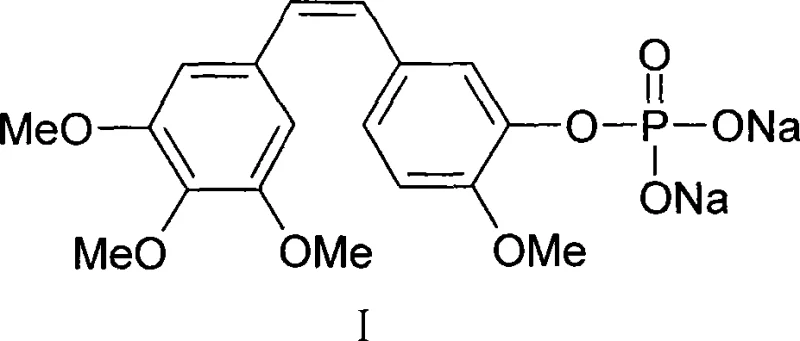
Advanced Phosphorylation Technology for Scalable Combretastatin A-4 Phosphate Production
Advanced Phosphorylation Technology for Scalable Combretastatin A-4 Phosphate Production
The development of potent vascular disrupting agents has placed significant demand on the supply chain for high-quality intermediates, specifically Combretastatin A-4 phosphate disodium salt (CA4P). Patent CN101220054A introduces a transformative preparation method that addresses critical bottlenecks in the synthesis of this vital anti-cancer compound. By shifting away from hazardous solvents and complex purification protocols, this technology enables a more robust and economically viable production pathway. The core innovation lies in the utilization of bis-alkoxyl phosphoryl halides coupled with a DMAP catalytic system, which stabilizes reaction temperatures and drastically reduces processing time. This approach not only enhances the chemical integrity of the final product but also aligns with modern green chemistry principles by eliminating toxic byproducts. For pharmaceutical manufacturers, this represents a pivotal shift towards more sustainable and cost-effective API intermediate sourcing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Combretastatin A-4 phosphate has been plagued by severe operational and environmental challenges that hinder efficient commercial scale-up of complex anti-cancer intermediates. Traditional routes often rely on dibenzyl phosphite reactions requiring trimethylchlorosilane and sodium iodide for deprotection, necessitating the use of highly toxic carbon tetrachloride as a solvent. Other methods employ expensive reagents like bis-(2,2,2-trichloroethyl) phosphoryl compounds, which drive up raw material costs and introduce severe reaction conditions that are difficult to control on a large scale. Furthermore, processes utilizing phosphorus oxychloride often result in excessive byproduct formation due to the high reactivity of the reagent, leading to lower yields and complicated quality control measures. The reliance on ion-exchange resins for purification in some prior art further exacerbates production costs and extends lead times, making these methods unsuitable for the rigorous demands of modern pharmaceutical supply chains.
The Novel Approach
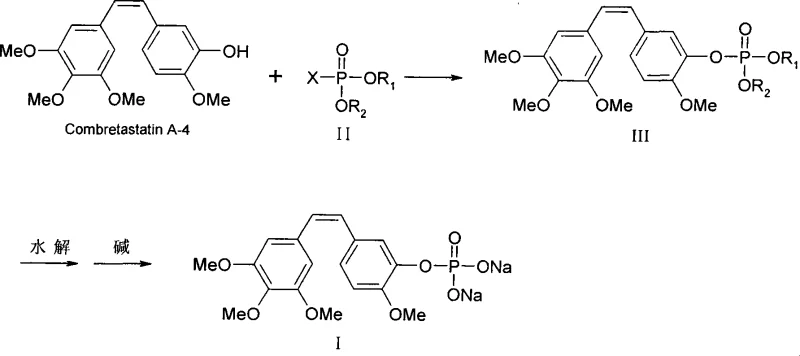
In stark contrast, the novel methodology disclosed in the patent utilizes bis-alkoxyl phosphoryl halides as the primary phosphorylation reagent, which are both inexpensive and readily available on the global market. This route introduces 4-Dimethylaminopyridine (DMAP) as a catalyst, which plays a crucial role in moderating reaction conditions and maintaining a stable temperature profile throughout the synthesis. The reaction proceeds efficiently at mild temperatures ranging from 20°C to 40°C, completing within a short timeframe of approximately 0.5 to 2 hours, thereby significantly increasing throughput capacity. Crucially, the phosphorylated intermediate generated in this process does not require purification before proceeding to the hydrolysis and salification steps, streamlining the workflow and reducing solvent consumption. This streamlined approach ensures high product quality and yield while simplifying the overall operational complexity for manufacturing teams.
Mechanistic Insights into DMAP-Catalyzed Phosphorylation
The mechanistic superiority of this synthesis lies in the nucleophilic catalysis provided by DMAP, which activates the phosphoryl halide towards the phenolic hydroxyl group of Combretastatin A-4. In the absence of such a catalyst, the reaction between the sterically hindered phenol and the phosphoryl halide would require much harsher conditions, potentially leading to the degradation of the sensitive cis-stilbene backbone of the molecule. The DMAP catalyst forms a highly reactive acylammonium intermediate that facilitates the transfer of the phosphoryl group under mild thermal conditions, preserving the stereochemical integrity of the double bond which is critical for biological activity. This controlled activation prevents the formation of poly-phosphorylated byproducts that are common when using more aggressive reagents like phosphorus oxychloride, ensuring a cleaner reaction profile. The stability of the intermediate formed allows for direct progression to the next step without isolation, minimizing exposure to conditions that could induce isomerization or decomposition.
Impurity control is inherently built into this mechanism through the selection of specific alkoxy protecting groups such as tert-butyl or benzyl, which can be cleanly removed under defined hydrolytic or hydrogenolytic conditions. The use of aprotic solvents like dichloromethane or trimethyl phosphate provides a homogeneous reaction medium that ensures consistent mixing and heat transfer, further reducing the risk of localized hot spots that could generate impurities. By avoiding the use of heavy metal scavengers or complex resin beds for purification, the final product exhibits a superior impurity profile with minimal residual metals or organic contaminants. This high level of chemical purity is essential for meeting the stringent regulatory requirements for injectable anti-cancer therapies, where even trace impurities can have significant safety implications. The robustness of this mechanism ensures batch-to-batch consistency, a key metric for reliable pharmaceutical intermediate suppliers.
How to Synthesize Combretastatin A-4 Phosphate Efficiently
The synthesis protocol outlined in the patent offers a standardized pathway for producing CA4P with high efficiency and reproducibility. The process begins with the dissolution of the starting material in a suitable aprotic solvent, followed by the controlled addition of the phosphorylating agent in the presence of the catalyst. Detailed operational parameters regarding stoichiometry, addition rates, and temperature gradients are critical to maximizing yield and minimizing side reactions. The subsequent hydrolysis and salification steps are equally important, requiring precise pH control to ensure complete conversion to the disodium salt without degrading the phosphate ester linkage.
- Dissolve Combretastatin A-4 in an aprotic solvent with an acid scavenger and DMAP catalyst, then add bis-alkoxyl phosphoryl halide at 20-40°C to form the phosphorylated intermediate.
- Hydrolyze the intermediate to remove protecting groups, followed by salification with sodium hydroxide and crystallization using acetone to isolate the final disodium salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology translates into tangible strategic advantages regarding cost structure and supply continuity. The elimination of exotic and expensive reagents such as silane derivatives and sodium iodide directly reduces the bill of materials, while the removal of ion-exchange purification steps lowers consumable costs and waste disposal fees. The simplified workflow reduces the total cycle time per batch, allowing manufacturing facilities to increase their annual output capacity without significant capital investment in new equipment. This efficiency gain is particularly valuable in the context of cost reduction in API manufacturing, where margin pressures are constantly driving the need for leaner production models. Furthermore, the use of common, non-proprietary solvents and reagents mitigates the risk of supply disruptions caused by single-source dependencies, ensuring a more resilient supply chain for critical oncology ingredients.
- Cost Reduction in Manufacturing: The process achieves significant economic optimization by replacing costly deprotection reagents with standard acidic or basic hydrolysis methods that are inexpensive and widely available. By eliminating the need for molecular distillation and complex resin-based purification, the operational expenditure associated with energy consumption and specialized equipment maintenance is drastically lowered. The high yield of the crude intermediate means that less starting material is wasted, improving the overall atom economy of the synthesis. These factors combine to create a substantially lower cost of goods sold (COGS), providing a competitive edge in pricing negotiations for bulk pharmaceutical contracts.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as dichloromethane, acetone, and standard phosphoryl halides ensures that raw material availability is not a bottleneck for production schedules. Unlike processes dependent on custom-synthesized reagents with long lead times, this method allows for rapid replenishment of inventory from multiple global suppliers. The robustness of the reaction conditions means that production is less susceptible to delays caused by minor fluctuations in environmental controls or utility availability. This reliability is crucial for reducing lead time for high-purity anti-cancer intermediates, enabling pharmaceutical partners to maintain consistent inventory levels and meet market demand without interruption.
- Scalability and Environmental Compliance: The mild reaction temperatures and absence of highly toxic solvents like carbon tetrachloride make this process inherently safer and easier to scale from pilot plant to commercial tonnage. The reduced generation of hazardous waste simplifies compliance with increasingly strict environmental regulations, lowering the administrative and financial burden of waste treatment. The straightforward crystallization process for the final product ensures that scale-up does not introduce new purification challenges, maintaining product quality regardless of batch size. This scalability supports the commercial expansion of oncology drug portfolios, allowing partners to confidently plan for long-term market growth.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. Understanding these details is essential for R&D teams evaluating process transfer and procurement teams assessing vendor capabilities. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation.
Q: What are the primary advantages of the DMAP-catalyzed method over traditional phosphorylation routes?
A: The DMAP-catalyzed method operates under mild conditions (20-40°C) compared to the harsh low-temperature or toxic solvent requirements of conventional methods. It eliminates the need for expensive silane reagents and complex ion-exchange purification, resulting in higher yields and simpler operations suitable for industrial scale-up.
Q: How does this process improve the purity profile of Combretastatin A-4 phosphate?
A: By utilizing stable bis-alkoxyl phosphoryl halides and avoiding highly active reagents like phosphorus oxychloride, the formation of side products is significantly minimized. The intermediate does not require purification before the final step, yet the final product achieves high purity through straightforward crystallization.
Q: Is this synthesis route scalable for commercial API production?
A: Yes, the process is designed for industrial suitability. It uses inexpensive, readily available reagents, avoids toxic carbon tetrachloride, and features short reaction times with stable temperature control, making it ideal for large-scale manufacturing of anti-cancer intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Combretastatin A-4 Phosphate Disodium Salt Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the development of life-saving oncology therapies. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to market supply is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Combretastatin A-4 phosphate meets the highest international standards for safety and efficacy. Our commitment to process excellence allows us to deliver high-purity pharmaceutical intermediates that support the complex regulatory filings of our global partners.
We invite you to collaborate with us to leverage this advanced phosphorylation technology for your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your supply chain efficiency and reduce overall project costs.
