Advanced Reppe Carbonylation for Commercial Scale-up of Trans-Strawberry Acid
Advanced Reppe Carbonylation for Commercial Scale-up of Trans-Strawberry Acid
The global demand for high-purity flavor and fragrance intermediates is driving a critical shift towards greener, more efficient synthetic methodologies. Patent CN113336637B introduces a groundbreaking approach to synthesizing trans-2-methyl-2-pentenoic acid, commonly known as strawberry acid, utilizing a sophisticated Reppe carbonylation strategy. This technology addresses the longstanding challenges of toxicity and waste associated with traditional production routes by employing a highly selective rhodium-based catalyst system. For R&D directors and procurement leaders, this represents a significant opportunity to optimize supply chains for fine chemical intermediates while adhering to increasingly stringent environmental regulations. The process leverages commodity feedstocks—piperylene, carbon monoxide, and water—to achieve exceptional atom economy, positioning it as a superior alternative for the reliable supply of strawberry acid in the competitive flavor industry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of strawberry acid has relied on pathways fraught with significant safety and economic drawbacks. One prevalent method involves the aldol condensation of propionaldehyde followed by oxidation using stoichiometric agents like silver nitrate or sodium chlorite. While effective, the silver-based processes incur prohibitive costs due to the expensive oxidant and generate substantial heavy metal waste, complicating disposal and increasing the environmental footprint. Alternatively, sodium chlorite oxidation produces large volumes of chlorine-containing wastewater and waste salts, creating severe pressure on safety and environmental compliance systems. Furthermore, older routes utilizing highly toxic cyanide reagents pose unacceptable risks to personnel and require complex containment infrastructure, making them unsuitable for modern sustainable manufacturing standards.
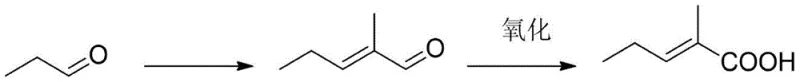
The Novel Approach
In stark contrast, the novel Reppe synthesis method described in the patent offers a streamlined, two-step pathway that fundamentally alters the economic and ecological profile of production. By reacting piperylene directly with carbon monoxide and water in the presence of a specialized rhodium catalyst, the process bypasses the need for hazardous oxidants entirely. This carbonylation step exhibits excellent regioselectivity, primarily yielding 2-methyl-3-pentenoic acid isomers which are then seamlessly converted to the target trans-isomer. The simplicity of using gaseous CO and water as reactants not only reduces raw material costs but also minimizes the generation of inorganic byproducts. This approach aligns perfectly with the principles of green chemistry, offering a scalable solution for the cost reduction in synthetic flavor manufacturing without compromising on yield or purity.
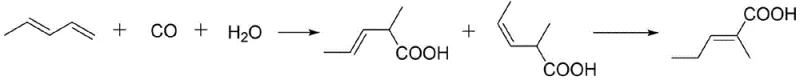
Mechanistic Insights into Rhodium-Catalyzed Carbonylation
The success of this synthesis hinges on the precise engineering of the catalytic system, specifically the synergy between the noble metal precursor and the organic ligand. The patent identifies rhodium salts, particularly Rh2(CO)4Cl2, as the optimal metal source due to their superior activity in hydrocarboxylation reactions compared to palladium or cobalt alternatives. However, the true breakthrough lies in the use of bulky spiro phosphine ligands, such as Ligand 4. These ligands possess a large cone angle and bite angle, creating a rigid steric environment around the rhodium center. This rigidity is crucial for controlling the orientation of the piperylene substrate during the migratory insertion of carbon monoxide, thereby suppressing the formation of unwanted linear or branched isomers and ensuring high selectivity for the desired 2-methyl-3-pentenoic acid backbone.
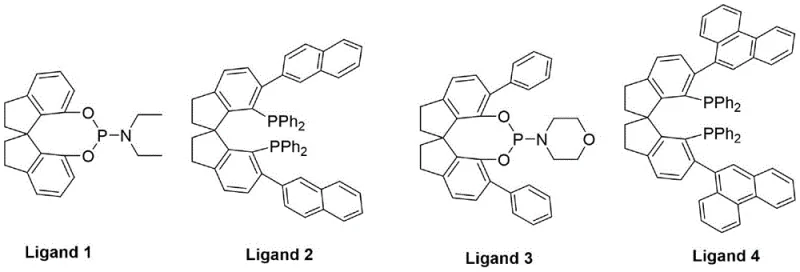
Following the carbonylation, the isomerization step employs a distinct mechanistic pathway driven by a zinc chloride and acetic acid binary catalyst system. Unlike pure zinc chloride, which activates the carbonyl group but fails to control stereochemistry effectively, the addition of acetic acid facilitates the formation of a specific carbenium ion intermediate. This intermediate allows for the thermodynamic equilibration of the double bond, exclusively favoring the formation of the more stable trans-configuration. This mechanistic nuance is vital for R&D teams focused on impurity control, as it eliminates the difficult separation of cis-isomers that plagues other methods. The result is a product stream with inherently high stereochemical purity, reducing the burden on downstream distillation columns and enhancing the overall efficiency of the commercial scale-up of complex flavor intermediates.
How to Synthesize Trans-2-Methyl-2-Pentenoic Acid Efficiently
The operational protocol for this synthesis is designed for robustness and reproducibility in an industrial setting. The process begins with the coordination of the rhodium salt and ligand in a polar aprotic solvent like DMI or HMPA, followed by the introduction of piperylene and water under controlled carbon monoxide pressure. The reaction conditions are mild yet effective, typically operating between 100°C and 130°C at pressures of 2.0 to 3.0 MPa. After the carbonylation is complete, the intermediate is isolated via vacuum rectification, allowing for the recovery and recycling of both the solvent and the valuable rhodium catalyst. The subsequent isomerization is conducted at lower temperatures (60-70°C) using the zinc chloride/acetic acid solution, followed by a simple aqueous workup and final distillation. Detailed standardized synthesis steps see the guide below.
- React piperylene, carbon monoxide, and water using a rhodium salt and spiro phosphine ligand catalyst system to form 2-methyl-3-pentenoic acid isomers.
- Separate the cis-trans isomeric intermediates via vacuum rectification, recovering the solvent and catalyst for reuse.
- Subject the intermediate to isomerization using a zinc chloride/acetic acid solution to quantitatively generate the trans-configured strawberry acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this Reppe-based methodology offers tangible strategic benefits beyond mere technical feasibility. The elimination of stoichiometric oxidants like silver nitrate removes a major variable cost driver and mitigates the supply risk associated with volatile precious metal markets. Furthermore, the ability to recycle the homogeneous rhodium catalyst significantly lowers the effective consumption of noble metals per kilogram of product, contributing to substantial cost savings in the long term. The use of commodity feedstocks such as piperylene and carbon monoxide ensures a stable and continuous supply chain, reducing lead time for high-purity flavor intermediates and insulating production schedules from the bottlenecks often seen with specialized reagents.
- Cost Reduction in Manufacturing: The process achieves significant economic optimization by replacing expensive, single-use oxidants with inexpensive carbon monoxide and water. The high atom economy means that nearly all input mass is converted into valuable product rather than waste, drastically reducing raw material expenditure. Additionally, the recyclability of the rhodium catalyst system minimizes the loss of high-value metals, further driving down the unit cost of production compared to traditional oxidation routes.
- Enhanced Supply Chain Reliability: By relying on widely available petrochemical derivatives like piperylene and industrial gases, the manufacturing process is decoupled from the supply constraints of niche chemical reagents. This ensures consistent production capacity and reduces the risk of delays caused by raw material shortages. The robust nature of the catalyst system also allows for extended campaign runs, enhancing overall equipment effectiveness and delivery reliability for downstream customers.
- Scalability and Environmental Compliance: The absence of hazardous cyanide reagents and chlorine-generating oxidants simplifies waste treatment protocols and reduces the regulatory burden on manufacturing facilities. The process generates minimal three wastes, aligning with modern environmental standards and facilitating easier permitting for capacity expansion. This clean profile supports sustainable growth and ensures long-term operational continuity in regions with strict environmental enforcement.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on performance metrics and operational parameters. Understanding these details is essential for stakeholders evaluating the feasibility of adopting this route for large-scale production.
Q: Why is the Rhodium/Spiro Ligand system superior to Palladium for this Reppe reaction?
A: While palladium salts are common in carbonylation, they often yield mixtures of 2-, 3-, and 4-pentenoic acids with poor selectivity. The specialized rhodium catalyst paired with bulky spiro phosphine ligands creates a rigid coordination environment that strictly directs the carbonyl insertion to the desired position, achieving yields over 82% with minimal byproducts.
Q: How does this method improve environmental compliance compared to traditional oxidation routes?
A: Traditional methods rely on stoichiometric oxidants like silver nitrate or sodium chlorite, generating massive amounts of heavy metal waste or chlorine-containing wastewater. This Reppe synthesis utilizes carbon monoxide and water as nucleophiles, resulting in near-perfect atom economy and eliminating the discharge of hazardous oxidant byproducts.
Q: What ensures the high trans-isomer purity in the final product?
A: The second step utilizes a specific zinc chloride/acetic acid catalyst system. Unlike pure zinc chloride which produces a mix of cis and trans isomers, the acetic acid component facilitates a carbenium ion mechanism that thermodynamically favors the rearrangement exclusively into the stable trans-configuration, simplifying downstream purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trans-2-Methyl-2-Pentenoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to maintain competitiveness in the global flavor and fragrance market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that monitor every batch for isotopic purity and trace impurities. Our capability to implement complex catalytic systems like the Rhodium-Spiro ligand framework demonstrates our technical depth and dedication to quality.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic advantages tailored to your volume needs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your sourcing strategy is built on the most efficient and sustainable chemical technologies available.
