Advanced Flow Chemistry Synthesis of 2,3-Dimethyl-6-Amino-2H-Indazole Hydrochloride for Scalable API Manufacturing
Advanced Flow Chemistry Synthesis of 2,3-Dimethyl-6-Amino-2H-Indazole Hydrochloride for Scalable API Manufacturing
The pharmaceutical industry is constantly seeking robust, safe, and scalable methods for producing complex oncology intermediates, particularly for tyrosine kinase inhibitors like Pazopanib. Patent CN114380748A introduces a groundbreaking synthetic methodology for 2,3-dimethyl-6-amino-2H-indazole hydrochloride, a critical building block in the value chain of this potent anticancer agent. This patent details a transition from hazardous batch processing to a sophisticated continuous flow chemistry platform, addressing long-standing challenges in nitration and hydrogenation safety. By leveraging tubular reactor technology, the invention achieves unprecedented control over exothermic reactions, resulting in superior product quality and operational safety. For R&D directors and supply chain leaders, this technology represents a pivotal shift towards greener, more efficient manufacturing paradigms that align with modern regulatory and environmental standards.
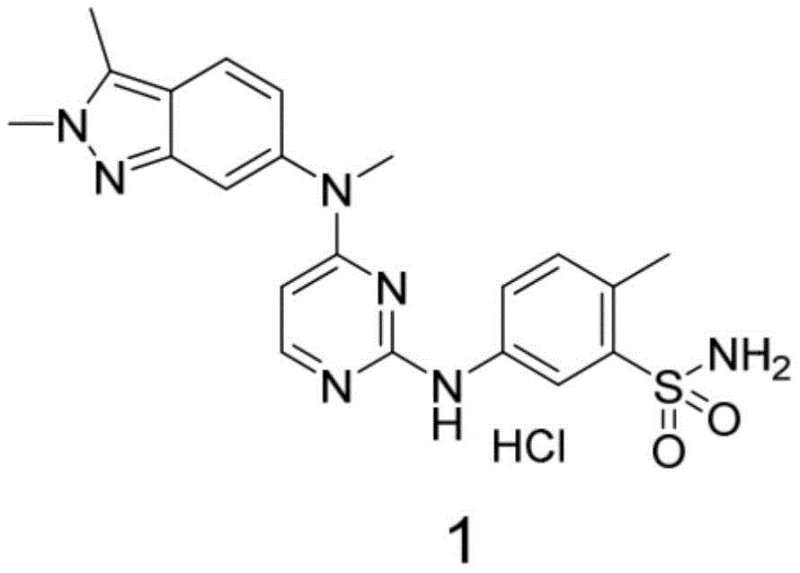
Pazopanib hydrochloride is a multi-target tyrosine kinase inhibitor approved for treating renal cell carcinoma and soft tissue sarcoma, functioning by inhibiting VEGFR-2, PDGFR, and c-Kit. The structural integrity of its key intermediate, 2,3-dimethyl-6-amino-2H-indazole hydrochloride, is paramount for the efficacy of the final drug substance. As shown in the molecular architecture, the indazole core serves as the anchor for the pyrimidine and sulfonamide functionalities. Traditional synthesis routes for this intermediate have been plagued by safety hazards and inconsistent quality, creating bottlenecks in the global supply of this life-saving medication. The new flow-based approach described in the patent offers a viable solution to these industrial pain points, promising a more reliable source of high-purity pharmaceutical intermediates for downstream API synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,3-dimethyl-6-amino-2H-indazole derivatives has relied on traditional batch kettle reactors, a method fraught with significant safety and efficiency drawbacks. The conventional pathway involves a sequence of high-risk reactions, including nitration, diazotization, and catalytic hydrogenation, each presenting unique hazards when performed in large volumes. Nitration is notoriously exothermic; in a batch setting, poor heat transfer can lead to local hot spots, triggering thermal runaways, fires, or even explosions. Furthermore, the diazotization step generates unstable diazonium salts that are prone to decomposition and potential detonation if temperature control slips even slightly. These safety concerns force manufacturers to operate with extreme caution, often at the expense of reaction speed and yield.
Beyond safety, the conventional batch process suffers from inherent inefficiencies that impact cost and purity. The mixing of gas, liquid, and solid phases in a stirred tank is often imperfect, leading to mass transfer limitations that prolong reaction times and increase energy consumption. For instance, catalytic hydrogenation in a batch reactor requires high pressure and extended durations to ensure complete conversion, yet catalyst recovery remains difficult and inefficient. These factors contribute to the formation of isomers and by-products, necessitating complex purification steps that erode overall yield. The cumulative effect is a manufacturing process that is not only dangerous but also economically burdensome and environmentally taxing due to higher solvent usage and waste generation.
The Novel Approach
In stark contrast, the novel approach detailed in patent CN114380748A utilizes a continuous flow tubular reactor system to revolutionize the synthesis of this key intermediate. By confining the reaction to a narrow tube with a high surface-area-to-volume ratio, the system achieves heat transfer efficiencies that are orders of magnitude greater than traditional kettles. This allows for the precise management of exothermic events, effectively neutralizing the risks associated with nitration and diazotization. The continuous flow of reagents ensures that only a minimal amount of hazardous material is present in the reactor at any given time, fundamentally altering the safety profile of the operation. This technological leap transforms high-risk batch operations into safe, controllable, and continuous processes.

The implementation of flow chemistry also brings about dramatic improvements in reaction kinetics and selectivity. The precise control over residence time and temperature prevents the over-reaction or degradation of sensitive intermediates, leading to a significant reduction in impurity profiles. For the hydrogenation step, the flow reactor facilitates intimate contact between the hydrogen gas, liquid substrate, and solid catalyst, enhancing reaction rates and allowing for milder conditions. This results in a streamlined process where the target product, 2,3-dimethyl-6-amino-2H-indazole hydrochloride, is obtained with exceptional purity and yield. The shift from batch to flow is not merely an equipment change; it is a fundamental re-engineering of the chemical process to maximize safety, efficiency, and product quality for commercial scale-up.
Mechanistic Insights into Flow-Chemistry Catalyzed Cyclization and Reduction
The core of this innovative synthesis lies in the intricate interplay between fluid dynamics and chemical kinetics within the tubular reactor. In the nitration step, the rapid mixing of 2-ethylaniline and nitric acid within the cooling module ensures that the heat of reaction is instantly dissipated, preventing the formation of dinitro by-products and oxidation impurities that typically plague batch nitrations. The subsequent diazotization and cyclization occur in a heated zone where the residence time is strictly controlled to seconds, allowing the unstable diazonium intermediate to cyclize into the indazole ring before it can decompose. This kinetic control is impossible to achieve in a batch reactor where heating and cooling ramps are slow and temperature gradients exist throughout the vessel.
Furthermore, the methylation and hydrogenation steps benefit immensely from the homogeneous environment created by the flow system. During methylation, the use of dimethyl carbonate as both solvent and reagent in a heated flow loop ensures complete conversion without the need for harsh alkylating agents. In the final hydrogenation stage, the slurry of Raney nickel catalyst is pumped continuously alongside the substrate and hydrogen gas. The turbulent flow within the reactor keeps the catalyst suspended and maximizes the gas-liquid-solid interface, driving the reduction of the nitro group to the amine with high efficiency. This mechanistic precision minimizes the formation of de-halogenated or over-reduced side products, ensuring that the final API intermediate meets the stringent purity specifications required for oncology drugs.
How to Synthesize 2,3-Dimethyl-6-Amino-2H-Indazole Hydrochloride Efficiently
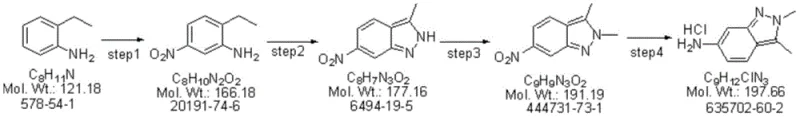
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this continuous manufacturing process. It involves four distinct reaction modules connected in series, each optimized for specific chemical transformations ranging from low-temperature nitration to high-pressure hydrogenation. The process begins with the preparation of feed streams which are metered precisely into the reactor system, ensuring stoichiometric accuracy that is critical for minimizing waste. Detailed operational parameters such as flow rates, temperatures, and pressures are rigorously defined to maintain the steady-state conditions necessary for consistent product quality. For a comprehensive understanding of the specific equipment setup and operational nuances, the standardized synthesis steps are detailed below.
- Nitration: Dissolve 2-ethylaniline in sulfuric acid and react with dilute nitric acid in a tubular reactor cooling module at 0-10°C to obtain the nitro-intermediate.
- Diazotization & Cyclization: React the nitro-intermediate with sodium nitrite in acetic acid within a temperature-controlled flow module to form the indazole ring structure.
- Methylation: Treat the cyclized intermediate with a methylating agent (e.g., dimethyl carbonate) in a heated flow reactor to introduce the N-methyl group.
- Hydrogenation: Reduce the nitro group to an amine using hydrogen gas and a Raney nickel catalyst in a pressurized flow system to yield the final hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this flow chemistry technology translates into tangible strategic advantages beyond mere technical superiority. The primary benefit is the drastic reduction in operational risk, which directly correlates to supply continuity. By eliminating the potential for catastrophic batch failures due to thermal runaways or explosions, manufacturers can guarantee uninterrupted production schedules. This reliability is crucial for maintaining the supply of critical cancer medications, where shortages can have severe consequences. Additionally, the continuous nature of the process allows for a smaller physical footprint and reduced inventory of hazardous raw materials on-site, lowering insurance costs and regulatory compliance burdens associated with storing large quantities of energetic chemicals.
- Cost Reduction in Manufacturing: The transition to flow chemistry drives cost efficiency through multiple mechanisms, primarily by enhancing yield and reducing waste. The superior selectivity of the flow process means that less raw material is lost to by-products, directly lowering the cost of goods sold. Furthermore, the ability to recover and reuse the Raney nickel catalyst more effectively in a continuous loop reduces the consumption of expensive precious metals. The elimination of extensive purification steps, necessitated by the high crude purity of the flow product, further decreases solvent usage and energy costs associated with distillation and crystallization, leading to substantial overall cost savings.
- Enhanced Supply Chain Reliability: Continuous manufacturing inherently supports a more agile and responsive supply chain. Unlike batch processing, which is limited by vessel size and cycle time, flow reactors can operate 24 hours a day, 7 days a week, scaling production simply by extending run times. This flexibility allows suppliers to respond rapidly to fluctuations in market demand for Pazopanib intermediates without the need for massive capital investment in larger tanks. The consistent quality of the output also reduces the risk of batch rejection during QC testing, ensuring that every shipment meets specifications and arrives on time, thereby strengthening the trust between supplier and pharmaceutical partner.
- Scalability and Environmental Compliance: From an environmental perspective, this technology aligns perfectly with green chemistry principles, a growing requirement for global pharma supply chains. The process generates significantly less waste (three wastes) due to higher atom economy and reduced solvent requirements. The containment of hazardous reagents within closed tubing minimizes operator exposure and fugitive emissions, facilitating easier compliance with strict environmental regulations. Scalability is achieved through 'numbering up' rather than 'scaling up,' meaning production capacity can be increased by adding parallel reactor units without changing the reaction chemistry, ensuring that the process remains robust and reproducible from pilot scale to full commercial production.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical feasibility and commercial viability of this flow chemistry synthesis. They are derived from the specific advancements detailed in the patent documentation, focusing on how this method overcomes traditional barriers in intermediate manufacturing. Understanding these aspects is vital for stakeholders evaluating the potential for technology transfer or long-term supply partnerships. The answers reflect the consensus on how continuous processing enhances safety and quality in the production of complex heterocyclic compounds.
Q: How does flow chemistry improve the safety of nitration and hydrogenation reactions?
A: Flow chemistry drastically reduces the inventory of hazardous reagents online at any given moment. The high surface-to-volume ratio of tubular reactors enables superior heat transfer, preventing thermal runaways common in batch nitration. For hydrogenation, the continuous mixing of gas and liquid phases eliminates explosion risks associated with large headspace hydrogen volumes in traditional kettles.
Q: What purity levels can be achieved with this continuous synthesis method?
A: The precise control over residence time and temperature in the flow reactor minimizes side reactions and isomer formation. Patent data indicates that the final product, 2,3-dimethyl-6-amino-2H-indazole hydrochloride, can achieve purity levels exceeding 99%, significantly higher than the 85-90% typically seen in conventional batch processes.
Q: Is this process suitable for large-scale commercial production of Pazopanib intermediates?
A: Yes, the continuous nature of the process allows for 24/7 operation without the downtime associated with batch charging and discharging. The scalability is inherent to flow chemistry; production capacity can be increased by running the reactor for longer periods or numbering up parallel reactor units, ensuring a reliable supply chain for high-volume API manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Dimethyl-6-Amino-2H-Indazole Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of continuous flow chemistry in the production of high-value pharmaceutical intermediates. As a dedicated CDMO partner, we possess the technical expertise and infrastructure to adapt complex synthetic routes like the one described in patent CN114380748A for commercial reality. Our facilities are equipped to handle diverse pathways, ranging from early-stage development to full-scale manufacturing, with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that ensure every batch complies with international regulatory standards for oncology drug substances.
We invite global pharmaceutical companies and procurement leaders to collaborate with us to leverage these advanced manufacturing capabilities. By partnering with NINGBO INNO PHARMCHEM, you gain access to a supply chain that is not only cost-effective but also resilient and sustainable. We encourage you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Let us provide you with specific COA data and route feasibility assessments to demonstrate how our flow chemistry solutions can optimize your supply of 2,3-dimethyl-6-amino-2H-indazole hydrochloride and secure your API production pipeline.
