Advanced Fed-Batch Fermentation Strategy For High-Purity L-Phenylalanine Manufacturing
Advanced Fed-Batch Fermentation Strategy For High-Purity L-Phenylalanine Manufacturing
The global demand for L-phenylalanine, a critical precursor for the artificial sweetener aspartame and various pharmaceutical intermediates, necessitates robust and high-yield production methods that can withstand the rigors of industrial scale-up. Patent CN101984066A introduces a sophisticated biological method utilizing the recombinant Escherichia coli strain WSH-Z06(pAP-B03) to address the longstanding challenge of acetate inhibition in high-density fermentation. This technology represents a significant leap forward in metabolic engineering, shifting away from simple batch processes to a dynamically controlled fed-batch system that precisely manages carbon source availability. By orchestrating the glucose feed rate across distinct growth phases—lag, logarithmic, and stationary—the process effectively decouples cell growth from byproduct formation, ensuring that the metabolic flux is channeled almost exclusively towards the synthesis of the target amino acid. For procurement specialists and R&D directors alike, this patent offers a blueprint for achieving superior titers without compromising the physiological health of the production organism, thereby securing a more stable and cost-effective supply chain for high-purity L-phenylalanine.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional batch fermentation processes for L-phenylalanine production often suffer from the "Crabtree-like" effect in Escherichia coli, where excessive glucose availability triggers overflow metabolism even under aerobic conditions. In standard batch setups, the initial glucose concentration is typically set high to support maximum growth, but this inevitably leads to the rapid accumulation of acetate, a toxic byproduct that severely inhibits cell division and product synthesis once concentrations exceed 0.5 g/L. This metabolic bottleneck not only caps the final biomass density but also diverts valuable carbon resources away from the shikimate pathway, resulting in disappointingly low yields; for instance, comparative data in the patent shows that a standard batch process with 20 g/L initial glucose produced a mere 4.54 g/L of L-phenylalanine. Furthermore, the accumulation of acetate complicates downstream processing, as the acidic byproduct requires additional neutralization and purification steps, driving up operational expenditures and extending production cycles unnecessarily for manufacturers relying on outdated fermentation protocols.
The Novel Approach
The innovative strategy disclosed in the patent circumvents these metabolic bottlenecks by implementing a multi-stage glucose feeding regime that aligns substrate availability with the physiological capacity of the recombinant strain. Instead of flooding the bioreactor with glucose at the outset, the method initiates fermentation with a controlled low concentration of 20 g/L during the lag phase, followed by an exponential feeding strategy during the logarithmic growth phase that matches the specific cell growth rate. This precise control prevents the onset of overflow metabolism, effectively suppressing acetate formation while maintaining rapid cellular proliferation. As the culture transitions into the stationary phase, the system switches to a constant rate feeding mode to sustain product accumulation without stimulating further biomass growth. This dynamic approach resulted in a dramatic increase in final L-phenylalanine concentration, reaching up to 45.5 g/L in optimized embodiments, thereby demonstrating a clear pathway for cost reduction in amino acid manufacturing through enhanced volumetric productivity and reduced waste generation.
Mechanistic Insights into Glucose-Controlled Fed-Batch Fermentation
The core of this technological advancement lies in the mathematical precision applied to the feeding strategy during the logarithmic growth phase, where the specific cell growth rate (μ) is maintained within a narrow optimal window of 0.2 to 0.3 h⁻¹. By adhering to this specific growth rate, the metabolic burden on the recombinant E. coli is balanced, ensuring that the enzymatic machinery responsible for L-phenylalanine biosynthesis operates at peak efficiency without being overwhelmed by excess carbon flux. The feeding rate (F) is calculated dynamically based on the current biomass (X), reactor volume (V), and the yield coefficient of cells on substrate (Yx/s), as defined by the exponential feeding equation. This ensures that glucose is supplied at the exact rate it is consumed for growth and maintenance, leaving no residual substrate to be converted into inhibitory acetate. Such tight regulation of the microenvironment within the fermenter is crucial for maintaining the stability of the plasmid pAP-B03 and ensuring consistent expression of the key biosynthetic genes pheA(fbr) and aroF(wt) throughout the extended fermentation cycle.
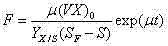
Beyond the feeding mechanics, the process also leverages the specific metabolic characteristics of the WSH-Z06 strain to maximize carbon efficiency. The strain is engineered to co-express feedback-resistant variants of key enzymes in the aromatic amino acid pathway, which removes the natural allosteric inhibition that would otherwise halt production as L-phenylalanine accumulates. When combined with the low-acetate environment created by the controlled feeding strategy, this genetic modification allows the cells to continue synthesizing the target product well into the stationary phase, a period where conventional cultures would typically cease production due to nutrient depletion or toxicity. The result is a fermentation profile where the majority of the product is formed after the primary growth phase has concluded, maximizing the conversion of expensive glucose substrate into high-value L-phenylalanine rather than unnecessary cellular biomass. This mechanism provides a robust foundation for commercial scale-up of complex amino acid intermediates, offering a predictable and reproducible process that minimizes batch-to-batch variability.
How to Synthesize L-Phenylalanine Efficiently
Implementing this high-yield fermentation process requires strict adherence to the defined temporal phases and feeding parameters outlined in the patent documentation to ensure optimal metabolic performance. The synthesis begins with the preparation of a specialized fermentation medium containing essential trace elements such as iron, manganese, and zinc, which serve as cofactors for the enzymatic reactions in the shikimate pathway. Operators must carefully monitor the dissolved oxygen and pH levels throughout the 62-hour cycle, adjusting the glucose feed rate in real-time to match the calculated exponential curve during the critical logarithmic phase. Detailed standardized synthetic steps regarding the specific timing of phase transitions and the exact composition of the trace element solution are provided in the technical guide below to assist process engineers in replicating these results.
- Inoculate recombinant E. coli WSH-Z06(pAP-B03) into a medium with a controlled initial glucose concentration of 20g/L during the 0-12 hour lag phase to establish biomass without triggering overflow metabolism.
- During the logarithmic growth phase (12-36 hours), implement exponential glucose feeding based on a specific cell growth rate (μ) between 0.2 and 0.3 h⁻¹ to maintain rapid synthesis while keeping acetate levels below inhibitory thresholds.
- Transition to a constant rate glucose feeding strategy (9-11 g/(h·L)) during the stationary phase (36-62 hours) to sustain product accumulation and achieve final titers exceeding 45 g/L.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this optimized fermentation technology translates directly into tangible operational efficiencies and risk mitigation strategies. The most immediate impact is the drastic increase in volumetric productivity, which means that the same amount of L-phenylalanine can be produced in a fraction of the fermentation volume compared to traditional batch methods. This reduction in required reactor capacity significantly lowers capital expenditure requirements for new facilities and allows existing plants to increase output without physical expansion, addressing the growing global demand for this essential amino acid derivative. Furthermore, the suppression of acetate not only improves yield but also simplifies the downstream purification process, as there is less acidic waste to neutralize and separate, leading to substantial cost savings in utilities and waste treatment. These factors combine to create a more resilient supply chain capable of delivering high-purity L-phenylalanine with greater consistency and lower overall manufacturing costs.
- Cost Reduction in Manufacturing: The elimination of acetate inhibition allows for much higher final titers, reaching over 45 g/L compared to the single-digit yields of batch processes, which drastically reduces the cost per kilogram of the final product. By maximizing the conversion efficiency of glucose to L-phenylalanine, the process minimizes raw material waste and reduces the energy intensity associated with heating, cooling, and aerating large volumes of fermentation broth for extended periods. Additionally, the simplified downstream processing resulting from lower byproduct levels decreases the consumption of purification resins and solvents, further driving down the variable costs associated with commercial scale-up of complex pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The robustness of the fed-batch control strategy ensures high batch-to-batch consistency, reducing the risk of failed runs that can disrupt supply schedules and delay deliveries to key customers. Because the process relies on controlling the feed rate of a common commodity chemical like glucose rather than expensive or scarce specialty substrates, it is less vulnerable to raw material price volatility or supply shortages. This stability allows suppliers to offer more reliable lead times and maintain safety stock levels with greater confidence, ensuring continuous availability of high-purity L-phenylalanine for critical applications in the food and pharmaceutical industries.
- Scalability and Environmental Compliance: The feeding strategy described is fully compatible with standard industrial bioreactor control systems, facilitating seamless technology transfer from pilot scale to multi-ton commercial production without the need for bespoke equipment. The significant reduction in acetate discharge also aligns with increasingly stringent environmental regulations regarding wastewater treatment, as the lower organic load in the effluent reduces the burden on biological treatment plants. This environmental advantage not only mitigates regulatory risk but also enhances the sustainability profile of the manufacturing process, appealing to eco-conscious partners and stakeholders in the global fine chemicals market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this advanced L-phenylalanine production method. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the technology's potential impact on your supply chain. Understanding these details is essential for R&D teams assessing process feasibility and procurement officers negotiating supply agreements for bulk amino acid ingredients.
Q: How does this process control acetate accumulation in E. coli fermentation?
A: The process strictly controls the specific cell growth rate during the logarithmic phase. By limiting glucose availability through exponential feeding matched to a growth rate of roughly 0.2-0.3 h⁻¹, the metabolic flux is directed towards biomass and product formation rather than overflow metabolism, keeping acetate concentrations below the critical inhibitory level of 0.5 g/L.
Q: What yield improvements can be expected compared to traditional batch fermentation?
A: Data from the patent indicates a substantial increase in productivity. While traditional batch fermentation with 20g/L initial glucose yielded only 4.54 g/L of L-phenylalanine, the optimized fed-batch strategy achieved final concentrations up to 45.5 g/L, representing a tenfold improvement in volumetric productivity.
Q: Is this fermentation method scalable for industrial production?
A: Yes, the method utilizes standard fed-batch techniques compatible with large-scale bioreactors. The reliance on glucose feeding profiles rather than complex genetic modifications or exotic substrates ensures that the process can be readily scaled from laboratory to commercial manufacturing volumes using existing infrastructure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Phenylalanine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable production technologies in maintaining a competitive edge in the global fine chemicals market. Our team of expert process engineers possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the fed-batch fermentation strategy described in CN101984066A can be successfully translated into industrial reality. We are committed to delivering high-purity L-phenylalanine that meets stringent purity specifications, supported by our rigorous QC labs which employ advanced analytical methods to verify identity and assay every batch before release. Our facility is designed to handle complex biological and chemical syntheses with the flexibility required to adapt to evolving customer needs while maintaining the highest standards of quality and safety.
We invite you to collaborate with us to leverage these advanced manufacturing capabilities for your specific project requirements. Please contact our technical procurement team to request a Customized Cost-Saving Analysis that demonstrates how optimizing your L-phenylalanine supply chain can improve your bottom line. We are ready to provide specific COA data and route feasibility assessments to support your development goals, ensuring that you have a reliable partner dedicated to your long-term success in the pharmaceutical and food additive sectors.
