Advanced Synthesis of Large Conjugated Diamine Monomers for High-Performance Polyimides
The landscape of high-performance polymer engineering is undergoing a significant transformation, driven by the demand for materials that combine thermal stability with processability and functional optical properties. Patent CN102153480A introduces a groundbreaking class of novel functional diamine monomers characterized by a large conjugated structure. These monomers are specifically designed to overcome the inherent solubility limitations of traditional polyimides while introducing unique aggregation-induced luminescence (AIE) capabilities. By leveraging a sophisticated synthetic strategy that integrates Wittig olefination with palladium-catalyzed cross-coupling, this technology enables the creation of non-planar molecular architectures that prevent tight chain packing. This structural innovation is critical for R&D directors seeking next-generation materials for microelectronics and optoelectronic devices where both mechanical robustness and light-emitting properties are paramount.
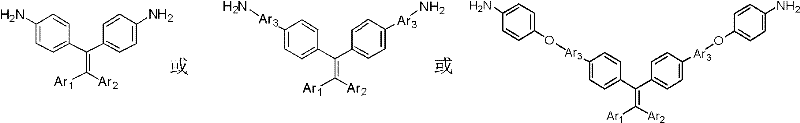
For procurement managers and supply chain heads, the significance of this patent lies in its reliance on accessible starting materials and robust reaction conditions. The synthesis avoids the prohibitive costs associated with fluorinated monomers, which have historically been the primary solution for enhancing polyimide solubility. Instead, it utilizes dihalogenated benzophenones and common boronic acid derivatives, creating a supply chain that is both resilient and cost-effective. The ability to produce high-purity intermediates through straightforward precipitation and crystallization steps further reduces downstream processing costs, making this a highly attractive route for the commercial scale-up of complex electronic chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the modification of polyimides to improve solubility and processability has relied heavily on three main strategies, each with distinct drawbacks that hinder widespread adoption in cost-sensitive markets. The first approach involves the incorporation of fluorine-containing monomers; while effective at reducing intermolecular forces, the exorbitant price of fluorinated precursors severely limits their application to niche, high-margin sectors. The second strategy employs alicyclic monomers to synthesize semi-aromatic polyimides, but this often comes at the expense of thermal stability and mechanical strength, which are the defining characteristics of high-performance engineering plastics. The third method utilizes bulky steric groups to disrupt chain linearity; however, many existing monomers in this category lack the sophisticated spatial design required to impart additional functionality, such as luminescence or specific electronic switching behaviors. Furthermore, traditional polyamic acid precursors are notoriously sensitive to moisture, complicating storage and logistics for supply chain managers who require long shelf-life stability.
The Novel Approach
The methodology disclosed in CN102153480A represents a paradigm shift by constructing a large, non-planar conjugated system that intrinsically balances solubility with thermal performance. The core innovation involves transforming a ketone carbonyl group into a double bond via a Wittig or Wittig-Horner reaction, creating a rigid yet twisted stilbene-like core. This central unit is then expanded through Suzuki coupling reactions, allowing for the precise attachment of diverse functional groups such as amines or hydroxyls. This modular approach not only ensures excellent solubility in common organic solvents—facilitating easier film casting and processing—but also imparts remarkable aggregation-induced luminescence. Unlike conventional fluorinated routes that merely solve solubility, this novel approach adds value by enabling the development of smart materials for polymer memory storage and light-emitting applications, thereby opening new revenue streams for manufacturers of specialty chemical intermediates.
Mechanistic Insights into Wittig-Suzuki Sequential Synthesis
The chemical elegance of this synthesis lies in the sequential application of carbon-carbon bond-forming reactions to build molecular complexity with high precision. The process initiates with the conversion of dihalogenated benzophenone into an aromatic dibromo-stilbene intermediate. In this critical step, a phosphonium ylide or phosphonate carbanion attacks the electrophilic carbonyl carbon, followed by elimination to form the central alkene double bond. This step is crucial as it establishes the non-planar geometry of the molecule; the steric hindrance around the double bond prevents the aromatic rings from lying flat, effectively disrupting pi-pi stacking interactions that typically lead to insolubility in polyimides. The reaction conditions, often involving strong bases like potassium tert-butoxide at elevated temperatures (around 150°C), ensure high conversion rates and minimize side products, resulting in intermediates with yields exceeding 90% as demonstrated in the patent examples.
Following the formation of the stilbene core, the synthesis proceeds via a palladium-catalyzed Suzuki-Miyaura cross-coupling reaction. This step links the dibromo-stilbene intermediate with substituted aryl boronic acids, such as p-aminophenylboronic acid or p-hydroxyphenylboronic acid. The catalytic cycle involves the oxidative addition of the palladium(0) species into the carbon-bromine bond, transmetallation with the boronate complex, and reductive elimination to forge the new carbon-carbon bond. This reaction is highly tolerant of functional groups, allowing for the direct introduction of amine or protected amine functionalities without the need for extensive protecting group chemistry. For variants requiring ether linkages, a nucleophilic aromatic substitution is employed followed by catalytic reduction of nitro groups using hydrazine hydrate and Pd/C. This multi-step cascade ensures that the final diamine monomer possesses a highly defined structure with minimal impurities, a critical factor for R&D teams focusing on the dielectric and optical consistency of the final polymer.
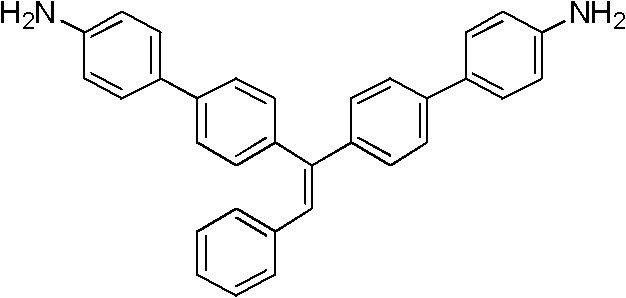
How to Synthesize 4',4''-(2-phenylethene-1,1-diyl)dibiphenyl-4-amine Efficiently
The synthesis of the representative monomer TriPEDBA serves as a benchmark for the entire class of compounds described in the patent. The process begins with the preparation of the key intermediate, 4,4'-(2-phenylethene-1,1-diyl)bis(bromobenzene), via a Wittig-Horner reaction between benzyl chloride derivatives and di-bromobenzophenone. This intermediate is then subjected to Suzuki coupling with p-aminophenylboronic acid hydrochloride in the presence of a palladium catalyst and a base like potassium carbonate. The reaction is typically conducted in a mixed solvent system of tetrahydrofuran and water under reflux conditions to ensure complete conversion. Detailed standardized synthesis steps, including exact molar ratios, temperature profiles, and workup procedures for maximizing yield and purity, are outlined below.
- Perform Wittig or Wittig-Horner reaction on dihalogenated benzophenone to convert the ketone carbonyl into a double bond, forming an aromatic dibromo-stilbene intermediate.
- Execute a Palladium-catalyzed Suzuki coupling reaction between the dibromo-stilbene intermediate and substituted boronic acids (such as aminophenylboronic acid) to extend the conjugated system.
- If utilizing the nitro-phenoxy route, perform a final catalytic reduction using hydrazine hydrate and Pd/C to convert nitro groups into the target primary amine functionalities.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers substantial strategic advantages for organizations looking to optimize their raw material costs and supply chain resilience. The shift away from fluorinated monomers eliminates a major cost driver, as fluorine chemistry often requires specialized equipment and expensive reagents that are subject to volatile market pricing. By utilizing commodity chemicals such as benzyl chloride, dibromobenzophenone, and boronic acids, manufacturers can leverage established global supply chains that offer greater stability and lower baseline costs. Furthermore, the high yields reported in the patent examples, often surpassing 80% for the final coupling steps, indicate a material-efficient process that minimizes waste generation and maximizes the output per batch, directly contributing to improved gross margins.
- Cost Reduction in Manufacturing: The elimination of expensive fluorinated precursors and the use of robust, high-yield catalytic reactions significantly lower the cost of goods sold. The purification process relies primarily on precipitation and recrystallization rather than energy-intensive distillation or chromatography, which further reduces utility costs and solvent consumption. This streamlined workflow allows for cost reduction in electronic chemical manufacturing without compromising the high-performance specifications required by end-users in the aerospace and microelectronics sectors.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials mitigates the risk of supply disruptions often associated with exotic or highly specialized reagents. The synthetic pathway is flexible, allowing for the substitution of various aryl boronic acids to tune properties without altering the core process infrastructure. This adaptability ensures that production schedules can be maintained even if specific minor reagents face temporary shortages, providing supply chain heads with the confidence needed for long-term planning and inventory management.
- Scalability and Environmental Compliance: The reaction conditions described, such as refluxing in THF or ethanol, are readily scalable from laboratory glassware to industrial reactors. The use of heterogeneous catalysts like Pd/C in the reduction step facilitates easy catalyst recovery and recycling, aligning with modern green chemistry principles. Additionally, the solid nature of the intermediates and final products simplifies handling and transport, reducing the logistical complexities and safety risks associated with liquid hazardous materials, thereby ensuring smoother commercial scale-up of complex polymer additives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel diamine monomer technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for stakeholders evaluating the feasibility of integrating these materials into their product portfolios.
Q: What are the key advantages of this diamine monomer over traditional fluorinated monomers?
A: Unlike expensive fluorinated monomers, this novel diamine utilizes a large non-planar conjugated structure that disrupts chain packing, enhancing solubility without the high cost of fluorine chemistry. Additionally, it introduces aggregation-induced luminescence (AIE), a property absent in standard polyimide precursors.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the patent explicitly states the process is suitable for industrial production due to simple synthesis steps and easy purification methods like precipitation and crystallization. The reactions utilize standard reagents like t-BuOK and Pd catalysts under manageable temperatures (70°C to 150°C).
Q: What specific optical properties do polymers derived from this monomer exhibit?
A: Polymers synthesized from these monomers exhibit significant aggregation-induced luminescence (AIE) effects. For instance, the TriPEDBA derivative shows a strong fluorescence peak at 464nm, making it highly valuable for optoelectronic applications and polymer storage materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Functional Diamine Monomer Supplier
As the demand for advanced polyimides with tailored optical and electrical properties continues to surge, partnering with an experienced CDMO is essential for translating laboratory innovations into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from pilot studies to full-scale manufacturing is seamless and efficient. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of functional diamine monomer meets the exacting standards required for high-performance polymer synthesis, minimizing the risk of downstream processing failures.
We invite you to collaborate with our technical team to explore how this patented technology can enhance your product lineup. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your operation. We encourage potential partners to contact our technical procurement team to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the highest level of technical confidence and supply security.
