Revolutionizing Protected Amino Acid Synthesis: A Safe Liquid-Phase Route for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high purity with operational safety, particularly for critical amino acid derivatives used in peptide drug manufacturing. Patent CN113135842A introduces a groundbreaking preparation method for O-tert-butyl-L-serine methyl ester and its subsequent conversion to O-tert-butyl-L-serine aqueous solutions, addressing long-standing inefficiencies in conventional synthesis. This technology leverages a fully liquid-phase reaction system that eliminates the need for hazardous gaseous reagents, thereby transforming the risk profile of large-scale production. By utilizing thionyl chloride and methanol for initial esterification followed by a novel tert-butyl acetate-mediated etherification, the process achieves a total yield exceeding 72 percent while maintaining exceptional optical integrity. For R&D Directors and Supply Chain Heads, this represents a pivotal shift towards more sustainable and controllable manufacturing protocols that minimize environmental impact and maximize batch consistency without compromising on the stringent quality standards required for active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for producing protected serine derivatives often rely heavily on the use of isobutene gas as the alkylating agent for the tert-butylation step, which introduces significant safety hazards and engineering complexities. Handling pressurized flammable gases requires specialized high-pressure reactors and rigorous safety interlocks, drastically increasing capital expenditure and operational overhead for manufacturing facilities. Furthermore, these gas-liquid reactions are notoriously difficult to control kinetically, often leading to inconsistent conversion rates and the formation of difficult-to-remove byproducts that compromise the optical purity of the final chiral intermediate. The necessity for cryogenic conditions in many traditional protocols to suppress racemization further escalates energy consumption and limits the scalability of the process, creating bottlenecks for procurement teams seeking reliable bulk supplies. Additionally, the multi-step nature of older methodologies frequently results in cumulative yield losses and generates substantial chemical waste, burdening downstream purification units and inflating the overall cost of goods sold for the final API.
The Novel Approach
In stark contrast, the innovative methodology disclosed in the patent utilizes tert-butyl acetate as a safe, liquid-phase source of the tert-butyl group, reacting directly with L-serine methyl ester hydrochloride under mild acidic catalysis. This substitution of a hazardous gas with a stable liquid solvent fundamentally simplifies the reactor design requirements, allowing the reaction to proceed efficiently at room temperature without the need for expensive pressure vessels or extreme cooling systems. The liquid-phase homogeneity ensures superior mass transfer and heat distribution, leading to more consistent reaction kinetics and significantly improved reproducibility across different batch sizes from laboratory to commercial scale. By avoiding the use of isobutene, the process inherently reduces the risk of explosion and fire, aligning perfectly with modern environmental health and safety regulations while streamlining the regulatory approval process for new manufacturing sites. This approach not only enhances the safety profile but also facilitates easier workup procedures, as the reaction mixture can be directly processed through standard extraction and crystallization techniques without complex gas scrubbing or venting systems.
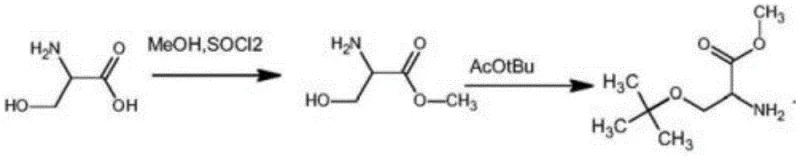
Mechanistic Insights into Acid-Catalyzed Etherification and Esterification
The core chemical transformation in this novel pathway involves a sophisticated dual-function mechanism where esterification and etherification are orchestrated to preserve the chiral center of the L-serine backbone. Initially, L-serine undergoes esterification with methanol in the presence of thionyl chloride, generating the hydrochloride salt which activates the carboxylic acid moiety while protecting the amine group in its protonated form. Subsequently, the addition of a strong acid catalyst, such as perchloric acid or sulfuric acid, promotes the cleavage of tert-butyl acetate to generate the reactive tert-butyl cation in situ within the liquid medium. This electrophilic species selectively attacks the hydroxyl group of the serine side chain to form the stable tert-butyl ether linkage, a critical protection group for preventing side reactions during subsequent peptide coupling steps. The choice of acid catalyst and the precise stoichiometric ratio of tert-butyl acetate are paramount, as they dictate the rate of cation generation and minimize the potential for acid-catalyzed racemization which could degrade the optical purity of the product. Understanding this mechanistic nuance allows process chemists to fine-tune reaction parameters to maximize yield while ensuring that the stereochemical integrity remains above 99.9 percent ee, a crucial metric for high-value pharmaceutical intermediates.
Impurity control is rigorously managed through the specific selection of reaction conditions and workup procedures that target the removal of unreacted starting materials and acidic byproducts. The patent details a meticulous purification strategy involving pH adjustment and liquid-liquid extraction, which effectively separates the organic product from inorganic salts and residual acids that could catalyze degradation during storage. By washing the organic layer with specific alkaline solutions and employing anhydrous drying agents, the process ensures that moisture content is reduced to negligible levels, preventing hydrolysis of the ester or ether groups. The final crystallization step, utilizing solvent systems like dioxane and n-hexane, is designed to selectively precipitate the desired needle-like crystals while leaving amorphous impurities in the mother liquor. This level of control over the solid-state form is essential for ensuring consistent flow properties and dissolution rates in downstream formulation processes, providing procurement managers with a material that meets strict pharmacopeial specifications for physical appearance and chemical identity.
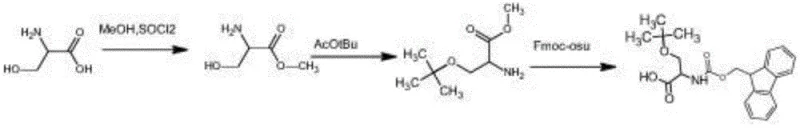
How to Synthesize O-tert-butyl-L-serine Methyl Ester Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for replicating this high-efficiency process, beginning with the careful addition of thionyl chloride to a methanol suspension of L-serine under controlled stirring conditions. Following the reflux and concentration steps to isolate the intermediate hydrochloride salt, the critical etherification is performed by mixing the salt with tert-butyl acetate and the chosen acid catalyst at ambient temperature for an extended period to ensure complete conversion. Detailed standardized synthesis steps see the guide below.
- Reflux L-serine with methanol and thionyl chloride to form L-serine methyl ester hydrochloride.
- React the ester hydrochloride with tert-butyl acetate and an acid catalyst at room temperature to achieve O-tert-butylation.
- Perform saponification and subsequent Fmoc protection to yield the final high-purity amino acid derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this liquid-phase synthesis route offers profound strategic advantages that extend beyond simple chemical yield improvements to impact the entire operational budget. By eliminating the requirement for high-pressure infrastructure and hazardous gas handling systems, manufacturing partners can significantly reduce capital investment costs and lower insurance premiums associated with chemical processing risks. The use of common, commercially available solvents like tert-butyl acetate and methanol ensures a stable supply chain that is less susceptible to the volatility often seen with specialized gaseous reagents, thereby enhancing supply continuity and reducing the risk of production stoppages. Furthermore, the ability to run reactions at room temperature drastically cuts energy consumption related to heating and cooling, contributing to a lower carbon footprint and aligning with corporate sustainability goals that are increasingly important for global pharmaceutical buyers. These operational efficiencies translate directly into a more competitive cost structure, allowing for better margin management without sacrificing the high-quality standards demanded by regulatory bodies.
- Cost Reduction in Manufacturing: The elimination of expensive high-pressure reactors and the associated safety monitoring systems leads to substantial capital expenditure savings for manufacturing facilities adopting this technology. By utilizing liquid reagents that are easier to store and handle, the process reduces logistical costs and minimizes the need for specialized containment equipment, resulting in a leaner and more cost-effective production line. The simplified workup procedure also decreases the consumption of auxiliary materials and reduces waste disposal fees, further driving down the overall cost of goods sold for the final protected amino acid derivative.
- Enhanced Supply Chain Reliability: Relying on stable liquid raw materials rather than pressurized gases mitigates the risk of supply disruptions caused by transportation restrictions or storage limitations. The robustness of the room-temperature reaction conditions ensures that production can be maintained consistently across different geographical locations without the need for highly specialized technical expertise or infrastructure. This reliability is crucial for maintaining just-in-time inventory levels and ensuring that downstream peptide synthesis operations are not delayed by raw material shortages or quality deviations.
- Scalability and Environmental Compliance: The liquid-phase nature of the reaction facilitates seamless scale-up from pilot plant to commercial tonnage production without the engineering challenges associated with gas-liquid mass transfer limitations. The process generates fewer hazardous emissions and utilizes solvents that are easier to recover and recycle, supporting stricter environmental compliance standards and reducing the regulatory burden on manufacturing sites. This scalability ensures that suppliers can meet increasing market demand for high-purity intermediates while maintaining a sustainable and environmentally responsible manufacturing footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route, based on the detailed data provided in the patent documentation. These insights are designed to clarify the operational benefits and quality assurances associated with the liquid-phase methodology for key decision-makers evaluating this technology for their supply chains.
Q: Why is the liquid-phase method superior to traditional isobutene gas methods?
A: The liquid-phase method eliminates the need for hazardous isobutene gas and high-pressure equipment, significantly improving operational safety and reducing capital expenditure on specialized reactors while maintaining high optical purity.
Q: What is the expected purity profile of the product using this novel route?
A: According to patent data, the process achieves an HPLC purity of over 99.5% with single impurity content controlled below 0.1%, ensuring suitability for sensitive peptide synthesis applications.
Q: Does this process require extreme temperature controls?
A: No, the key etherification step proceeds effectively at room temperature, removing the energy costs and engineering complexities associated with cryogenic conditions often found in traditional serine protection strategies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable O-tert-butyl-L-serine Methyl Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to maintain competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering products with stringent purity specifications and rigorous QC labs testing every batch to guarantee consistency and compliance with international pharmacopeial standards. Our dedication to technical excellence allows us to offer customized solutions that meet the specific needs of complex peptide synthesis projects, providing our partners with a secure and high-quality source of essential building blocks.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your current supply chain and reduce overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this safer and more efficient liquid-phase process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project requirements, ensuring that your development timelines are met with the highest quality materials available in the market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
