handling. Consequently, the barrier to entry for manufacturing this key antifungal intermediate is lowered, fostering a more competitive and resilient supply market that can better withstand regional disruptions or raw material shortages.
- Cost Reduction in Manufacturing: The economic implications of replacing HMPA and silica gel chromatography are profound, leading to substantial cost savings throughout the production lifecycle. HMPA is not only expensive to purchase but also costly to dispose of due to its toxicity, whereas the solvents used in this new method, such as isopropanol and toluene, are inexpensive and easily recoverable through standard distillation. Moreover, the removal of the silica gel column purification step represents a massive reduction in consumable costs and labor hours, as column chromatography is notoriously slow and solvent-intensive on an industrial scale. By streamlining the workflow to a simple extract-and-crystallize process, manufacturers can significantly reduce the cost of goods sold (COGS), allowing for more competitive pricing strategies in the global antifungal market without sacrificing margin.
- Enhanced Supply Chain Reliability: The robustness of this chemical process directly correlates to improved supply chain continuity and reduced lead times for high-purity pharmaceutical intermediates. Because the reaction does not require strict anhydrous or anaerobic conditions, the risk of batch failure due to moisture ingress or equipment leakage is virtually eliminated, ensuring consistent production schedules. The use of common, non-hazardous chemicals also means that supply is less susceptible to regulatory restrictions or transportation bans that often affect dangerous goods like sodium hydride. This reliability allows downstream API manufacturers to plan their production runs with greater confidence, knowing that their intermediate supply is secure and less prone to the delays associated with handling specialized hazardous materials.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method aligns perfectly with modern green chemistry principles and increasingly strict global regulations. The absence of carcinogenic solvents and the reduction in waste generation (by avoiding silica gel and minimizing solvent use) simplify the permitting process for new manufacturing lines and reduce the burden on wastewater treatment facilities. The process is inherently scalable, moving seamlessly from kilogram-level R&D batches to multi-ton commercial production without the need for complex engineering controls required for pyrophoric reagents. This ease of scale-up ensures that suppliers can rapidly respond to surges in market demand for antifungal medications, providing a stable and compliant source of material for long-term partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route for Sertaconazole Nitrate. These answers are derived directly from the comparative data and experimental results presented in the patent documentation, focusing on the practical implications for industrial application. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this newer, safer methodology over established but riskier legacy processes.
Q: Why is the new synthesis route for Sertaconazole Nitrate considered safer than previous methods?
A: The novel process eliminates the use of sodium hydride (NaH), which is highly flammable and requires strict anhydrous/inert conditions, and replaces the carcinogenic solvent HMPA with safer alternatives like isopropanol and toluene.
Q: How does this process improve product purity and decolorization?
A: By avoiding high-temperature phase transfer catalysis which often leads to reddish-brown byproducts, this mild room-temperature coupling followed by acid salt formation yields a product that is significantly easier to decolorize and purify without silica gel chromatography.
Q: Is this method suitable for large-scale commercial production?
A: Yes, the removal of hazardous reagents like NaH and toxic solvents like HMPA, combined with the elimination of complex column chromatography steps, makes this route highly amenable to multi-ton scale manufacturing with reduced operational risks.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sertaconazole Nitrate Supplier
The technological advancements detailed in CN1974567B underscore the potential for safer, more efficient production of critical antifungal intermediates, a capability that NINGBO INNO PHARMCHEM is uniquely positioned to deliver. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this optimized synthesis are fully realized at an industrial level. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Sertaconazole Nitrate meets the highest international pharmacopoeia standards while maintaining the cost and safety advantages of this novel route.
We invite global partners to leverage our technical expertise to secure a stable supply of this vital intermediate. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your supply chain resilience and drive down your overall production costs.
