Revolutionizing High-Performance Polymers: N-Substituted Polybenzimidazole Amide Commercialization
Revolutionizing High-Performance Polymers: N-Substituted Polybenzimidazole Amide Commercialization
The landscape of high-performance engineering plastics is undergoing a significant transformation driven by the innovations detailed in patent CN102604095B. This pivotal intellectual property introduces a novel class of N-substituted polybenzimidazole amide compounds that effectively address the longstanding limitations of traditional polybenzimidazole (PBI) materials. For R&D directors and procurement strategists in the aerospace and electronics sectors, this technology represents a critical advancement in material science, offering a unique combination of exceptional thermal stability and enhanced processability. The core innovation lies in the strategic modification of the polymer backbone, where the substitution of hydrogen atoms on the benzimidazole nitrogen atoms eliminates the instability associated with N-H bonds while simultaneously improving solubility. This breakthrough not only expands the application horizon for high-temperature polymers but also provides a robust foundation for developing next-generation composite materials capable of withstanding extreme operational environments without compromising structural integrity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional polybenzimidazole (PBI) has long been revered for its outstanding thermal resistance, yet its widespread adoption has been severely hindered by intrinsic processing challenges. The primary bottleneck is the extremely poor solubility of conventional PBI in common organic solvents, which necessitates complex and often hazardous processing methods that limit its utility in precision engineering applications. Furthermore, the presence of N-H bonds in the standard PBI structure renders the material susceptible to oxidative degradation in air, thereby reducing its long-term thermal stability and reliability in critical aerospace components. These structural deficiencies mean that manufacturers often face prohibitive costs and technical barriers when attempting to mold or coat substrates with traditional PBI, forcing them to seek alternative materials that may not offer the same level of performance. The inability to easily process these materials via solution casting or standard melt techniques has historically restricted their use to niche applications where no other alternatives exist, creating a supply chain vulnerability for industries demanding high-performance thermoplastics.
The Novel Approach
The methodology outlined in the patent data presents a paradigm shift by introducing an N-substituted architecture that fundamentally alters the physicochemical properties of the polymer. By synthesizing bis(benzimidazole) intermediates and reacting them with difluorinated amide compounds, the process creates a polymer chain where the nitrogen atoms are fully substituted, effectively removing the reactive N-H sites that cause instability. This structural modification results in a material that retains the rigid, heat-resistant backbone of PBI while gaining significant solubility in solvents like N,N-dimethylacetamide and dimethyl sulfoxide. 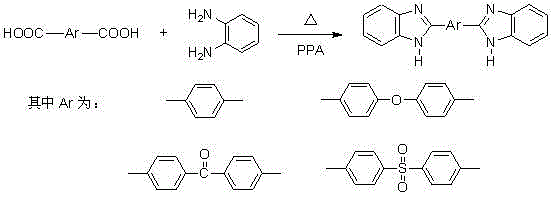 The ability to process this material via both melt and solution techniques offers unprecedented flexibility for manufacturing complex geometries and thin films. This novel approach transforms a difficult-to-process specialty polymer into a versatile engineering material, opening new avenues for cost reduction in advanced materials manufacturing by enabling standard injection molding and coating processes that were previously impossible with unmodified PBI.
The ability to process this material via both melt and solution techniques offers unprecedented flexibility for manufacturing complex geometries and thin films. This novel approach transforms a difficult-to-process specialty polymer into a versatile engineering material, opening new avenues for cost reduction in advanced materials manufacturing by enabling standard injection molding and coating processes that were previously impossible with unmodified PBI.
Mechanistic Insights into Polycondensation and N-Substitution
The chemical elegance of this synthesis lies in its multi-step polycondensation mechanism which ensures high molecular weight and structural regularity. The process begins with the formation of bis(benzimidazole) monomers through the cyclodehydration of aromatic dicarboxylic acids and o-phenylenediamine in a polyphosphoric acid medium at elevated temperatures around 200°C. This step is crucial for establishing the rigid heterocyclic units that provide the thermal backbone of the final polymer. Subsequently, the difluorinated amide comonomers are prepared via nucleophilic acyl substitution, where aromatic diamines react with 4-fluorobenzoyl chloride. The final polymerization step is a nucleophilic displacement reaction where the nitrogen atoms of the benzimidazole rings attack the fluorine-substituted carbons of the amide monomers. 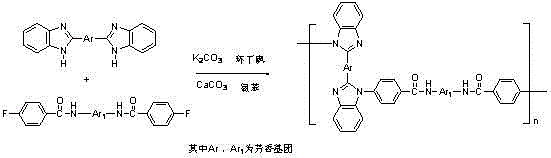 This reaction is catalyzed by anhydrous potassium carbonate in a high-boiling solvent system comprising sulfolane and chlorobenzene, with calcium carbonate acting as a fluoride scavenger. The precise control of stoichiometry and the use of specific additives ensure that the polymerization proceeds to high conversion, minimizing the presence of low-molecular-weight oligomers that could act as impurities. The resulting zigzag chain structure increases the free volume within the polymer matrix, which is directly responsible for the observed improvements in toughness and impact resistance compared to linear analogues.
This reaction is catalyzed by anhydrous potassium carbonate in a high-boiling solvent system comprising sulfolane and chlorobenzene, with calcium carbonate acting as a fluoride scavenger. The precise control of stoichiometry and the use of specific additives ensure that the polymerization proceeds to high conversion, minimizing the presence of low-molecular-weight oligomers that could act as impurities. The resulting zigzag chain structure increases the free volume within the polymer matrix, which is directly responsible for the observed improvements in toughness and impact resistance compared to linear analogues.
From an impurity control perspective, the choice of reagents and conditions is meticulously designed to minimize side reactions that could compromise the thermal performance of the final product. The use of calcium carbonate is particularly strategic, as it effectively traps fluoride ions generated during the polymerization, preventing them from catalyzing degradation pathways or remaining as corrosive residues in the final resin. Furthermore, the purification steps involving precipitation in water and extraction with methanol ensure that residual salts and unreacted monomers are thoroughly removed. This rigorous attention to purity is essential for applications in the semiconductor and aerospace industries, where even trace ionic contaminants can lead to catastrophic failure of components under high-stress conditions. The mechanism inherently supports the production of high-purity advanced materials by leveraging well-understood organic synthesis principles that are easily monitored and controlled at scale.
How to Synthesize N-Substituted Polybenzimidazole Amide Efficiently
The synthesis protocol described in the patent offers a clear roadmap for producing these high-performance polymers with consistent quality. The process is divided into three distinct stages: the preparation of the bis(benzimidazole) intermediate, the synthesis of the difluorinated amide monomer, and the final polycondensation. Each stage utilizes readily available industrial chemicals, such as terephthalic acid, o-phenylenediamine, and 4-fluorobenzoyl chloride, which simplifies the sourcing strategy for procurement teams. The reaction conditions, while requiring elevated temperatures up to 210°C for the final step, are well within the capabilities of standard stainless steel reactors equipped with nitrogen protection and water separators. Detailed standardized synthesis steps see the guide below.
- Prepare bis(benzimidazole) intermediates by reacting aromatic dicarboxylic acids with o-phenylenediamine in polyphosphoric acid at 200°C.
- Synthesize difluorinated amide monomers by reacting aromatic diamines with 4-fluorobenzoyl chloride using triethylamine as a catalyst.
- Perform polycondensation of the two intermediates in sulfolane with potassium carbonate and calcium carbonate at temperatures up to 210°C.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement managers, the adoption of this N-substituted polybenzimidazole amide technology offers substantial strategic benefits beyond mere technical performance. The reliance on commodity chemicals for the monomer synthesis significantly de-risks the supply chain, as these raw materials are produced globally in large volumes, ensuring continuity of supply even during market fluctuations. The elimination of exotic or highly specialized catalysts further reduces the complexity of the manufacturing process, leading to significant cost reduction in polymer manufacturing. By avoiding the need for expensive transition metal catalysts or complex purification trains required for other high-performance polymers, producers can achieve a more favorable cost structure that makes these advanced materials competitive with lower-tier engineering plastics. This economic efficiency is critical for expanding the market penetration of high-performance polymers into broader industrial applications where cost sensitivity is a primary decision factor.
- Cost Reduction in Manufacturing: The synthetic route leverages inexpensive and widely available starting materials such as aromatic dicarboxylic acids and simple diamines, which drastically lowers the raw material cost basis compared to proprietary monomers used in competing technologies. The process avoids the use of precious metal catalysts, relying instead on abundant inorganic bases like potassium carbonate, which eliminates the need for costly metal removal steps and reduces waste disposal expenses. Furthermore, the high yield and straightforward workup procedures minimize solvent consumption and energy usage per kilogram of product, contributing to a leaner and more cost-effective production model that enhances overall margin potential for manufacturers.
- Enhanced Supply Chain Reliability: Since the key monomers like terephthalic acid and p-phenylenediamine are established industrial commodities with multiple global suppliers, the risk of single-source dependency is virtually eliminated. This diversification of the supply base ensures that production schedules remain robust against regional disruptions or logistical bottlenecks that might affect more specialized chemical feedstocks. The stability of the supply chain is further reinforced by the fact that the synthesis does not require moisture-sensitive or pyrophoric reagents that demand specialized handling and storage infrastructure, allowing for safer and more flexible logistics operations across international borders.
- Scalability and Environmental Compliance: The polymerization process is designed for scalability, utilizing standard batch reactor configurations that can be easily scaled from pilot plant to commercial production volumes without fundamental changes to the chemistry. The use of calcium carbonate as a fluoride scavenger simplifies waste treatment by converting hazardous fluoride ions into stable solid waste that is easier to manage and dispose of in compliance with environmental regulations. Additionally, the ability to process the final polymer via melt molding reduces the reliance on large volumes of organic solvents during the fabrication phase, aligning with increasingly stringent global standards for volatile organic compound (VOC) emissions and promoting a greener manufacturing footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of N-substituted polybenzimidazole amides. These insights are derived directly from the experimental data and claims within the patent documentation, providing a reliable basis for evaluating the technology's fit for your specific project requirements. Understanding these details is crucial for R&D teams assessing material compatibility and for procurement officers validating supplier capabilities.
Q: What are the thermal stability characteristics of N-substituted polybenzimidazole amides?
A: According to patent CN102604095B, these polymers exhibit glass transition temperatures (Tg) exceeding 210°C and 10% weight loss temperatures above 450°C in nitrogen, indicating exceptional thermal resistance suitable for harsh environments.
Q: How does N-substitution improve the solubility of polybenzimidazole derivatives?
A: The elimination of N-H bonds through N-substitution reduces intermolecular hydrogen bonding, while the introduction of zigzag structures and polar groups like carbonyl and sulfone enhances solubility in common organic solvents such as DMAc and DMSO.
Q: Is this polymerization process scalable for industrial production?
A: Yes, the process utilizes industrially available monomers like terephthalic acid and p-phenylenediamine, and employs standard polycondensation conditions with accessible catalysts like potassium carbonate, facilitating commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Substituted Polybenzimidazole Amide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the N-substituted polybenzimidazole amide technology in driving innovation across the aerospace, electronics, and advanced materials sectors. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory concept to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped to handle the high-temperature polycondensation processes required for this synthesis, maintaining stringent purity specifications through our rigorous QC labs to guarantee that every batch meets the exacting standards demanded by high-tech applications. We are committed to delivering not just a chemical product, but a comprehensive solution that accelerates your time-to-market.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis route to your specific volume and purity requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages of switching to this novel polymer architecture. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that will empower your team to make informed decisions about integrating this high-performance material into your next generation of products.
