Advanced Oxidative Cleavage Strategy for High-Purity Bilastine Manufacturing
Advanced Oxidative Cleavage Strategy for High-Purity Bilastine Manufacturing
The pharmaceutical industry continuously seeks robust synthetic pathways that balance efficiency with environmental sustainability, particularly for second-generation antihistamines like Bilastine. Patent CN103214455A introduces a transformative approach to synthesizing this critical active pharmaceutical ingredient by leveraging a mild oxidative cleavage strategy. Unlike traditional methods that rely on harsh acidic environments or multi-step ester hydrolysis, this innovation utilizes a hypochlorite-based system facilitated by phase transfer catalysis. This technical breakthrough addresses long-standing challenges in impurity control and yield optimization, positioning it as a superior choice for manufacturers aiming to secure a reliable pharmaceutical intermediate supplier status. The methodology not only simplifies the operational workflow but also aligns with green chemistry principles by reducing the reliance on corrosive mineral acids.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of Bilastine has been hindered by the limitations inherent in acid-catalyzed hydrolysis and base-mediated ester saponification. As illustrated in prior art such as CN 1176964A, the use of strong inorganic acids like hydrochloric or sulfuric acid often leads to the fracture of sensitive ether bonds within the molecular structure. 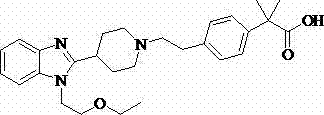 This degradation results in the formation of complex impurity profiles that are notoriously difficult to remove during downstream processing, ultimately compromising the overall yield which typically hovers around 72% to 80%. Furthermore, alternative routes involving ester intermediates, as seen in other patents, require rigorous temperature control and extended reaction times to achieve complete conversion, often necessitating the use of volatile organic solvents that complicate waste management and increase operational costs.
This degradation results in the formation of complex impurity profiles that are notoriously difficult to remove during downstream processing, ultimately compromising the overall yield which typically hovers around 72% to 80%. Furthermore, alternative routes involving ester intermediates, as seen in other patents, require rigorous temperature control and extended reaction times to achieve complete conversion, often necessitating the use of volatile organic solvents that complicate waste management and increase operational costs.
The Novel Approach
In stark contrast, the method disclosed in CN103214455A employs a sophisticated oxidative cleavage mechanism that bypasses the pitfalls of traditional hydrolysis. By utilizing an aqueous hypochlorite solution in the presence of a phase transfer catalyst, specifically tetrabutylammonium hydrogen sulfate, the process achieves efficient ring-opening of the oxazoline moiety under remarkably mild conditions. 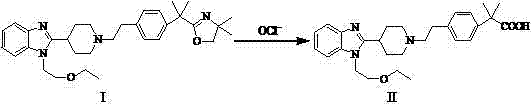 This approach eliminates the need for corrosive mineral acids, thereby preserving the integrity of the ether linkages and preventing the formation of degradation by-products. The reaction proceeds smoothly at temperatures ranging from 0°C to 80°C, offering flexibility in thermal management while consistently delivering yields as high as 93%, representing a significant improvement over legacy technologies.
This approach eliminates the need for corrosive mineral acids, thereby preserving the integrity of the ether linkages and preventing the formation of degradation by-products. The reaction proceeds smoothly at temperatures ranging from 0°C to 80°C, offering flexibility in thermal management while consistently delivering yields as high as 93%, representing a significant improvement over legacy technologies.
Mechanistic Insights into Hypochlorite-Mediated Oxidative Cleavage
The core of this technological advancement lies in the unique interaction between the hypochlorite ion and the oxazoline ring system, facilitated by the phase transfer catalyst. The quaternary ammonium salt acts as a molecular shuttle, transporting the hypochlorite anion from the aqueous phase into the organic phase where the substrate resides. This intimate contact allows for a selective nucleophilic attack on the electron-deficient carbon of the oxazoline ring, initiating a cascade that ultimately cleaves the C-N and C-O bonds to reveal the carboxylic acid functionality. Unlike acid hydrolysis which relies on protonation and water attack, this oxidative pathway avoids the generation of highly acidic microenvironments that trigger unwanted side reactions. The result is a cleaner reaction matrix where the desired Bilastine molecule is formed with high specificity, minimizing the burden on purification units.
From an impurity control perspective, this mechanism offers distinct advantages for R&D teams focused on quality by design. The absence of strong acids prevents the hydrolysis of the ethoxyethyl group attached to the benzimidazole nitrogen, a common failure point in conventional syntheses. Additionally, the mild alkaline or neutral conditions post-reaction allow for straightforward workup procedures involving simple acidification and extraction. This reduces the accumulation of inorganic salts and organic tars that often plague acid-based processes. Consequently, the final product exhibits a superior purity profile right out of the reactor, reducing the need for extensive recrystallization cycles and ensuring that the material meets the stringent specifications required for regulatory submission and commercial distribution.
How to Synthesize Bilastine Efficiently
Implementing this synthesis route requires careful attention to the stoichiometry of the oxidant and the efficiency of the phase transfer system. The process begins by dissolving the oxazoline precursor in a suitable organic solvent such as ethyl acetate, followed by the addition of the catalyst and the controlled introduction of the hypochlorite solution. Detailed standardized operating procedures regarding mixing rates, temperature ramps, and quenching protocols are essential to maximize the benefits of this chemistry. For a comprehensive guide on the specific execution of this protocol, please refer to the structured synthesis steps provided below.
- Dissolve the oxazoline precursor in an organic solvent such as ethyl acetate and add a phase transfer catalyst like tetrabutylammonium hydrogen sulfate.
- Introduce an aqueous hypochlorite solution and maintain the reaction temperature between 0°C and 80°C for 1 to 24 hours to effect oxidative cleavage.
- Acidify the reaction mixture, extract the product, and perform a secondary hydrolysis if necessary to obtain high-purity Bilastine solid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this hypochlorite-based methodology translates into tangible strategic benefits beyond mere chemical elegance. The shift away from corrosive mineral acids significantly reduces the corrosion load on manufacturing equipment, extending the lifespan of reactors and piping systems while lowering maintenance expenditures. Furthermore, the simplified workup procedure, which avoids complex neutralization and salt removal steps associated with strong acid usage, streamlines the production cycle. This operational simplicity directly contributes to cost reduction in pharmaceutical intermediate manufacturing by minimizing utility consumption and labor hours per batch, allowing for more competitive pricing structures in a crowded market.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous mineral acids, coupled with the high yield of the reaction, drives down the cost of goods sold significantly. By avoiding the loss of material due to side reactions and ether bond cleavage, the effective throughput of the plant is increased without additional capital investment. The use of commodity chemicals like sodium hypochlorite further ensures that raw material costs remain stable and predictable, shielding the supply chain from volatility associated with specialty reagents.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures consistent batch-to-batch quality, which is critical for maintaining uninterrupted supply to downstream API manufacturers. The mild reaction conditions reduce the risk of runaway exotherms or equipment failures, enhancing overall plant safety and operational continuity. Additionally, the availability of raw materials such as hypochlorite and common organic solvents means that production is not bottlenecked by the sourcing of exotic or restricted chemicals, thereby reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: This process is inherently scalable, having been demonstrated to work effectively from gram to kilogram scales with linear performance characteristics. The reduction in hazardous waste generation, particularly acidic wastewater and heavy metal contaminants often associated with other catalytic systems, simplifies environmental compliance and waste disposal logistics. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Bilastine synthesis route. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances is vital for stakeholders evaluating the feasibility of integrating this technology into their existing production portfolios.
Q: What are the primary advantages of the hypochlorite oxidation method over acid hydrolysis?
A: The hypochlorite oxidation method described in CN103214455A operates under significantly milder conditions compared to traditional strong acid hydrolysis, reducing the risk of ether bond fracture and minimizing difficult-to-remove impurities, thereby achieving yields up to 93%.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the process utilizes easily available raw materials like sodium hypochlorite and common organic solvents, with simple operational steps that facilitate safe and efficient scale-up from laboratory to commercial manufacturing volumes.
Q: How does this method impact the purity profile of the final API?
A: By avoiding harsh acidic conditions that typically degrade sensitive ether linkages, this novel route substantially reduces the generation of side products, resulting in a cleaner crude product that simplifies downstream purification and ensures stringent quality specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bilastine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the hypochlorite oxidation route are translated into reliable industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Bilastine intermediate we produce meets the highest international standards, providing our partners with the confidence needed to accelerate their drug development timelines.
We invite you to collaborate with us to leverage this cutting-edge technology for your supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing processes can drive value and efficiency for your organization.
