Advanced Chiral Pool Synthesis Strategies for Commercial Scale (S)-Metolachlor Production
Advanced Chiral Pool Synthesis Strategies for Commercial Scale (S)-Metolachlor Production
The global demand for high-efficiency, environmentally friendly agrochemicals has driven a significant shift towards single-enantiomer herbicides, with (S)-metolachlor standing out as a premier example of this evolution. Traditional manufacturing often relied on the production of racemic mixtures followed by inefficient separation processes, resulting in substantial waste of the inactive (R)-isomer. However, recent advancements detailed in patent CN101367746A introduce a robust, full-synthesis route that leverages chiral pool technology to achieve superior optical purity without the need for post-synthesis resolution. This innovative approach utilizes (D)-lactic acid esters as the foundational chiral source, systematically transforming them through a series of highly controlled chemical modifications including tosylation, nucleophilic substitution, reduction, acylation, and methylation. By anchoring the synthesis on a naturally occurring chiral building block, this method effectively bypasses the complexities associated with asymmetric catalysis while maintaining rigorous control over stereochemistry.
For procurement specialists and supply chain directors, the implications of this technology are profound, offering a pathway to cost reduction in agrochemical intermediate manufacturing that is both sustainable and economically viable. The elimination of resolution steps inherently doubles the theoretical yield compared to racemic synthesis, directly impacting the bottom line by maximizing raw material utilization. Furthermore, the reliance on commodity chemicals such as p-toluenesulfonyl chloride and standard reducing agents ensures a stable supply chain, mitigating the risks associated with sourcing specialized chiral catalysts. As the industry moves towards stricter regulatory standards regarding pesticide residues and environmental impact, securing a reliable agrochemical intermediate supplier capable of delivering high-purity (S)-metolachlor through such efficient routes becomes a critical strategic priority for downstream formulators and agricultural chemical giants.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of metolachlor has been dominated by two primary methodologies: the synthesis of the racemate followed by enzymatic or chemical resolution, and asymmetric catalytic hydrogenation of imines. While asymmetric hydrogenation represents a technological marvel, it imposes significant barriers to entry for many manufacturers due to the requirement for sophisticated high-pressure reactor systems and precious metal catalysts, such as iridium complexes with chiral ligands. These catalysts are not only exorbitantly expensive but also sensitive to poisoning, requiring ultra-pure feedstocks and stringent operational controls that drive up capital expenditure and operational costs. On the other hand, resolution methods suffer from an intrinsic ceiling on yield, as a maximum of 50% of the produced material corresponds to the desired active (S)-isomer, with the remaining (R)-isomer often discarded or requiring energy-intensive recycling processes. This inherent inefficiency translates to higher waste generation and a larger carbon footprint, which is increasingly untenable in the modern green chemistry landscape.
The Novel Approach
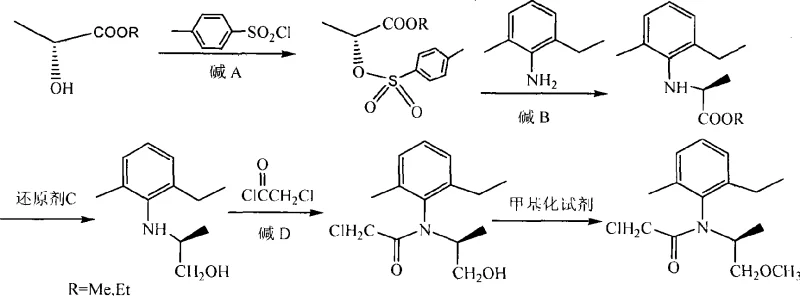
In stark contrast, the methodology disclosed in the referenced patent circumvents these bottlenecks by employing a chiral pool strategy that begins with optically pure (D)-lactate esters. This approach fundamentally alters the economic equation by removing the need for expensive chiral catalysts and high-pressure equipment entirely. The synthesis proceeds through a logical sequence where the chirality of the starting material is faithfully transferred to the final product through stereospecific reactions. For instance, the initial conversion of the hydroxyl group to a tosylate and its subsequent displacement by 2-methyl-6-ethylaniline occurs with predictable stereochemical outcomes, ensuring that the optical integrity established at the beginning is preserved throughout the multi-step sequence. This results in a process that operates under relatively mild conditions, typically ranging from sub-zero temperatures for sensitive steps to moderate heating for substitutions, thereby reducing energy consumption. The ability to achieve high enantiomeric excess (e.e.) values, often exceeding 90% and reaching up to 99% in optimized examples, without a single resolution step, marks a paradigm shift in the commercial scale-up of complex agrochemical intermediates.
Mechanistic Insights into Chiral Transfer and Substitution Dynamics
The core of this synthesis lies in the precise manipulation of the chiral center located at the alpha-position of the lactate derivative. The process initiates with the activation of the secondary hydroxyl group of (D)-methyl or ethyl lactate using p-toluenesulfonyl chloride in the presence of a base such as triethylamine. This tosylation step converts the poor leaving group (-OH) into an excellent leaving group (-OTs), setting the stage for the subsequent nucleophilic attack. Crucially, this step must be conducted at controlled low temperatures, typically between -10°C and 30°C, to prevent any potential racemization or side reactions that could compromise the optical purity. The resulting (R)-2-(p-toluenesulfonyloxy)propionate retains the configuration necessary for the next transformation, acting as a pivotal electrophile in the synthesis chain.
Following activation, the introduction of the aromatic amine moiety is achieved through a nucleophilic substitution reaction with 2-methyl-6-ethylaniline. This step is thermally driven, often requiring temperatures between 110°C and 160°C, and utilizes a base to scavenge the generated acid. The mechanism involves the nitrogen lone pair of the aniline attacking the chiral carbon, displacing the tosylate group. Depending on the specific conditions and solvent environment, this substitution can proceed via an SN2 mechanism which would invert the configuration, or through pathways that retain stereochemistry depending on the specific ion-pairing dynamics. The patent data indicates that the process successfully yields the S-(-)-N-(2-methyl-6-ethylphenyl)alanine ester with high optical purity, demonstrating that the reaction conditions are finely tuned to favor the desired stereoisomer. Subsequent reduction of the ester group to a primary alcohol using hydride sources like sodium borohydride modified with lithium chloride or lithium aluminum hydride further functionalizes the molecule without affecting the chiral center, preparing it for the final acylation and methylation steps that complete the herbicide structure.
How to Synthesize (S)-Metolachlor Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and optical purity. The process is divided into five distinct operational stages, beginning with the preparation of the chiral tosylate and concluding with the final methylation. Each stage demands specific control over temperature, stoichiometry, and workup procedures to ensure the removal of impurities that could affect downstream performance. The following guide outlines the standardized operational framework derived from the patent examples, providing a roadmap for laboratory validation and pilot plant scaling.
- Activation of (D)-lactate ester with p-toluenesulfonyl chloride to form the tosylate intermediate with inversion potential.
- Nucleophilic substitution with 2-methyl-6-ethylaniline to establish the chiral amine backbone.
- Reduction of the ester moiety to the corresponding alcohol using borohydride or aluminum hydride reagents.
- Acylation with chloroacetyl chloride to form the chloroacetamide precursor.
- Final O-methylation using methylating agents like dimethyl sulfate or methyl iodide under basic conditions.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this chiral pool synthesis route offers compelling advantages that extend beyond simple chemical yield. The primary driver for cost optimization is the elimination of the resolution step, which traditionally acts as a massive efficiency drain in chiral manufacturing. By utilizing a starting material that is already enantiomerically enriched, the process effectively doubles the mass efficiency compared to racemic synthesis, leading to substantial raw material savings. Furthermore, the reagents employed—such as lactate esters, tosyl chloride, and common hydride reducers—are commodity chemicals with stable global supply chains, reducing the risk of procurement bottlenecks associated with specialized chiral ligands or enzymes. This stability allows for more accurate long-term forecasting and inventory management, crucial for maintaining continuous production lines in the volatile agrochemical market.
- Cost Reduction in Manufacturing: The economic benefits are realized through the simplification of the process flow. By removing the need for expensive transition metal catalysts and the associated recovery systems, capital expenditure is significantly lowered. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, while the high selectivity minimizes the formation of by-products, thereby lowering waste disposal costs. The overall result is a leaner manufacturing process with a lower cost of goods sold (COGS), enabling more competitive pricing in the global herbicide market without sacrificing margin.
- Enhanced Supply Chain Reliability: Dependence on complex catalytic systems often introduces single points of failure in the supply chain. This alternative route relies on robust, well-understood organic transformations that can be performed in standard glass-lined or stainless steel reactors available in most fine chemical facilities. The accessibility of raw materials ensures that production schedules are less susceptible to disruptions caused by the scarcity of niche reagents. This reliability is paramount for meeting the seasonal demands of the agricultural sector, ensuring that high-purity agrochemical intermediates are available precisely when needed for formulation.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the successful execution of reactions in multi-hundred gram scales in the patent examples without loss of efficiency. The avoidance of heavy metals aligns with increasingly stringent environmental regulations regarding residual metals in agricultural products. This compliance reduces the regulatory burden and testing costs associated with product registration. Moreover, the simplified waste stream, devoid of complex metal-ligand residues, facilitates easier treatment and disposal, supporting corporate sustainability goals and enhancing the overall environmental profile of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on the feasibility and performance characteristics of the route.
Q: What is the primary advantage of this chiral pool method over asymmetric hydrogenation?
A: This method utilizes readily available chiral starting materials like (D)-lactate, eliminating the need for expensive transition metal catalysts and complex high-pressure hydrogenation equipment required in asymmetric catalysis.
Q: How is optical purity maintained throughout the synthesis?
A: Optical purity is preserved by leveraging the inherent chirality of the lactate starting material. The reaction conditions, particularly the mild temperatures during tosylation and reduction, prevent racemization, ensuring high e.e. values in the final product.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process avoids cryogenic conditions and uses common organic solvents and reagents. The absence of resolution steps significantly improves overall yield and throughput, making it highly scalable for commercial agrochemical production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-Metolachlor Supplier
The transition to more efficient chiral synthesis routes represents a significant opportunity for agrochemical manufacturers to enhance their competitive positioning. NINGBO INNO PHARMCHEM stands at the forefront of this technological evolution, possessing the technical expertise and infrastructure to translate complex laboratory protocols into robust commercial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot to plant is seamless and risk-mitigated. We understand that consistency is key in the agrochemical sector; therefore, our stringent purity specifications and rigorous QC labs guarantee that every batch of (S)-metolachlor meets the highest international standards for optical purity and chemical identity.
We invite industry partners to engage with us to explore how this advanced synthesis route can be integrated into your supply chain. By collaborating with our technical procurement team, you can obtain a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and profitability in your herbicide production portfolio.
