Optimizing Agrochemical Intermediate Production: A Deep Dive into Advanced Acetophenone Synthesis
The global demand for high-efficiency triazole fungicides continues to surge, driving the need for robust and scalable production methods for their key precursors. Central to this supply chain is 2-chloro-4-(4-chlorophenoxy)-acetophenone, a critical intermediate in the synthesis of difenoconazole, a broad-spectrum agricultural fungicide. Recent technological advancements, specifically detailed in patent CN113816838A, have introduced a transformative synthetic methodology that addresses long-standing inefficiencies in traditional manufacturing. This innovation not only enhances the purity profile of the final product but also introduces a sophisticated solvent recovery mechanism that aligns with modern green chemistry principles. For R&D directors and procurement strategists, understanding the nuances of this patented process is essential for securing a competitive edge in the agrochemical market. By shifting from conventional acylation protocols to this optimized Lewis acid-catalyzed route, manufacturers can achieve substantial improvements in yield consistency and operational safety.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 2-chloro-4-(4-chlorophenoxy)-acetophenone has relied on processes such as those described in earlier patents like CN101434528A, which utilized acetic anhydride as the acylating agent. These legacy methods suffer from significant thermodynamic and kinetic drawbacks, primarily stemming from complex post-reaction processing. A major bottleneck in these conventional routes is the inefficient solvent recovery; the processes often involve multiple extraction and distillation steps that fail to capture the majority of the organic solvent, leading to elevated production costs and increased environmental burden. Furthermore, the recrystallization stages in older methodologies frequently employ methanol, a solvent that exhibits non-negligible solubility for the target ketone even at low temperatures. This physicochemical property results in substantial product loss during the purification phase, capping the overall yield at approximately 90% and necessitating larger batch sizes to meet volume targets. The operational complexity of hydrolyzing before solvent treatment further complicates the workflow, creating opportunities for process deviations and quality inconsistencies.
The Novel Approach
In stark contrast, the novel approach outlined in the recent patent data leverages acetyl chloride as the acylating source in conjunction with a Lewis acid catalyst, fundamentally altering the reaction kinetics and downstream processing capabilities. This method introduces a streamlined workflow where solvent removal and hydrolysis are sequenced to maximize material retention. By employing solvents such as dichloroethane or o-dichlorobenzene, the process facilitates a highly efficient distillation step where up to 95% of the solvent fraction can be recovered and directly recycled into subsequent batches. This closed-loop capability drastically reduces raw material consumption and minimizes waste generation. Additionally, the switch to petroleum ether for the final recrystallization step eliminates the solubility losses associated with methanol, ensuring that the precipitated solid retains maximum mass. The result is a simplified production architecture that is not only easier to automate but also delivers a consistently higher purity product, making it an ideal candidate for cost reduction in agrochemical intermediates manufacturing.
Mechanistic Insights into Lewis Acid-Catalyzed Friedel-Crafts Acylation
The core of this synthetic breakthrough lies in the precise control of the Friedel-Crafts acylation mechanism, where the interaction between the Lewis acid catalyst and the acyl chloride generates a highly reactive acylium ion species. In this specific transformation, catalysts such as aluminum trichloride, stannic chloride, or lanthanum nitrate coordinate with the carbonyl oxygen of the acetyl chloride, enhancing its electrophilicity and facilitating the attack on the electron-rich aromatic ring of the diphenyl ether substrate. The regioselectivity of this reaction is paramount, as the goal is to introduce the acetyl group at the para-position relative to the ether linkage while managing the ortho-chlorine substituent. The patent data highlights that the choice of catalyst and the strict regulation of reaction temperature are critical determinants of success. For instance, maintaining the reaction environment at sub-zero temperatures, specifically around -15°C, suppresses the formation of poly-acylated byproducts and prevents the degradation of the sensitive ether linkage. This thermal control ensures that the electrophilic substitution proceeds with high fidelity, resulting in a crude reaction mixture that is significantly cleaner than those produced under ambient or elevated temperature conditions.
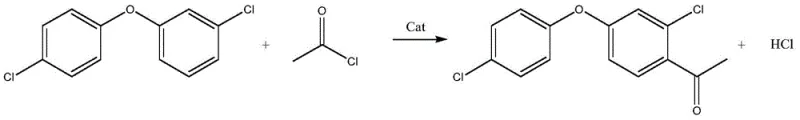
Beyond the primary reaction pathway, the impurity control mechanism is equally sophisticated, relying on the differential solubility and phase behavior of the reaction components during the workup phase. The process dictates a specific sequence where the solvent is removed prior to hydrolysis, which forces the catalyst-complexed intermediates into a solid state that can be cleanly separated from the bulk liquid. Upon the addition of deionized water for hydrolysis, the aluminum salts and other inorganic byproducts are solubilized into the aqueous phase, while the organic ketone partitions into the oil layer. The patent specifies a washing and extraction protocol that recovers residual product from the aqueous washings using the same solvent, ensuring that no valuable material is lost to the waste stream. This meticulous attention to phase separation and extraction efficiency is what allows the process to achieve normalization results exceeding 99%, satisfying the rigorous demands for high-purity agrochemical intermediates required by downstream formulation scientists.
How to Synthesize 2-Chloro-4-(4-Chlorophenoxy)-Acetophenone Efficiently
Implementing this advanced synthesis route requires a disciplined adherence to the specified operational parameters to fully realize the benefits of yield enhancement and solvent conservation. The procedure begins with the careful preparation of the reaction vessel, ensuring that moisture is excluded to prevent premature deactivation of the Lewis acid catalyst. Following the acylation and the strategic solvent recovery distillation, the hydrolysis step must be conducted with precise temperature control to facilitate clean phase separation without emulsification. The final purification via petroleum ether recrystallization is the defining step that locks in the product quality, removing trace isomers and unreacted starting materials. For technical teams looking to adopt this methodology, the following standardized guide outlines the critical operational milestones derived from the patent examples.
- Conduct Friedel-Crafts acylation by reacting diphenyl ether with acetyl chloride using a Lewis acid catalyst in a solvent like dichloroethane at controlled low temperatures.
- Remove the solvent via distillation for recycling, then hydrolyze the solid residue with deionized water to separate organic and aqueous layers.
- Extract the aqueous layer to recover residual product, combine oil layers, and perform recrystallization using petroleum ether to obtain the refined ketone.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this novel synthetic route offers profound advantages that extend well beyond simple chemical yield improvements. For procurement managers tasked with optimizing the cost structure of agrochemical supply chains, the ability to recycle the primary reaction solvent represents a direct and significant reduction in variable manufacturing costs. Unlike traditional methods where solvent loss is an accepted overhead, this process treats the solvent as a reusable asset, thereby insulating the production budget from volatile petrochemical price fluctuations. Furthermore, the simplification of the unit operations—merging synthesis and solvent recovery into a tighter loop—reduces the overall cycle time per batch. This efficiency gain translates into enhanced throughput capacity without the need for capital-intensive expansion of reactor farms, allowing suppliers to respond more agilely to market demand spikes. The robustness of the crystallization process also ensures a more predictable output schedule, mitigating the risk of delays caused by difficult filtrations or low recovery rates common in older methanol-based systems.
- Cost Reduction in Manufacturing: The elimination of expensive solvent losses and the reduction in raw material consumption per kilogram of product create a leaner cost base. By recovering and reusing dichloroethane or o-dichlorobenzene, the process minimizes the purchase frequency of bulk chemicals, leading to substantial long-term savings. Additionally, the higher yield means less waste disposal cost, further improving the bottom line for large-scale producers.
- Enhanced Supply Chain Reliability: The use of readily available reagents like acetyl chloride and diphenyl ether, combined with a process that tolerates standard industrial equipment, ensures a stable supply flow. The improved yield consistency reduces the need for safety stock buffers, allowing for a more just-in-time inventory strategy. This reliability is crucial for maintaining the continuity of fungicide production schedules during peak agricultural seasons.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex agrochemical intermediates, with clear protocols for heat management and phase separation that translate well from pilot to plant scale. The reduced generation of aqueous waste containing organic residues simplifies effluent treatment, helping manufacturers meet increasingly stringent environmental regulations without costly retrofitting of wastewater facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios. The answers reflect the balance between chemical performance and operational practicality that defines modern fine chemical production.
Q: Why is the new synthesis method superior to conventional acetic anhydride routes?
A: The novel method utilizes acetyl chloride instead of acetic anhydride, enabling a more efficient solvent recovery loop and avoiding the low recovery rates associated with methanol recrystallization in older processes.
Q: What represents the optimal reaction temperature for maximizing yield?
A: Experimental data indicates that maintaining the synthesis reaction temperature at approximately -15°C yields the highest conversion rates and total product yield, minimizing side reactions.
Q: How does this process impact environmental compliance and waste generation?
A: By implementing a closed-loop solvent recovery system where dichloroethane is distilled and reused, the process significantly reduces hazardous waste discharge and raw material consumption.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloro-4-(4-Chlorophenoxy)-Acetophenone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with both technical depth and manufacturing muscle. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN113816838A are fully realized in practice. We operate stringent purity specifications and maintain rigorous QC labs equipped to verify the identity and quality of every batch against global pharmacopeial and agrochemical standards. Our commitment to process excellence means that we do not just supply chemicals; we deliver supply chain security through consistent, high-quality output that meets the exacting needs of R&D directors and production planners alike.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific application. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic impact of switching to our superior grade intermediates. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project requirements, ensuring that your development timelines remain on track with the highest quality materials available in the market.
