Advanced Isomerization Technology for High-Purity Chiral Citronellal Production and Commercial Scale-Up
The global demand for high-purity chiral intermediates continues to surge, driven by the pharmaceutical and fine fragrance sectors' relentless pursuit of enantiomerically pure compounds. Patent CN110981707A introduces a groundbreaking methodology for the isomerization of nerol or geraniol into chiral citronellal, addressing critical bottlenecks in traditional synthesis routes. This technology leverages a novel water-soluble metal catalyst system that operates under mild conditions, offering a transformative approach to producing this valuable monoterpene aldehyde. For R&D Directors and Supply Chain Heads, this innovation represents a significant leap forward in process reliability and economic feasibility. By utilizing a water-oil two-phase system, the method ensures that the precious metal catalyst remains in the aqueous layer post-reaction, facilitating effortless separation and extensive recycling capabilities. This not only minimizes heavy metal contamination risks but also drastically reduces the overall cost of goods sold (COGS) associated with noble metal usage. As a reliable Flavor & Fragrance Intermediates supplier, understanding such technological advancements is crucial for maintaining a competitive edge in the market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral citronellal has been plagued by significant technical and economic challenges that hinder large-scale commercial viability. Traditional methods often rely on the asymmetric hydrogenation of neral or geranial, which presents a formidable separation dilemma due to the nearly identical boiling points of these cis-trans isomers. Achieving high optical purity requires extremely pure starting materials, necessitating energy-intensive distillation processes that inflate production costs and reduce overall yield. Furthermore, alternative routes involving myrcene require the use of hazardous reagents like lithium diethylamide, which poses severe safety risks regarding moisture sensitivity and operational complexity. These conventional pathways frequently suffer from low atom economy and generate substantial chemical waste, creating environmental compliance burdens for manufacturing facilities. The reliance on organic solvents for catalyst recovery further complicates the downstream processing, leading to higher solvent consumption and increased VOC emissions. Consequently, procurement teams face volatile pricing and supply instability when sourcing citronellal produced via these outdated and inefficient methodologies.
The Novel Approach
In stark contrast, the innovative method disclosed in CN110981707A utilizes a sophisticated water-soluble chiral metal catalyst to drive the isomerization of nerol or geraniol with exceptional efficiency. This approach eliminates the need for harsh reducing or oxidizing agents, operating instead under mild thermal conditions that preserve the integrity of the sensitive terpene structure. The core breakthrough lies in the design of the catalyst, which incorporates hydrophilic functional groups such as sulfonate or phosphate moieties on the chiral ligands. This modification ensures that the catalyst preferentially partitions into the water phase, while the hydrophobic citronellal product accumulates in the organic oil phase. Such a biphasic system allows for a simple decantation process to separate the product, bypassing the need for complex chromatographic purification or high-vacuum distillation. This streamlined workflow not only enhances safety by avoiding pyrophoric reagents but also significantly improves the environmental profile of the manufacturing process. For stakeholders focused on cost reduction in Pharmaceutical Intermediates manufacturing, this technology offers a sustainable pathway to high-value chiral building blocks.
Mechanistic Insights into Water-Soluble Ruthenium-Catalyzed Isomerization
The catalytic cycle underpinning this transformation is a masterpiece of organometallic chemistry, designed to maximize both activity and stereoselectivity. The active species is generated in situ through the coordination of a metal-diphosphine complex, preferably ruthenium-based, with a chiral diamine ligand in the presence of a base. The water-solubility is ingeniously engineered by selecting ligands such as 5,5'-disulfonate-BINAP or sulfonated diamine derivatives, which impart sufficient hydrophilicity to the metal center without compromising its catalytic performance. During the reaction, the allylic alcohol substrate coordinates to the metal center, undergoing a hydride shift that results in the formation of the chiral aldehyde. The chiral environment created by the binaphthyl backbone of the phosphine ligand dictates the facial selectivity of the reaction, ensuring the predominant formation of the desired (R)-enantiomer. Crucially, the aqueous medium stabilizes the transition state and facilitates proton transfer steps, enhancing the overall turnover number of the catalyst. This mechanistic robustness allows the system to tolerate minor impurities in the feedstock, making it highly suitable for industrial applications where feedstock consistency can vary.
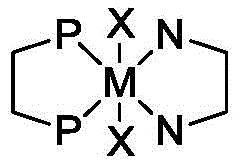
Impurity control is inherently built into this process design, primarily due to the high chemoselectivity of the catalyst towards the allylic alcohol functionality. Unlike non-selective oxidation methods that might over-oxidize the substrate to carboxylic acids or degrade the double bonds, this isomerization preserves the carbon skeleton and the olefinic unsaturation. The mild reaction temperature, typically ranging between 25°C and 80°C, prevents thermal degradation pathways that often lead to polymerization or racemization of the product. Furthermore, the phase separation acts as an intrinsic purification step; water-soluble byproducts or inorganic salts remain in the aqueous phase, while the organic phase contains the crude citronellal with high purity. This reduces the load on subsequent refining columns and minimizes the formation of heavy ends or tars. For Quality Control teams, this translates to a more consistent impurity profile and easier compliance with stringent pharmacopeial standards. The ability to recycle the aqueous catalyst phase multiple times without significant loss of activity further ensures that batch-to-batch variability is minimized, securing a stable supply of high-purity Chiral Citronellal.
How to Synthesize Chiral Citronellal Efficiently
Implementing this synthesis route requires precise attention to the preparation of the catalytic solution and the management of the biphasic reaction system. The process begins with the activation of the catalyst precursors in a polar solvent like ethanol, followed by the introduction of water to create the aqueous catalytic phase. Substrates such as nerol or geraniol are then introduced, and the mixture is agitated under an inert atmosphere to prevent oxidation of the sensitive allylic species. Reaction monitoring is typically conducted via gas chromatography to track conversion and enantiomeric excess, ensuring the endpoint is reached before any potential side reactions occur. The simplicity of the workup procedure is a major advantage, as it requires only settling and phase separation to recover the bulk of the product. Detailed standard operating procedures for scaling this reaction from laboratory to pilot plant are critical for maintaining the delicate balance of the emulsion and ensuring efficient mass transfer between phases.
- Prepare the water-soluble catalyst by coordinating a metal-diphosphine complex with a chiral diamine ligand in ethanol, followed by the addition of water and base.
- Introduce the substrate, nerol or geraniol, into the reactor containing the catalyst solution under an inert nitrogen atmosphere.
- Maintain the reaction mixture at 25-80°C for 3-12 hours, then allow phase separation to isolate the product from the recyclable aqueous catalyst layer.
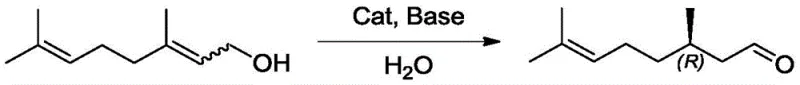
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this patented technology offers substantial advantages that directly impact the bottom line and supply chain resilience. The primary driver of value is the drastic reduction in catalyst consumption costs achieved through the innovative recycling mechanism. Since the catalyst remains in the water phase, it can be recovered and reused for multiple batches without the need for complex regeneration processes, effectively amortizing the cost of expensive noble metals over a much larger production volume. This stands in sharp contrast to homogeneous systems where the catalyst is lost with the product or requires destructive extraction. Additionally, the use of readily available raw materials like nerol and geraniol mitigates supply risk, as these feedstocks are produced on a massive scale for the fragrance industry and are not subject to the same scarcity issues as specialized precursors like pure neral. The mild reaction conditions also translate to lower energy consumption for heating and cooling, contributing to a smaller carbon footprint and reduced utility costs. These factors combined create a robust economic model that supports long-term price stability for buyers.
- Cost Reduction in Manufacturing: The elimination of expensive stoichiometric reagents and the ability to recycle the aqueous catalyst phase significantly lowers the variable cost per kilogram of product. By avoiding the use of hazardous lithium amides or high-pressure hydrogenation equipment, capital expenditure requirements for plant infrastructure are also reduced. The simplified downstream processing reduces solvent usage and waste disposal fees, further enhancing the overall cost efficiency of the manufacturing operation. This economic structure allows suppliers to offer more competitive pricing while maintaining healthy margins, benefiting the entire value chain.
- Enhanced Supply Chain Reliability: Utilizing stable and abundant starting materials ensures that production schedules are not disrupted by raw material shortages. The robustness of the catalytic system against minor feedstock variations means that quality is maintained even if supplier specifications fluctuate slightly. Furthermore, the scalability of the water-oil two-phase system is well-understood in the chemical industry, allowing for rapid capacity expansion to meet surging demand without prolonged engineering delays. This reliability is crucial for pharmaceutical customers who require guaranteed continuity of supply for their critical drug substance manufacturing lines.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste, as the aqueous catalyst stream can be treated or reused rather than disposed of as toxic sludge. Operating at near-atmospheric pressure and moderate temperatures reduces the safety risks associated with high-pressure reactors, simplifying regulatory compliance and insurance costs. The high atom economy of the isomerization reaction means less raw material is wasted as byproducts, aligning with green chemistry principles and corporate sustainability goals. This environmental stewardship enhances the brand reputation of companies adopting this technology and future-proofs their supply chains against tightening environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this isomerization technology. They are derived from the specific beneficial effects and experimental data disclosed in the patent documentation. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this process into their existing manufacturing portfolios. The answers reflect the practical realities of scaling aqueous biphasic catalysis and the specific advantages it holds over traditional organic synthesis methods.
Q: How does the water-soluble catalyst system improve cost efficiency?
A: The catalyst dissolves in the aqueous phase while the product remains in the oil phase, allowing for simple physical separation and multiple reuse cycles without complex purification, significantly lowering noble metal consumption.
Q: What optical purity can be achieved with this isomerization method?
A: The patented process consistently achieves high enantiomeric excess (ee) values, often exceeding 98% for the (R)-configuration, which is critical for downstream menthol synthesis.
Q: Are the raw materials nerol and geraniol readily available for scale-up?
A: Yes, unlike neral or geranial which are difficult to separate, nerol and geraniol are stable, commercially available isomers that facilitate a more robust and continuous supply chain.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Citronellal Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in CN110981707A for the production of high-value fine chemicals. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are successfully translated into robust industrial processes. Our facility is equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, including precise enantiomeric excess measurements critical for chiral intermediates. We understand that consistency is key for our partners, and our manufacturing protocols are designed to deliver batch-after-batch reliability that meets the exacting standards of the global pharmaceutical and fragrance industries. Our commitment to technical excellence ensures that every gram of Chiral Citronellal we supply adheres to the highest quality benchmarks.
We invite you to collaborate with us to leverage this cutting-edge synthesis route for your specific application needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this water-soluble catalyst system for your supply chain. Please contact us to request specific COA data and route feasibility assessments tailored to your volume requirements. By partnering with NINGBO INNO PHARMCHEM, you gain access to not just a product, but a comprehensive technical solution that drives efficiency and value for your organization.
