Advanced Copper-Catalyzed Synthesis of Optically Active Menthol for Industrial Scale-Up
Advanced Copper-Catalyzed Synthesis of Optically Active Menthol for Industrial Scale-Up
The global demand for high-purity optically active menthol continues to surge across the flavor, fragrance, and pharmaceutical sectors, driving the need for more efficient and cost-effective manufacturing technologies. Patent CN112573996B introduces a groundbreaking four-step synthetic route that addresses the critical limitations of traditional extraction and noble-metal catalysis. This novel methodology leverages a copper-salt chiral phosphine catalyst system to achieve high enantioselectivity starting from readily available precursors like methyl 5-methyl-4-hexenoate. For R&D directors and procurement leaders, this technology represents a paradigm shift towards sustainable, scalable, and economically viable production of L-menthol, eliminating the reliance on volatile agricultural supplies and expensive rhodium complexes.
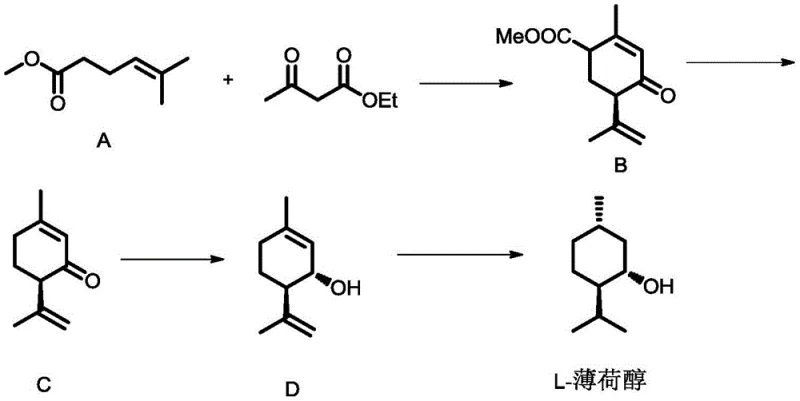
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industry has relied heavily on two primary sources for optically active menthol: natural extraction from mint leaves and chemical synthesis using expensive noble metal catalysts. Natural extraction is plagued by inherent inefficiencies, including low yields dependent on crop seasons, complex purification processes to remove uncertain impurities, and significant environmental waste generation. On the synthetic side, established methods such as the Takasago process utilize rhodium-BINAP complexes which, while effective, involve prohibitively high catalyst costs and critical challenges in catalyst recovery and recycling. Furthermore, alternative routes like the hydrogenation of thymol often result in complex mixtures of diastereoisomers requiring energy-intensive separation and resolution steps, ultimately depressing overall process yields and increasing the cost of goods sold.
The Novel Approach
The patented methodology offers a robust alternative by utilizing a copper-based catalytic system that is both economically accessible and highly selective. The process initiates with the cyclization of methyl 5-methyl-4-hexenoate and ethyl acetoacetate, facilitated by a chiral copper-phosphine complex, to generate a key intermediate with high optical purity. A distinct advantage of this route is the integration of a crystallization purification step early in the sequence, which effectively upgrades the enantiomeric excess of the intermediate before subsequent transformations. This strategic design minimizes the propagation of impurities and allows for the use of milder reaction conditions throughout the decarboxylation, reduction, and final hydrogenation steps, making it exceptionally suitable for large-scale industrial application without the technical bottlenecks associated with noble metal recovery.
Mechanistic Insights into Copper-Catalyzed Asymmetric Cyclization
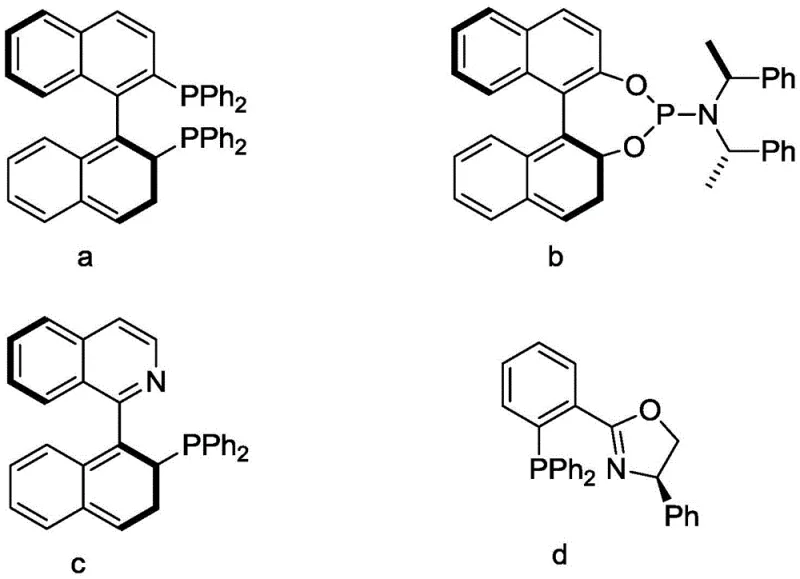
The cornerstone of this synthesis is the enantioselective cyclization in the first step, which establishes the chiral center essential for the final L-menthol configuration. The reaction employs a metal precursor, preferably copper sulfate pentahydrate, coordinated with specific chiral phosphine ligands. As illustrated in the ligand structures below, the steric and electronic properties of ligands such as structure d play a pivotal role in inducing asymmetry during the ring-closing reaction. The interaction between the copper center and the chiral ligand creates a defined chiral pocket that directs the approach of the substrates, ensuring the formation of the desired enantiomer with high fidelity. Optimization data indicates that a molar ratio of ligand to copper between 0.5:1 and 2:1 yields the best balance of activity and selectivity, achieving conversion rates exceeding 93% with selectivity up to 99%.
Following the initial cyclization, the process incorporates a sophisticated impurity control mechanism through solution crystallization. By carefully selecting solvents such as ethyl acetate and controlling the cooling profile from 90°C down to 20°C, the process effectively purifies the intermediate compound B. This step is critical for R&D teams focused on impurity profiles, as it elevates the optical purity from approximately 95% ee to over 99.9% ee in a single pass. Subsequent steps, including base-mediated decarboxylation and hydride reduction, proceed with high chemoselectivity, preserving the established stereochemistry. The final hydrogenation step utilizes heterogeneous catalysts like Pd-C or Raney Nickel under controlled hydrogen pressure, ensuring complete saturation of the double bonds without racemization, thereby delivering the final optically active menthol with consistent quality.
How to Synthesize Optically Active Menthol Efficiently
The synthesis of optically active menthol via this patented route involves a streamlined sequence of reactions designed for operational simplicity and high throughput. The process begins with the pretreatment of the starting material followed by the key asymmetric cyclization, which sets the stereochemical foundation for the molecule. Detailed optimization of reaction parameters such as temperature, catalyst loading, and solvent choice is essential to maximize yield and enantiomeric excess. For technical teams looking to implement this chemistry, the following guide outlines the standardized operational framework derived from the patent examples, ensuring reproducible results from laboratory to pilot scale.
- Pretreat methyl 5-methyl-4-hexenoate with alkali, then cyclize with ethyl acetoacetate using a copper-chiral phosphine catalyst to form compound B.
- Perform decarboxylation on compound B under alkaline conditions to generate optically active isomenthadienone (compound C).
- Reduce compound C using a reducing agent like NaBH4 to obtain isomenthadienol (compound D), followed by catalytic hydrogenation to yield optically active menthol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this copper-catalyzed route offers substantial strategic benefits beyond mere technical feasibility. The replacement of scarce and volatile noble metals like rhodium with abundant copper salts drastically reduces raw material cost exposure and mitigates supply risk. Furthermore, the use of commodity chemicals such as methyl 5-methyl-4-hexenoate and ethyl acetoacetate ensures a stable supply chain that is not subject to the agricultural fluctuations affecting natural mint oil. The mild reaction conditions and simplified workup procedures also translate to lower energy consumption and reduced waste treatment costs, aligning with modern sustainability goals and regulatory compliance requirements.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior due to the elimination of expensive chiral noble metal catalysts. By utilizing copper salts and recyclable phosphine ligands, the direct material cost per kilogram of product is significantly lowered. Additionally, the high selectivity of the reaction minimizes the formation of by-products, reducing the burden on downstream purification units and lowering solvent consumption. The ability to upgrade optical purity through crystallization rather than complex chromatographic separations further drives down operational expenditures, offering a clear path to improved margins in fine chemical manufacturing.
- Enhanced Supply Chain Reliability: Dependence on natural extracts or specialized noble metals introduces significant vulnerability to supply disruptions. This synthetic route relies on petrochemical-derived intermediates that are produced in vast quantities globally, ensuring consistent availability and price stability. The robustness of the catalytic system allows for continuous or large-batch processing without the frequent catalyst replenishment cycles required by sensitive rhodium systems. This reliability enables manufacturers to commit to long-term supply contracts with confidence, knowing that production capacity can be maintained regardless of seasonal or geopolitical factors affecting raw material sourcing.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard unit operations such as stirred tank reactors and crystallizers that are common in existing fine chemical facilities. The mild temperatures and pressures reduce the safety risks associated with high-energy processes, facilitating easier regulatory approval and safer plant operations. Moreover, the reduction in heavy metal usage simplifies wastewater treatment and waste disposal protocols, helping companies meet increasingly stringent environmental standards. The overall atom economy and reduced solvent intensity contribute to a smaller carbon footprint, making this a preferred technology for environmentally conscious corporate procurement strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this optically active menthol synthesis technology. These insights are derived directly from the experimental data and process descriptions within the patent documentation, providing a factual basis for evaluating its suitability for your specific production needs. Understanding these details is crucial for making informed decisions about technology adoption and partnership.
Q: What represents the primary cost advantage of this menthol synthesis route?
A: The primary cost advantage lies in replacing expensive noble metal catalysts like Rhodium-BINAP complexes with economical copper salts and chiral phosphine ligands, significantly lowering catalyst procurement costs while maintaining high enantioselectivity through crystallization.
Q: How does this method improve supply chain stability compared to extraction?
A: Unlike mint leaf extraction which suffers from seasonal variability and low yields, this chemical synthesis utilizes readily available industrial intermediates like methyl 5-methyl-4-hexenoate, ensuring consistent year-round production capacity and reliable delivery schedules.
Q: What is the achieved optical purity of the final menthol product?
A: Through a combination of asymmetric catalysis and optimized crystallization purification steps, the process achieves an optical purity ranging from 93% ee to 99% ee, meeting stringent specifications for high-value flavor and pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Optically Active Menthol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to maintain competitiveness in the global flavor and fragrance market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of optically active menthol meets the highest international standards for enantiomeric excess and chemical purity required by top-tier pharmaceutical and food-grade applications.
We invite you to collaborate with us to leverage this cutting-edge technology for your supply chain. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this copper-catalyzed route can optimize your bottom line. Please contact us today to request specific COA data and route feasibility assessments, and let us partner with you to secure a sustainable and cost-effective source of high-quality optically active menthol.
