Advanced Synthesis of N-Methyl-P-Nitroaniline: Technical Breakthroughs and Commercial Scalability
The chemical industry continuously seeks optimization in the synthesis of critical organic intermediates, particularly those serving dual roles in pharmaceutical and energetic material sectors. Patent CN101580473A introduces a transformative methodology for the preparation of N-methyl-p-nitroaniline, a compound of significant utility as a stabilizer in gunpowder propellants and as a benchmark substance in analytical chemistry. This technical disclosure addresses long-standing inefficiencies in traditional manufacturing by proposing a three-step sequence that prioritizes mild reaction conditions and exceptional product quality. By shifting away from hazardous reagents and extreme physical parameters, this innovation offers a robust framework for reliable agrochemical intermediate supplier networks and fine chemical manufacturers alike. The following analysis dissects the technical merits of this approach, evaluating its potential to redefine cost reduction in electronic chemical manufacturing and broader specialty chemical sectors through enhanced safety and yield profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of N-methyl-p-nitroaniline has been plagued by severe safety hazards and operational complexities inherent to legacy synthetic routes. As illustrated in prior art, one common pathway involves the reaction of p-nitrochlorobenzene with potassium fluoride at temperatures ranging from 170 to 175°C, utilizing tetramethylammonium chloride as a catalyst.
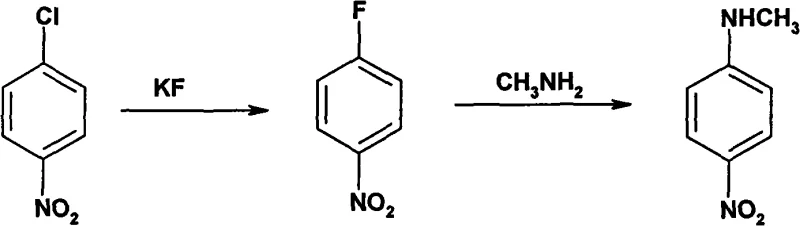
This specific methodology, while capable of achieving yields around 70%, generates p-nitrofluorobenzene and hydrofluoric acid (HF) as intermediates or by-products, both of which are notoriously toxic and highly corrosive. The presence of HF necessitates the use of specialized, expensive corrosion-resistant reactor linings and imposes rigorous safety protocols that inflate operational expenditures. Furthermore, alternative routes involving sulfonation require massive quantities of concentrated sulfuric acid, creating substantial waste treatment burdens and equipment degradation issues. Another direct ammonolysis method demands high-temperature and high-pressure conditions, restricting the choice of reactor materials and limiting the flexibility of batch processing. These cumulative factors create significant bottlenecks for supply chain heads seeking consistent delivery schedules and procurement managers aiming for predictable cost structures.
The Novel Approach
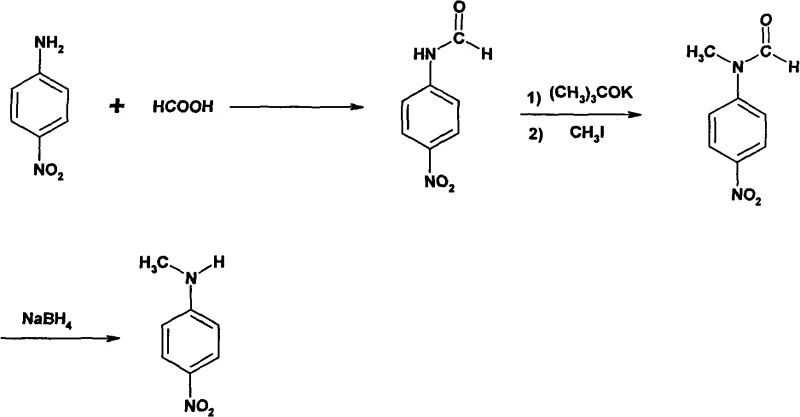
In stark contrast to these hazardous legacy methods, the technology disclosed in CN101580473A presents a refined, three-step synthetic strategy that operates under significantly milder conditions. The process initiates with the formylation of p-nitroaniline using formic acid, followed by a precise N-methylation step utilizing potassium tert-butoxide and methyl iodide in a dimethylformamide solvent system. The final stage employs sodium borohydride for reduction, a reagent known for its selectivity and ease of handling compared to stronger reducing agents. This novel approach eliminates the generation of hydrofluoric acid entirely and avoids the need for high-pressure autoclaves, thereby drastically simplifying the engineering requirements for commercial scale-up of complex polymer additives or pharmaceutical precursors. The reaction sequence is designed such that each step achieves a yield greater than 90%, culminating in an overall yield exceeding 80%, which represents a substantial improvement over the 70% ceiling of older technologies. Moreover, the workup procedures involve simple aqueous precipitation and filtration, removing the need for complex distillation columns or hazardous extraction processes.
Mechanistic Insights into Formylation and Reductive Methylation
The core of this technological advancement lies in the strategic protection and activation of the amine functionality through a formyl group, which serves as a versatile handle for subsequent alkylation. In the first stage, p-nitroaniline reacts with formic acid at temperatures between 80 and 105°C. This formylation step is critical as it converts the nucleophilic amine into an amide, which is less susceptible to oxidation and more manageable during the subsequent alkylation phase. The resulting N-formyl-p-nitroaniline precipitates upon cooling and dilution with water, allowing for easy isolation with a yield of approximately 95.7% to 96.1% as demonstrated in the patent examples. This high efficiency in the initial step sets a strong foundation for the overall process mass intensity, ensuring that raw material consumption is minimized from the outset.
Following isolation, the N-formyl intermediate undergoes methylation in the presence of a strong base, potassium tert-butoxide, and methyl iodide. The base deprotonates the amide nitrogen, generating a nucleophilic anion that attacks the methyl iodide to install the methyl group. This reaction proceeds at room temperature over an 8-hour period, showcasing the mildness of the conditions compared to the 170°C required in traditional fluorination routes. The final reduction step utilizes sodium borohydride in ethanol to cleave the formyl group, regenerating the secondary amine structure of N-methyl-p-nitroaniline. The use of sodium borohydride is particularly advantageous for impurity control, as it selectively reduces the amide without affecting the nitro group on the aromatic ring, a common side reaction with stronger hydride sources. This selectivity ensures that the final product maintains a purity of ≥98% with a sharp melting point of 152 to 153°C, meeting the rigorous standards expected by a reliable pharma intermediates supplier.
How to Synthesize N-Methyl-P-Nitroaniline Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and temperature control to maximize the benefits of the patented method. The process is divided into three distinct operational units: formylation, methylation, and reduction, each with specific quenching and isolation protocols that ensure high recovery rates. Operators must adhere to the specified molar ratios, particularly the 1:1 to 1.5:1 ratio of potassium tert-butoxide and methyl iodide relative to the substrate, to prevent over-alkylation or incomplete conversion. The detailed standardized synthesis steps, including exact solvent volumes and stirring times, are outlined below to facilitate technology transfer and pilot plant trials.
- React p-nitroaniline with formic acid at 80-105°C to form N-formyl-p-nitroaniline.
- Methylate the intermediate using potassium tert-butoxide and methyl iodide in DMF at room temperature.
- Reduce the N-methyl-N-formyl derivative using sodium borohydride in ethanol to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this patented synthesis route offers compelling economic and logistical advantages that extend beyond simple yield improvements. By eliminating the reliance on hydrofluoric acid and high-pressure reactors, manufacturers can significantly reduce capital expenditure on specialized equipment and lower the ongoing costs associated with safety compliance and hazardous waste disposal. The simplified workup procedure, which relies on water precipitation rather than complex distillation or chromatography, translates directly into reduced energy consumption and shorter batch cycle times. These operational efficiencies contribute to substantial cost savings in fine chemical intermediates manufacturing, allowing suppliers to offer more competitive pricing without compromising on margin. Furthermore, the use of readily available reagents like formic acid and sodium borohydride mitigates the risk of raw material supply disruptions, enhancing the overall resilience of the supply chain.
- Cost Reduction in Manufacturing: The elimination of corrosive hydrofluoric acid removes the necessity for expensive Hastelloy or lined reactors, allowing for the use of standard stainless steel equipment which drastically lowers infrastructure investment. Additionally, the high per-step yields (>90%) minimize raw material waste, ensuring that the cost of goods sold is optimized through efficient atom economy. The avoidance of high-temperature and high-pressure conditions further reduces energy utility costs, as reactions can be driven by standard heating and cooling systems rather than specialized thermal oil or steam setups. These factors combine to create a leaner manufacturing process that is inherently more cost-effective than traditional sulfonation or fluorination routes.
- Enhanced Supply Chain Reliability: The reliance on common, non-controlled reagents such as formic acid and methyl iodide ensures a stable supply of inputs, reducing the risk of production stoppages due to raw material shortages. The robustness of the reaction conditions, which tolerate minor fluctuations in temperature and mixing without significant yield loss, enhances process reliability and consistency across different production batches. This stability is crucial for maintaining just-in-time delivery schedules for downstream customers in the pharmaceutical and agrochemical sectors. By simplifying the purification process to basic filtration and recrystallization, the lead time for high-purity pharmaceutical intermediates is significantly shortened, enabling faster response to market demand spikes.
- Scalability and Environmental Compliance: The absence of toxic HF by-products simplifies environmental permitting and waste treatment, as effluent streams are less hazardous and easier to neutralize. The process generates minimal solid waste, primarily consisting of inorganic salts that can be managed through standard industrial waste protocols. This environmental profile aligns with increasingly stringent global regulations on chemical manufacturing, reducing the regulatory burden on production facilities. The straightforward nature of the unit operations facilitates seamless scale-up from laboratory grams to multi-ton commercial production, ensuring that quality remains consistent regardless of batch size. This scalability supports long-term supply agreements and provides confidence to partners seeking a sustainable source of critical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a transparent view of the process capabilities. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their specific application needs.
Q: Why is the new synthesis method safer than traditional routes?
A: Traditional methods often utilize highly toxic hydrofluoric acid or require high-temperature sulfonation. The patented method operates under mild conditions (20-105°C) without corrosive HF, significantly enhancing operational safety.
Q: What purity levels can be achieved with this process?
A: The process consistently yields products with a purity of ≥98% and a melting point of 152-153°C, meeting stringent requirements for high-performance applications.
Q: Is this synthesis route suitable for large-scale manufacturing?
A: Yes, the method features simple post-treatment involving aqueous precipitation and filtration, avoiding complex high-pressure equipment, which makes it highly scalable for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Methyl-P-Nitroaniline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global fine chemicals market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent to practice is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, leveraging our rigorous QC labs to verify every batch against the high standards set by patents like CN101580473A. Our infrastructure is designed to handle complex organic syntheses safely, allowing us to capitalize on the cost and safety benefits of this novel route while guaranteeing supply continuity for our partners.
We invite you to collaborate with us to optimize your supply chain for N-methyl-p-nitroaniline and related intermediates. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your R&D and production goals effectively.
