Advanced Synthetic Route for Lentinan Core Fragment Heptasaccharide: Scalability and Purity Insights
Advanced Synthetic Route for Lentinan Core Fragment Heptasaccharide: Scalability and Purity Insights
The pharmaceutical industry's pursuit of potent immunomodulators has increasingly focused on defined oligosaccharide structures derived from medicinal fungi. Patent CN114736315B, published in March 2023, introduces a groundbreaking method for synthesizing the heptasaccharide core fragment of lentinan, featuring a beta-(1→6) branched chain on a beta-(1→3) main chain. This specific structural motif is critical for the antineoplastic activity of lentinan, yet obtaining it via natural extraction often results in heterogeneous mixtures with uncertain molecular weights. The disclosed invention provides a robust chemical synthesis pathway that ensures structural homogeneity and high purity, addressing a significant bottleneck in the development of next-generation carbohydrate-based therapeutics. By utilizing a strategic combination of trichloroacetimidate donors and thioglycoside acceptors activated by trimethylsilyl trifluoromethanesulfonate (TMSOTf), the process achieves high stereocontrol and yield. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this technology represents a pivotal shift towards more controllable and scalable manufacturing of complex glycans.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of lentinan fragments has been plagued by inefficient protection group strategies and cumbersome purification steps. Prior art, such as the methods described in CN1303857A and CN1336387A, often relied on allyl groups for terminal protection. While allyl groups offer orthogonality, their removal typically necessitates the use of noble metal catalysts like palladium, which introduces significant cost implications and the risk of heavy metal residues in the final API. Furthermore, traditional routes frequently involve repetitive cycles of protection and deprotection for every glycosidic bond formation. This stepwise approach not only extends the production timeline drastically but also accumulates yield losses at each stage, making the commercial scale-up of complex oligosaccharides economically challenging. The reliance on toxic reagents for deprotection and the difficulty in removing certain protecting groups completely have hindered the widespread adoption of synthetic lentinan fragments in clinical settings.
The Novel Approach
The methodology outlined in CN114736315B circumvents these historical hurdles through a cleverly designed convergent strategy. Instead of relying on difficult-to-remove allyl groups, the inventors utilize a combination of acetyl, benzyl, and isopropylidene protecting groups that can be manipulated under milder and more cost-effective conditions. A standout feature of this novel approach is the implementation of a stereoselective one-pot synthesis for the tetrasaccharide acceptor. By precisely regulating the addition sequence and reaction time of different glycosyl donors, the process constructs the tetrasaccharide backbone without isolating unstable intermediates. This consolidation of steps significantly enhances the utilization rate of raw materials and simplifies the operational workflow. The final assembly involves coupling a pre-formed trisaccharide donor with the tetrasaccharide acceptor, followed by a global deprotection sequence that yields the target heptasaccharide with exceptional purity. This streamlined route directly translates to cost reduction in API manufacturing by minimizing unit operations and solvent usage.
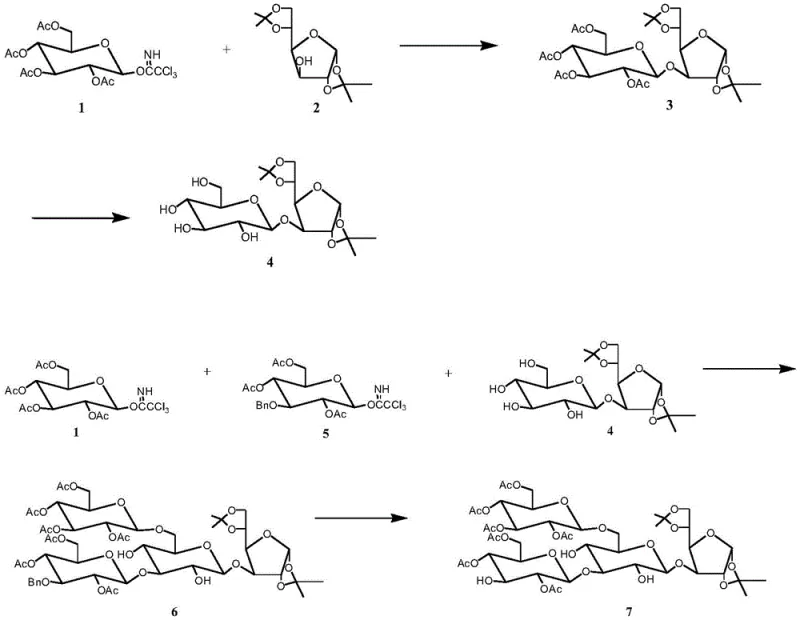
Mechanistically, the success of this synthesis hinges on the precise activation of glycosyl donors using Lewis acids. The process predominantly employs TMSOTf as a catalyst to activate trichloroacetimidate donors, which are highly reactive and allow for efficient glycosidic bond formation at mild temperatures, typically around 25°C. The use of 2,3,4,6-tetra-O-acetyl glucose trichloroacetimidate as a recurring building block ensures consistency in the stereochemical outcome, favoring the formation of the desired beta-linkages essential for biological activity. The reaction conditions are optimized to prevent side reactions such as hydrolysis or rearrangement, which are common pitfalls in oligosaccharide synthesis. By maintaining anhydrous conditions in dichloromethane and carefully controlling the stoichiometry of the donors and acceptors, the method achieves high conversion rates. The subsequent deprotection steps, involving methanolamine for acetyl removal and acidic hydrolysis for isopropylidene groups, are designed to be orthogonal, ensuring that the sensitive glycosidic bonds remain intact while the protecting groups are cleanly stripped away.
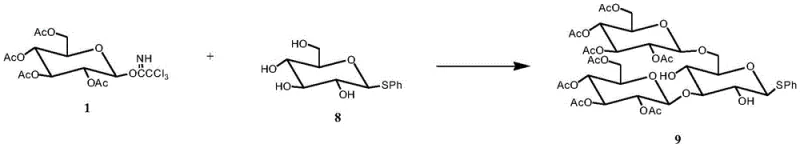
Impurity control is another critical aspect where this patent demonstrates superiority. In conventional syntheses, incomplete deprotection or over-reaction can lead to complex impurity profiles that are difficult to separate from the target molecule. The current method mitigates this by using protecting groups that yield volatile or water-soluble byproducts upon removal. For instance, the removal of acetyl groups generates acetamide derivatives that are easily washed away, while the cleavage of isopropylidene groups produces acetone. The final hydrogenolysis or chemical deprotection of benzyl groups is performed under conditions that minimize degradation of the sugar backbone. This rigorous control over the chemical environment ensures that the final heptasaccharide meets stringent purity specifications required for pharmaceutical applications. The ability to produce a substance with a definite structure and low levels of common impurities facilitates regulatory approval and ensures batch-to-batch consistency, a key requirement for any reliable pharmaceutical intermediate supplier.
How to Synthesize Lentinan Heptasaccharide Efficiently
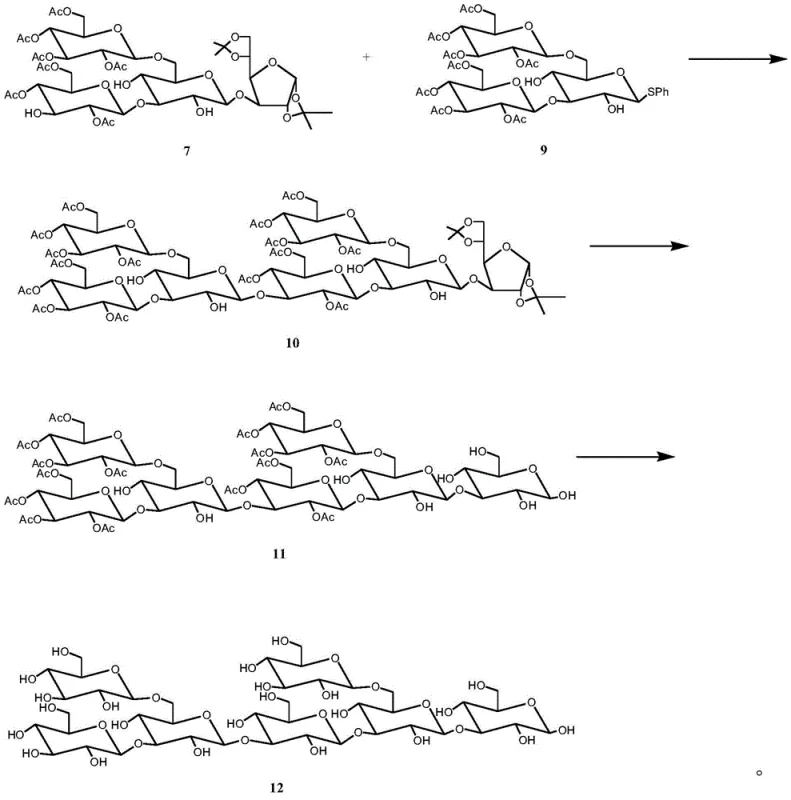
The synthesis protocol detailed in the patent offers a clear roadmap for producing the bioactive heptasaccharide. The process begins with the preparation of key building blocks: a tetrasaccharide acceptor and a trisaccharide donor. These fragments are assembled using TMSOTf-mediated glycosylation in dichloromethane. The detailed standardized synthesis steps, including specific molar ratios, reaction times, and workup procedures, are provided in the technical guide below to ensure reproducibility and safety during scale-up.
- Prepare the tetrasaccharide acceptor by coupling 2,3,4,6-tetra-O-acetyl glucose trichloroacetimidate with 1,2: 5,6-di-O-isopropylidene glucose, followed by deacetylation and further coupling with benzyl-protected donors.
- Synthesize the trisaccharide donor by coupling phenylthioglucose with glucose trichloroacetimidate derivatives under Lewis acid conditions.
- Couple the tetrasaccharide acceptor and trisaccharide donor using TMSOTf to form the heptasaccharide skeleton, then remove all protecting groups including isopropylidene, acetyl, and benzyl groups to yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from extraction-based or legacy synthetic methods to this new protocol offers substantial strategic benefits. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By eliminating the need for specialized allyl-protected sugars and noble metal catalysts, the process relies on commodity chemicals that are readily available in the global market. This shift reduces the risk of supply disruptions caused by the scarcity of exotic reagents. Furthermore, the one-pot nature of the tetrasaccharide synthesis reduces the number of isolation and purification steps, which directly correlates to lower labor costs and reduced facility occupancy time. The overall efficiency gains mean that manufacturers can respond more rapidly to market demand fluctuations, enhancing supply chain reliability.
- Cost Reduction in Manufacturing: The economic impact of this synthesis route is profound, primarily driven by the elimination of expensive catalytic systems and the reduction of process steps. Traditional methods often require palladium-catalyzed deprotection, which not only incurs high material costs but also necessitates expensive downstream processing to remove trace metals to meet regulatory limits. By avoiding these noble metals entirely, the new method removes a significant cost center. Additionally, the high yields reported in the patent examples, such as the 96.5% yield in the final deprotection step, indicate a highly efficient atom economy. This efficiency minimizes waste generation and maximizes the output per kilogram of starting material, leading to substantial cost savings in large-scale production without compromising quality.
- Enhanced Supply Chain Reliability: Stability in the supply of critical intermediates is paramount for continuous drug manufacturing. This synthetic route enhances reliability by utilizing robust protecting group chemistry that is less sensitive to minor variations in reaction conditions compared to more labile groups. The reagents used, such as trichloroacetimidates and thioglycosides, are stable and can be stored for extended periods, allowing for better inventory management. Moreover, the simplified workflow reduces the dependency on highly specialized operators, making it easier to train staff and maintain consistent production schedules. This operational resilience ensures that the lead time for high-purity immunomodulators can be kept predictable, safeguarding the downstream formulation processes against delays.
- Scalability and Environmental Compliance: Scaling complex carbohydrate synthesis is notoriously difficult due to solubility issues and the accumulation of byproducts. However, the use of dichloromethane as a primary solvent, combined with the high solubility of the intermediates, facilitates smooth scale-up from laboratory to pilot and commercial scales. The environmental footprint is also improved; the avoidance of toxic allyl removal reagents and heavy metals simplifies waste treatment protocols. The byproducts generated are largely organic and manageable within standard industrial waste streams. This alignment with green chemistry principles not only reduces disposal costs but also ensures compliance with increasingly stringent environmental regulations, making the process sustainable for long-term commercial scale-up of complex glycans.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of lentinan fragments. These answers are derived directly from the experimental data and technical disclosures within patent CN114736315B, providing clarity on the feasibility and advantages of this specific manufacturing route for stakeholders evaluating its adoption.
Q: What are the advantages of this synthesis method over previous lentinan fragment routes?
A: Unlike prior art (e.g., CN1336387A) which utilizes allyl groups requiring toxic reagents and noble metal catalysts for removal, this method employs acetyl and benzyl protections that are removed under milder, cost-effective conditions, significantly simplifying the purification process and reducing heavy metal contamination risks.
Q: How does the one-pot strategy impact production efficiency?
A: The patent describes a stereoselective one-pot method for preparing the tetrasaccharide acceptor. By regulating the addition sequence and reaction time of glycosyl donors, intermediate isolation steps are minimized, leading to higher overall yields and reduced solvent consumption compared to stepwise protection-deprotection sequences.
Q: Is this heptasaccharide suitable for clinical antineoplastic applications?
A: Yes, the synthesized heptasaccharide corresponds to the active repeating unit of lentinan, which possesses immunoregulatory and anti-tumor activities. The chemical synthesis ensures a definite structure and high purity, overcoming the molecular weight uncertainty and variability associated with natural extraction.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lentinan Heptasaccharide Supplier
The technological advancements presented in CN114736315B underscore the potential for producing high-value oligosaccharides with unprecedented efficiency and purity. At NINGBO INNO PHARMCHEM, we recognize the complexity involved in translating such sophisticated laboratory protocols into commercial reality. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with state-of-the-art reactors capable of handling sensitive glycosylation reactions under strict anhydrous conditions, ensuring that the stringent purity specifications required for antineoplastic intermediates are consistently met. Our rigorous QC labs employ advanced chromatographic and spectroscopic techniques to verify the structural integrity and beta-linkage stereochemistry of every batch, guaranteeing that the final product matches the bioactivity profile of the natural lentinan core.
We invite pharmaceutical developers and procurement leaders to collaborate with us to leverage this innovative synthesis route for your pipeline. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to evaluate how this optimized manufacturing process can accelerate your drug development timelines and reduce overall production costs. Let us be your trusted partner in bringing this promising immunotherapeutic candidate from bench to bedside.
