Advanced Green Synthesis of 4-(4'-n-alkyl cyclohexyl)cyclohexanone for High-Performance Liquid Crystals
The rapidly evolving landscape of display technologies demands increasingly sophisticated materials, particularly within the realm of liquid crystals where stability and response speed are paramount. Patent CN101337870A introduces a pivotal advancement in the synthesis of 4-(4'-n-alkyl cyclohexyl)cyclohexanone, a critical intermediate for next-generation cyclohexane-based liquid crystals. This patent discloses a novel oxidation methodology that replaces hazardous traditional reagents with a benign tungsten-catalyzed system utilizing hydrogen peroxide. For R&D directors and procurement specialists seeking a reliable liquid crystal intermediate supplier, this technology represents a significant leap forward in process safety and environmental compliance. The structural integrity of the target molecule, as depicted in the general formula below, is preserved through mild yet effective oxidative conditions.
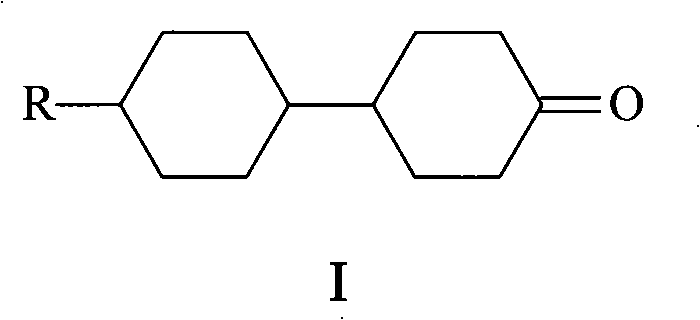
By shifting away from stoichiometric heavy metal oxidants, this process addresses the growing regulatory pressure on chemical manufacturing facilities to minimize toxic effluent. The core innovation lies in the specific combination of sodium tungstate dihydrate and phosphotungstic acid, which generates a highly active peroxo-species in situ. This approach not only ensures high conversion rates but also facilitates a cleaner workup procedure, directly impacting the bottom line for manufacturers focused on cost reduction in electronic chemical manufacturing. As the industry moves towards greener supply chains, adopting such methodologies becomes a strategic imperative for maintaining competitive advantage.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the oxidation of secondary alcohols to ketones, a fundamental transformation in organic synthesis, has relied heavily on reagents that pose significant environmental and operational challenges. Traditional methods often employ chromium trioxide in sulfuric acid (Jones reagent), which generates vast quantities of toxic chromium(VI) waste that requires expensive and complex remediation protocols before discharge. Alternatively, dimethyl sulfoxide (DMSO) based oxidations, while effective, produce difficult-to-remove sulfur-containing byproducts and emit unpleasant odors that complicate plant operations. Furthermore, hypochlorite oxidation methods frequently suffer from poor selectivity, leading to the formation of chlorinated impurities that are notoriously difficult to separate from the desired product, thereby lowering the overall yield and purity.
The Novel Approach
In stark contrast, the methodology described in CN101337870A utilizes a catalytic amount of tungsten species coupled with hydrogen peroxide, an ideal "green" oxidant whose only byproduct is water. This system operates effectively in N-methylpyrrolidinone (NMP), a high-boiling polar aprotic solvent that stabilizes the reaction intermediates. The reaction scheme illustrates the direct conversion of the alcohol precursor (II) to the ketone product (I) without the introduction of halogen atoms or heavy metal residues.

This novel approach eliminates the need for extensive aqueous workups designed to remove heavy metals, streamlining the isolation process to simple solvent distillation and extraction. The absence of chlorinated side products means that the crude product profile is significantly cleaner, reducing the burden on purification columns and crystallization steps. For supply chain managers, this translates to a more robust process with fewer variables that could lead to batch failures or delays, ensuring a consistent supply of high-purity 4-(4'-n-alkyl cyclohexyl)cyclohexanone.
Mechanistic Insights into Tungsten-Catalyzed Oxidation
The efficacy of this synthesis relies on the formation of active peroxotungstate complexes generated from the interaction between sodium tungstate, phosphotungstic acid, and hydrogen peroxide. Mechanistically, the tungsten center coordinates with the peroxide anion to form an electrophilic oxygen donor species. This activated oxygen is then transferred to the hydroxyl group of the 4-(4'-n-alkyl cyclohexyl)cyclohexanol substrate. The presence of phosphotungstic acid acts as a co-catalyst or promoter, likely enhancing the acidity of the medium and stabilizing the peroxo-complex, thereby accelerating the rate-determining step of oxygen transfer. This catalytic cycle allows for the use of sub-stoichiometric amounts of tungsten, making the process economically viable despite the cost of the metal salts.
From an impurity control perspective, the mechanism inherently suppresses over-oxidation or ring-opening reactions that might occur under harsher acidic conditions typical of chromic acid oxidations. The mild nature of the hydrogen peroxide/tungsten system ensures that the sensitive cyclohexyl rings remain intact, preserving the stereochemical and structural properties required for liquid crystal applications. By avoiding radical pathways often associated with other oxidants, the formation of polymeric tars or degradation products is minimized. This high level of chemoselectivity is crucial for achieving the stringent purity specifications demanded by the display industry, where even trace impurities can adversely affect the electro-optical performance of the final liquid crystal mixture.
How to Synthesize 4-(4'-n-alkyl cyclohexyl)cyclohexanone Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for scaling this reaction from laboratory to production scales. The process begins with the careful preparation of the oxidant solution, followed by the controlled addition of the substrate and solvent. Maintaining the temperature between 80°C and 90°C is critical to balancing reaction rate and selectivity. Detailed standard operating procedures regarding molar ratios and workup techniques are essential for reproducibility.
- Prepare the oxidant by mixing sodium tungstate dihydrate and phosphotungstic acid, followed by the addition of hydrogen peroxide under stirring.
- Add the substrate 4-(4'-n-alkyl cyclohexyl)cyclohexanol and the solvent N-methylpyrrolidinone to the oxidant mixture.
- Heat the reaction mixture to 80-90°C for 5-8 hours, then recover the solvent via vacuum distillation and extract the product with petroleum ether.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this tungsten-catalyzed oxidation route offers tangible benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic simplification of waste management protocols. By eliminating hexavalent chromium and chlorinated organics from the waste stream, facilities can avoid the high costs associated with hazardous waste disposal and regulatory compliance reporting. This shift not only reduces direct operational expenditures but also mitigates the risk of production shutdowns due to environmental violations, thereby enhancing overall supply chain resilience.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the use of hydrogen peroxide, which is a commodity chemical available at a fraction of the cost of specialized oxidants like DMSO-activators or chromium reagents. Furthermore, the ability to recover and reuse the N-methylpyrrolidinone solvent through vacuum distillation significantly lowers the raw material consumption per kilogram of product. The high yields reported (exceeding 90% in examples) mean that less starting material is wasted, directly improving the atom economy and reducing the cost of goods sold (COGS) for the final intermediate.
- Enhanced Supply Chain Reliability: Relying on widely available, non-restricted reagents like sodium tungstate and hydrogen peroxide reduces the risk of supply disruptions compared to sourcing specialized or regulated chemicals. The robustness of the reaction conditions, which tolerate a range of alkyl chain lengths (C1 to C7), allows manufacturers to produce a family of related intermediates on the same production line with minimal changeover time. This flexibility enables suppliers to respond more agilely to fluctuating market demands for different liquid crystal formulations, ensuring shorter lead times for customers.
- Scalability and Environmental Compliance: The exothermic nature of peroxide oxidations is well-managed in this protocol through controlled addition and temperature maintenance, making it suitable for large-scale batch reactors. The absence of toxic heavy metals simplifies the qualification of equipment and reduces the cleaning validation burden between batches. From a sustainability standpoint, the "green" label of this process aligns with the corporate social responsibility (CSR) goals of major electronics brands, potentially opening up preferred vendor status for manufacturers who can certify their processes as environmentally friendly.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. Understanding these details is vital for stakeholders evaluating the feasibility of integrating this technology into their existing manufacturing portfolios. The answers are derived directly from the experimental data and claims presented in the patent documentation.
Q: What are the environmental advantages of this tungsten-catalyzed oxidation method?
A: Unlike traditional chromium-based oxidations which generate toxic heavy metal waste, this method uses hydrogen peroxide as the terminal oxidant, producing only water as a byproduct. This significantly simplifies wastewater treatment and aligns with green chemistry principles.
Q: How does the selectivity of this process compare to hypochlorite oxidation?
A: The tungsten-phosphotungstic acid catalyst system offers superior chemoselectivity compared to hypochlorite methods. It avoids the formation of chlorinated by-products, thereby enhancing the purity profile of the final liquid crystal intermediate and reducing downstream purification burdens.
Q: Is the solvent N-methylpyrrolidinone recoverable in this process?
A: Yes, the process design allows for the recovery of N-methylpyrrolidinone through vacuum distillation after the reaction is complete. The recovered solvent can be dehydrated and reused, contributing to substantial cost reductions in raw material consumption.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-(4'-n-alkyl cyclohexyl)cyclohexanone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the performance of advanced display materials. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to industrial manufacturing is seamless. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of 4-(4'-n-alkyl cyclohexyl)cyclohexanone meets the exacting standards required for liquid crystal applications. Our commitment to green chemistry principles aligns perfectly with the efficient, low-waste methodology described in CN101337870A.
We invite global partners to collaborate with us to leverage this advanced synthesis technology for their supply chains. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential clients to reach out for specific COA data and route feasibility assessments to verify how our capabilities can support your long-term production goals and reduce your overall manufacturing footprint.
