Advanced Sulfonated Phosphine Oxide Monomers: Scalable Synthesis for High-Performance Polymer Applications
Advanced Sulfonated Phosphine Oxide Monomers: Scalable Synthesis for High-Performance Polymer Applications
The development of high-performance polymeric materials for demanding applications such as proton exchange membrane fuel cells and advanced water treatment systems relies heavily on the availability of specialized monomers with precise structural integrity. Patent CN101723978B introduces a groundbreaking efficient preparation method for sulfonated phosphine oxide monomers and their salts, specifically targeting the synthesis of bis(4-halophenyl)(3-sulfonic acid phenyl)phosphine oxide derivatives. This technological advancement addresses critical bottlenecks in the manufacturing of polyaryl ether phosphine oxide polymers, offering a route that significantly enhances the utilization rate of sulfur trioxide while drastically simplifying downstream processing. For R&D directors and procurement specialists seeking reliable sulfonated phosphine oxide monomer supplier partnerships, understanding the nuances of this synthesis is vital for securing supply chains for next-generation electronic and energy materials.
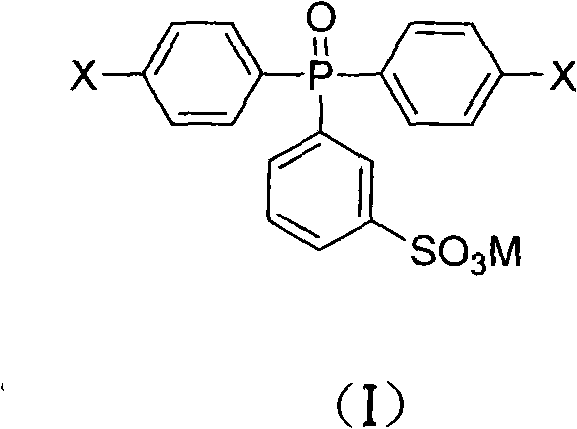
The strategic importance of this patent lies in its ability to produce monomers that facilitate the creation of polymers with superior mechanical properties and proton conductivity compared to traditional sulfonated polyethersulfones. By integrating phosphine oxide structural units, the resulting materials exhibit enhanced thermal stability and chemical resistance, which are paramount for longevity in harsh operational environments. The method described allows for the precise control of sulfonation degrees without compromising the integrity of the phosphine oxide backbone, ensuring that the final polymer exhibits the desired ion exchange capacity. This level of control is essential for manufacturers aiming to produce high-purity fuel cell membrane materials that meet rigorous international performance standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for bis(4-halophenyl)(3-sulfophenyl)phosphine oxide monomers have historically been plagued by inefficiencies that render them economically unviable for large-scale industrial production. Conventional methods typically rely on high concentrations of oleum and often result in a sulfur trioxide utilization rate as low as 20.6%, leading to excessive generation of acidic waste liquid that poses significant environmental and disposal challenges. Furthermore, the purification of the crude product in these legacy processes invariably requires column chromatography, a technique that is notoriously difficult to scale, consumes vast quantities of organic solvents, and extends the overall preparation cycle time considerably. These factors combine to create a high-cost manufacturing profile that limits the commercial availability of these critical intermediates for the broader polymer industry.
The Novel Approach
In stark contrast, the novel approach detailed in the patent data utilizes a optimized molar ratio of raw materials to oleum, specifically between 1:1.5-2.5, coupled with a controlled reaction temperature range of 80-110°C to maximize efficiency. This refined process achieves a sulfur trioxide utilization rate of up to 50.0%, representing a substantial improvement that directly correlates to reduced raw material costs and a smaller environmental footprint. Crucially, the post-treatment workflow has been re-engineered to eliminate the need for column chromatography entirely, relying instead on a sequence of filtration, extraction, and salt precipitation to isolate the product. This shift not only shortens the preparation period but also reduces the volume of organic solvents required, making the process inherently more suitable for the commercial scale-up of complex specialty chemicals.
Mechanistic Insights into Electrophilic Aromatic Sulfonation
The core chemical transformation involves an electrophilic aromatic substitution where the sulfonic acid group is introduced onto the phenyl ring attached to the phosphorus atom. The presence of the phosphine oxide group influences the electron density of the aromatic rings, necessitating precise control over the acidity and temperature of the reaction medium to ensure regioselectivity. By maintaining the oleum concentration within the preferred range of 15-20%, the reaction kinetics are tuned to favor mono-sulfonation at the meta-position relative to the phosphine oxide linkage, avoiding over-sulfonation or degradation of the sensitive phosphorus-oxygen bond. This mechanistic precision is what allows for the high yields observed, often exceeding 90% when calculated based on reacted raw materials, ensuring a consistent impurity profile.
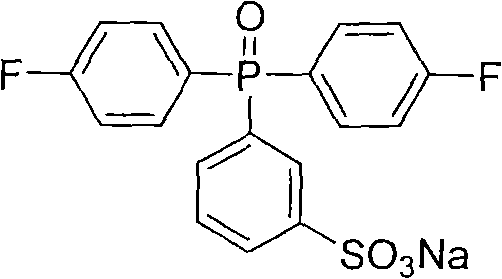
Impurity control is further enhanced by the specific workup procedure which leverages the solubility differences between the sulfonated product and unreacted starting materials. The addition of sodium chloride to the aqueous filtrate induces the precipitation of oily intermediates, which are then extracted with butanone, effectively separating the target monomer from inorganic salts and acidic byproducts. Subsequent neutralization with alkali converts the sulfonic acid into its corresponding salt form, such as the sodium or potassium salt, which exhibits different crystallization properties that facilitate final purification via methanol recrystallization. This multi-stage purification logic ensures that the final white solid product meets stringent purity specifications required for polymerization into high-performance membranes without the need for additional refining steps.
How to Synthesize Bis(4-halophenyl)(3-sulfonic acid phenyl)phosphine Oxide Efficiently
Implementing this synthesis route in a production environment requires strict adherence to the specified reaction parameters to replicate the high efficiency and purity reported in the patent literature. The process begins with the careful mixing of bis(4-halophenyl)phenylphosphine oxide and oleum, followed by a controlled heating phase that must be monitored to prevent thermal runaway while ensuring complete conversion. Detailed standardized synthesis steps see the guide below, which outlines the precise quenching, extraction, and neutralization protocols necessary to achieve the reported 50.0% sulfur trioxide utilization. Operators must be trained to handle the exothermic nature of the sulfonation and the subsequent acid quench safely to maintain batch-to-batch consistency.
- Mix bis(4-halophenyl)phenylphosphine oxide with 15-20% oleum at a molar ratio of 1: 1.5-2.5 and react at 80-110°C for 1.5-5 hours.
- Quench the reaction in ice water, filter solids, extract filtrate with ethyl acetate, and precipitate oily matter using sodium chloride.
- Extract with butanone, neutralize with alkali to pH 7, evaporate solvents, and recrystallize from methanol to obtain the white solid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this optimized synthesis method translates into tangible operational benefits that extend beyond simple chemical yield improvements. The elimination of column chromatography removes a major bottleneck in production throughput, allowing for continuous or larger batch processing that significantly enhances supply chain reliability for critical polymer intermediates. By reducing the dependency on large volumes of organic solvents and minimizing acidic waste generation, manufacturers can achieve substantial cost savings in both raw material procurement and waste disposal compliance, aligning with modern green chemistry initiatives. This process robustness ensures that lead times for high-purity polymer intermediates can be reduced, providing a competitive edge in markets where speed to market is crucial.
- Cost Reduction in Manufacturing: The dramatic increase in sulfur trioxide utilization means that less raw oleum is required per kilogram of finished product, directly lowering the variable cost of goods sold. Additionally, the removal of the column chromatography step eliminates the capital and operational expenses associated with silica gel, solvent recovery, and the extended labor hours typically needed for such purification. These efficiencies compound to offer a much more attractive cost structure for cost reduction in polymer synthesis additives manufacturing, allowing buyers to negotiate better pricing tiers without sacrificing quality.
- Enhanced Supply Chain Reliability: Simplified processing equipment requirements mean that production can be scaled across multiple facilities with lower technical barriers, reducing the risk of single-point failures in the supply network. The use of common industrial solvents like ethyl acetate and butanone, rather than specialized chromatographic eluents, ensures that raw material sourcing remains stable even during global supply fluctuations. This resilience is critical for reducing lead time for high-purity polymer intermediates, ensuring that downstream polymer producers receive their monomers on schedule to maintain their own production schedules.
- Scalability and Environmental Compliance: The reduction in acidic waste liquid and organic solvent usage simplifies the environmental permitting process for new production lines, accelerating the time required to bring new capacity online. The process is inherently designed for large-scale industrial production, meaning that technology transfer from lab to plant is straightforward with minimal re-engineering of the core chemistry. This scalability supports the growing demand for advanced materials in the energy sector, ensuring a steady supply of monomers needed for the commercial scale-up of complex specialty chemicals like fuel cell membranes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these sulfonated phosphine oxide monomers. The answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to ensure accuracy for engineering teams. Understanding these details helps stakeholders evaluate the feasibility of integrating these materials into their existing product portfolios.
Q: How does this new method improve sulfur trioxide utilization compared to conventional sulfonation?
A: The optimized process increases sulfur trioxide utilization from approximately 20.6% in traditional methods to 50.0%, significantly reducing acidic waste generation and raw material consumption.
Q: Is column chromatography required for purifying these sulfonated monomers?
A: No, the novel purification protocol eliminates the need for column chromatography by utilizing salt precipitation and solvent extraction, making it highly suitable for large-scale industrial manufacturing.
Q: What specific cation forms of the sulfonated phosphine oxide monomer can be produced?
A: The process is versatile enough to produce various salt forms including lithium, sodium, potassium, cesium, and ammonium salts, as well as the free acid form, depending on the neutralization base used.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bis(4-halophenyl)(3-sulfonic acid phenyl)phosphine Oxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality monomers play in the development of next-generation functional polymers and energy materials. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from R&D to full-scale manufacturing is seamless and efficient. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of sulfonated phosphine oxide monomer meets the exacting standards required for high-performance applications in fuel cells and water treatment membranes.
We invite you to collaborate with our technical team to explore how this optimized synthesis route can benefit your specific project requirements. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your supply chain goals and drive innovation in your product development pipeline.
