Scalable Production of 3,6-Branched Glucan Hexaose via Convergent Glycosylation Strategies
Scalable Production of 3,6-Branched Glucan Hexaose via Convergent Glycosylation Strategies
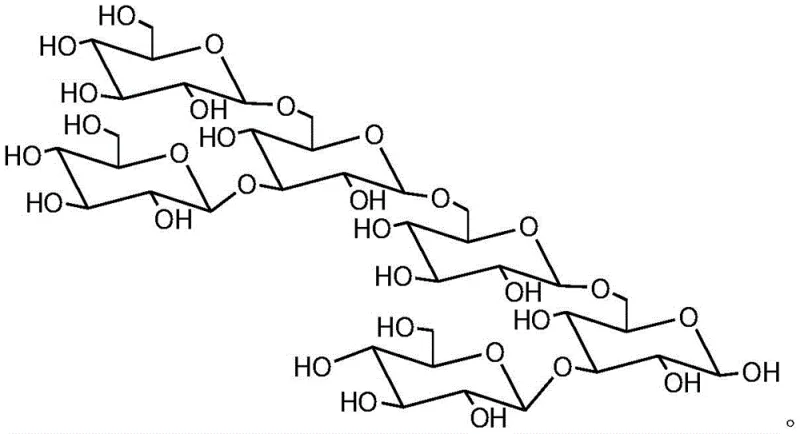
The agricultural biotechnology sector is increasingly recognizing the critical role of oligosaccharide elicitors in sustainable crop protection, driving demand for high-purity intermediates like 3,6-branched glucan hexaose. Patent CN108892740B, published in early 2022, introduces a groundbreaking synthesis method that overcomes the historical bottlenecks of low yield and complex processing associated with this valuable bio-active molecule. This specific oligosaccharide, originally isolated from the cell walls of Phytophthora sojae, functions as a potent immune system activator in plants, triggering defense genes without the ecological risks of traditional pesticides. The disclosed technology leverages a convergent [3+3] synthetic strategy, utilizing trichloroacetimidate glycosyl donors and tert-butyldimethylsilyl (TBS) protected acceptors to achieve superior efficiency. For global supply chain leaders, this patent represents a pivotal shift towards more reliable agrochemical intermediate supplier capabilities, ensuring consistent availability of this high-value plant growth regulator.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of branched oligosaccharides has been plagued by inefficient linear approaches that suffer from diminishing returns as the chain length increases. Traditional methods often rely on stepwise elongation, where each additional sugar unit requires a full cycle of protection, activation, coupling, and deprotection, leading to a drastic reduction in overall yield. Furthermore, the stereochemical control required to establish the specific beta-1,6 main chain and beta-1,3 side chain linkages is notoriously difficult to maintain over multiple steps without specialized catalysts. These legacy processes frequently result in complex impurity profiles that are costly to separate, thereby inflating the cost reduction in agrochemical manufacturing efforts. The reliance on harsh reaction conditions in older protocols also limits the scalability, making it difficult to transition from gram-scale laboratory experiments to ton-scale commercial production without compromising product quality.
The Novel Approach
In stark contrast, the methodology outlined in CN108892740B employs a highly efficient convergent strategy that constructs the hexaose skeleton by coupling two pre-formed trisaccharide blocks. This [3+3] approach effectively halves the number of sequential coupling steps required for the final assembly, significantly mitigating the yield loss typically seen in linear synthesis. The process utilizes robust benzoyl (Bz) and tert-butyldimethylsilyl (TBS) protecting groups which offer orthogonal stability, allowing for precise manipulation of specific hydroxyl groups without affecting the rest of the molecule. By optimizing the molar ratios of donors to acceptors and utilizing mild Lewis acid catalysis, the invention achieves high raw material utilization rates throughout the entire process. This streamlined design not only simplifies the operational workflow but also enhances the feasibility of large-scale industrial production, positioning it as a preferred route for commercial scale-up of complex oligosaccharides.
Mechanistic Insights into TMSOTf-Catalyzed Glycosylation
The core of this synthetic breakthrough lies in the precise application of trimethylsilyl trifluoromethanesulfonate (TMSOTf) as a promoter for glycosylation reactions involving trichloroacetimidate donors. Mechanistically, the TMSOTf activates the trichloroacetimidate leaving group at the anomeric center, generating a highly reactive oxocarbenium ion intermediate that is susceptible to nucleophilic attack by the acceptor hydroxyl group. This activation occurs efficiently at room temperature (25°C), which is crucial for preserving the integrity of the acid-sensitive glycosidic bonds and preventing side reactions such as elimination or rearrangement. The use of dichloromethane as the solvent further facilitates this interaction by providing a non-polar environment that stabilizes the ionic intermediates while maintaining solubility for the lipophilic protected sugar derivatives. The stereoselectivity is carefully managed through the participation of neighboring group effects from the C-2 benzoyl esters, ensuring the formation of the desired beta-linkages with high fidelity.
Impurity control is inherently built into this mechanism through the strategic selection of protecting groups and reaction stoichiometry. The benzoyl groups not only direct stereochemistry but also increase the crystallinity of the intermediates, facilitating purification via standard column chromatography or recrystallization techniques. The final deprotection sequence involves a two-stage hydrolysis: first, mild acid hydrolysis removes the terminal TBS and benzylidene groups, followed by basic hydrolysis (Zemplén deprotection) to cleave the benzoyl esters. This sequential removal prevents the formation of partially deprotected byproducts that often contaminate the final batch. The result is a final product with a clean impurity profile, meeting the stringent purity specifications required for biological applications in agriculture. This rigorous control over the reaction pathway ensures that the synthesized compound retains its biological activity as a potent plant immune elicitor.
How to Synthesize 3,6-Branched Glucan Hexaose Efficiently
The practical implementation of this synthesis route is designed for reproducibility and ease of handling, making it accessible for both research laboratories and pilot plants. The process begins with the independent preparation of the trisaccharide donor and acceptor blocks, which can be stockpiled to streamline the final coupling step. Detailed operational parameters, including specific solvent volumes, catalyst loading (typically roughly 10 mol% TMSOTf), and reaction times ranging from 2 to 6 hours, are optimized to maximize conversion. The subsequent workup procedures utilize standard extraction and drying techniques, avoiding the need for specialized equipment. For a comprehensive breakdown of the exact reagent quantities and step-by-step instructions, please refer to the standardized guide below.
- Synthesize the glucose trisaccharide donor by coupling benzoyl glucose trichloroacetimidate with tert-butyldimethylsilyl-alpha-D-glucopyranose using TMSOTf catalysis, followed by benzoyl protection and activation.
- Prepare the glucose trisaccharide acceptor by coupling specific glucose donors with tert-butyldimethylsilyl-alpha-D-glucopyranose, followed by selective hydrolysis of the 4,6-benzylidene group.
- Couple the equimolar trisaccharide donor and acceptor under TMSOTf catalysis at room temperature, then perform sequential acid and base hydrolysis to remove protecting groups and obtain the final free hexaose.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis method offers tangible benefits that extend beyond mere chemical elegance. The simplification of the synthetic route directly translates to reduced operational expenditures (OpEx) by minimizing the number of unit operations and man-hours required per batch. The high raw material utilization rate means less waste generation and lower costs associated with raw material procurement, addressing the critical need for cost reduction in agrochemical manufacturing. Furthermore, the ability to conduct key coupling steps at room temperature eliminates the energy costs associated with heating or cryogenic cooling, contributing to a smaller carbon footprint and aligning with modern sustainability goals. These factors collectively enhance the economic viability of producing this high-value elicitor at a commercial scale.
- Cost Reduction in Manufacturing: The convergent nature of the synthesis drastically reduces the number of purification cycles needed compared to linear methods, which significantly lowers solvent consumption and silica gel usage. By eliminating the need for expensive transition metal catalysts and relying on readily available Lewis acids, the process avoids the costly downstream removal of heavy metal residues. This streamlined approach ensures that the cost of goods sold (COGS) remains competitive, allowing for better margin management in the final agrochemical formulation.
- Enhanced Supply Chain Reliability: The reliance on stable, commercially available starting materials such as glucose derivatives and standard protecting group reagents mitigates the risk of supply disruptions. The robustness of the reaction conditions means that production is less susceptible to minor fluctuations in environmental parameters, ensuring consistent batch-to-batch quality. This reliability is essential for reducing lead time for high-purity oligosaccharides, enabling manufacturers to respond quickly to seasonal demands in the agricultural sector without compromising on delivery schedules.
- Scalability and Environmental Compliance: The process is inherently scalable due to its simple operational steps and the use of common organic solvents that are easily recovered and recycled. The final product is fully biodegradable and non-toxic, aligning with strict environmental regulations regarding pesticide residues and ecological impact. This compliance reduces the regulatory burden on manufacturers and facilitates faster market entry for new plant protection products based on this active ingredient, securing long-term market access.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 3,6-branched glucan hexaose. These insights are derived directly from the technical specifications and experimental data provided in the patent literature, ensuring accuracy and relevance for industry stakeholders. Understanding these details is crucial for evaluating the feasibility of integrating this intermediate into your existing product pipelines.
Q: What represents the primary advantage of the 3+3 convergent synthesis strategy described in CN108892740B?
A: The primary advantage is the significant improvement in overall yield and raw material utilization compared to traditional linear synthesis. By constructing two trisaccharide blocks independently and coupling them in the final stage, the process minimizes the accumulation of impurities and reduces the number of purification steps required for long-chain intermediates.
Q: Which catalyst system is utilized for the glycosylation reactions in this protocol?
A: The protocol utilizes trimethylsilyl trifluoromethanesulfonate (TMSOTf) as a Lewis acid catalyst. This catalyst is highly effective for activating trichloroacetimidate donors at room temperature (25°C), allowing for mild reaction conditions that preserve the stereochemical integrity of the sensitive glycosidic linkages.
Q: How does this synthesis method address environmental and scalability concerns?
A: The method addresses these concerns through high atom economy and the use of robust protecting groups like benzoyl and TBS which allow for efficient purification. The final product is biodegradable and non-polluting, and the simplified operational steps facilitate easier scale-up from laboratory to industrial production without requiring exotic reagents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,6-Branched Glucan Hexaose Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced oligosaccharide synthesis in modern agriculture and are committed to bridging the gap between academic innovation and industrial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry described in CN108892740B can be seamlessly translated into large-scale manufacturing. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3,6-branched glucan hexaose meets the highest standards of quality and consistency required by global agrochemical companies.
We invite you to collaborate with us to optimize your supply chain and reduce your production costs through our advanced manufacturing capabilities. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our expertise can support your strategic goals in the development of next-generation plant immune elicitors.
