Advanced Synthesis of 1,3-Dibromoacetone: Technical Upgrade and Commercial Scalability
Advanced Synthesis of 1,3-Dibromoacetone: Technical Upgrade and Commercial Scalability
The pharmaceutical and fine chemical industries constantly seek robust synthetic routes that balance high purity with economic viability, particularly for halogenated intermediates like 1,3-dibromoacetone. Patent CN1956941A introduces a transformative methodology that addresses the longstanding limitations of acetone bromination, shifting the paradigm from equilibrium-limited reactions to crystallization-driven high-yield processes. This technology leverages a sophisticated understanding of halogen exchange equilibria and phase behavior to isolate 1,3-dibromoacetone with exceptional purity, serving as a critical precursor for epichlorohydrin and various agrochemical agents. By integrating rapid mixing protocols with catalytic equilibration using hydrogen bromide, the process effectively suppresses the formation of undesirable poly-brominated byproducts such as tetrabromoacetone. For R&D directors and procurement specialists, this patent represents a significant opportunity to optimize supply chains for reliable pharma intermediates supplier networks, ensuring consistent quality while mitigating the volatility associated with traditional halogenation chemistries.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,3-dihaloacetones has been plagued by poor selectivity and complex purification requirements that inflate operational expenditures. Direct chlorination of acetone, for instance, notoriously produces substantial quantities of 1,1-dichloroacetone and trichloroacetone derivatives, necessitating energy-intensive distillation columns to achieve acceptable purity levels. Alternative approaches, such as the iodine-catalyzed chlorination described in older literature, introduce prohibitive costs due to the high price of iodine and the generation of stoichiometric amounts of hydrogen chloride waste. Furthermore, previous bromination strategies, such as those relying on bisulfite adduct formation, involve multiple discrete chemical steps including addition, filtration, acid decomposition, and recrystallization. These multi-step sequences not only reduce overall throughput but also generate significant volumes of aqueous waste containing sulfur compounds and unreacted brominated species. The thermodynamic equilibrium of acetone dibromination typically caps the concentration of the desired 1,3-isomer at roughly 70%, leaving a substantial portion of the reaction mass as unusable or difficult-to-separate mono- and tri-brominated impurities.
The Novel Approach
The methodology disclosed in CN1956941A circumvents these thermodynamic and kinetic barriers through a clever integration of reaction engineering and separation science. Instead of accepting the 70% equilibrium limit, the process employs a dynamic equilibration strategy where the reaction mixture is treated with hydrogen bromide to facilitate isomer interchange, actively converting 1,1-dibromoacetone and tribromoacetone into the desired 1,3-isomer. Crucially, this equilibration is coupled with reactive crystallization, where the 1,3-dibromoacetone is selectively precipitated from the mixture as it forms. By physically removing the product from the liquid phase, Le Chatelier's principle is exploited to drive the equilibrium further toward completion, potentially achieving concentrations exceeding 95%. This approach eliminates the need for complex adduct chemistry and allows for the direct use of brominated acetone mixtures as solvents for subsequent batches, thereby drastically simplifying the workflow. For stakeholders focused on cost reduction in fine chemical manufacturing, this represents a move from a linear, waste-generating process to a circular, efficiency-maximizing operation.
Mechanistic Insights into HBr-Catalyzed Equilibration and Crystallization
The core chemical innovation lies in the manipulation of the brominated acetone derivative distribution through acid-catalyzed enolization and halogen exchange. When acetone reacts with two moles of bromine, a complex mixture is initially formed containing monobromoacetone, 1,1-dibromoacetone, 1,3-dibromoacetone, and tribromoacetone. The presence of hydrogen bromide acts as a potent catalyst for the keto-enol tautomerism required for halogen migration. Under these acidic conditions, the less stable isomers undergo reversible deprotonation and re-halogenation, gradually shifting the statistical distribution toward the thermodynamically favored 1,3-dibromoacetone. This equilibration is not merely a static waiting game but is dynamically coupled with temperature control to induce crystallization. As the solution is cooled, typically to temperatures between -30°C and the melting point of the pure product, the solubility limit of 1,3-dibromoacetone is exceeded. The nucleation and growth of crystals effectively lock the 1,3-isomer into a solid state, preventing it from participating in further reverse equilibration reactions.
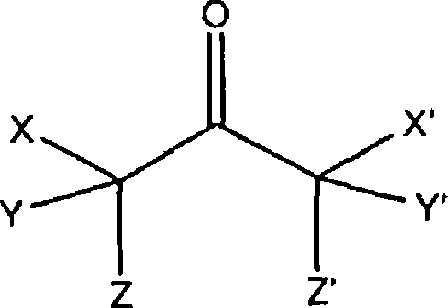
The structural diversity of the reaction intermediates is illustrated by the general formula found in the patent documentation, where the central carbonyl group is flanked by carbon atoms bearing varying degrees of bromination. In this schematic, X represents a bromine atom, while Z and Z' are hydrogen atoms, and the positions Y, Y', and X' can independently be hydrogen or bromine. This variability underscores the challenge of selectivity; without the specific equilibration and crystallization protocol, the reaction yields a statistical mess of these variants. However, by maintaining a hydrogen bromide concentration between 0.01% and 10% during the cooling phase, the system ensures that any 1,1-isomer or tribromo-species remaining in the mother liquor continues to convert into the 1,3-form. This continuous conversion during the crystallization phase is what allows the process to break the traditional 70% yield barrier, delivering a solid product that requires minimal downstream purification to meet stringent pharmaceutical specifications.
How to Synthesize 1,3-Dibromoacetone Efficiently
Implementing this synthesis route requires precise control over mixing dynamics and thermal profiles to ensure safety and reproducibility at scale. The initial bromination step is exothermic and must be managed carefully to prevent runaway reactions or the formation of over-brominated tars. The patent emphasizes the critical importance of rapid mixing upon the addition of bromine to acetone; this homogenizes the reactants before the autocatalytic hydrogen bromide is generated in significant quantities, thereby minimizing localized hot spots that favor tetrabromoacetone formation. Following the initial reaction, the mixture is subjected to an equilibration period, often aided by the addition of exogenous HBr or by recycling HBr-rich off-gases. The final isolation is achieved through controlled cooling crystallization, where the rate of temperature descent dictates the crystal size and purity profile. Detailed standardized operating procedures for scaling this from laboratory glassware to industrial reactors are essential for maintaining these tight controls.
- React acetone with two molar equivalents of bromine under rapid mixing conditions to minimize over-bromination.
- Equilibrate the resulting mixture of brominated acetone derivatives in the presence of hydrogen bromide to maximize 1,3-dibromoacetone concentration.
- Perform reactive crystallization by cooling the equilibrated mixture to isolate solid 1,3-dibromoacetone with purity exceeding 95%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates directly into enhanced margin protection and supply security. The most immediate impact is seen in raw material utilization; by recycling the mother liquors and converting byproduct bromides back into elemental bromine, the process significantly reduces the net consumption of expensive halogen reagents. Traditional methods often discard bromide-rich waste streams, representing a direct loss of valuable atomic resources. In contrast, this closed-loop approach allows for the regeneration of bromine from hydrogen bromide byproducts using standard oxidation techniques, effectively creating an internal supply of one of the most costly inputs. Furthermore, the elimination of iodine catalysts removes a major cost driver and supply chain bottleneck, as iodine prices are historically volatile and subject to geopolitical constraints. The simplification of the purification train—from multiple chemical treatments to a single crystallization step—also reduces utility consumption, specifically steam for distillation and water for washing, leading to substantial operational expenditure savings.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior due to the drastic simplification of unit operations. By avoiding the formation of bisulfite adducts, the plant eliminates the need for sulfur-based reagents and the associated waste treatment costs for sulfate streams. Additionally, the ability to use the brominated acetone mixture itself as a solvent for subsequent batches reduces the demand for external organic solvents like ethyl acetate or dichloromethane. This solvent reduction not only lowers purchase costs but also diminishes the volume of VOC emissions, aligning with increasingly strict environmental regulations. The high selectivity achieved means that less feedstock is wasted on generating off-spec isomers, maximizing the yield per ton of acetone charged and improving the overall asset turnover ratio of the production facility.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the robustness of the chemistry against feedstock variations. Since the process relies on an equilibration mechanism that corrects for initial isomer imbalances, it is more forgiving of minor fluctuations in reactant quality compared to rigid stoichiometric processes. The capability to recycle bromine internally acts as a buffer against external market shortages of elemental bromine, ensuring that production schedules are not disrupted by supplier delays. Moreover, the production of 1,3-dichloroacetone from the dibromo-precursor offers a strategic alternative to direct chlorination, diversifying the sourcing options for downstream epichlorohydrin manufacturers. This flexibility allows supply chain planners to hedge against regional regulatory changes affecting chlorination technologies, securing a long-term, reliable source of high-purity intermediates.
- Scalability and Environmental Compliance: From an EHS (Environment, Health, and Safety) perspective, the process offers distinct advantages for commercial scale-up of complex halogenated ketones. The reactive crystallization occurs at relatively mild temperatures, reducing the thermal load on cooling systems and minimizing the risk of thermal decomposition. The reduction in waste generation is particularly notable; by converting bromide byproducts back to bromine and recycling chloride salts, the process approaches a zero-discharge ideal for halogen species. This minimizes the burden on wastewater treatment plants and reduces the liability associated with hazardous waste disposal. The simplified workflow also reduces the number of potential leak points and operator interventions, enhancing overall plant safety and making it easier to obtain the necessary permits for capacity expansion in regulated jurisdictions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and claims within CN1956941A, providing a factual basis for feasibility assessments. Understanding these nuances is critical for technical teams evaluating the transition from legacy processes to this advanced equilibration-crystallization platform. The data supports the viability of this method for producing both 1,3-dibromoacetone and its chlorinated derivatives with high efficiency.
Q: How does the new process improve yield compared to traditional bromination?
A: Traditional methods are limited by equilibrium to approximately 70% concentration. This patent utilizes reactive crystallization to continuously remove the product from the equilibrium mixture, driving conversion to near completion and achieving concentrations above 95%.
Q: What are the advantages regarding halogen waste management?
A: The process includes a closed-loop system where hydrogen bromide byproducts and residual bromides from the chlorination step are converted back into elemental bromine and recycled, significantly reducing hazardous waste disposal costs.
Q: Can this method be adapted for 1,3-dichloroacetone production?
A: Yes, the isolated 1,3-dibromoacetone serves as a superior precursor. It reacts efficiently with various chloride sources (such as alkali metal chlorides or ammonium salts) to produce 1,3-dichloroacetone with high selectivity, avoiding the isomeric impurities common in direct chlorination.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3-Dibromoacetone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN1956941A are fully realized in practical application. We maintain stringent purity specifications and operate rigorous QC labs equipped to analyze complex halogenated impurity profiles, guaranteeing that every batch of 1,3-dibromoacetone meets the exacting standards required for pharmaceutical and agrochemical synthesis. Our commitment to quality extends beyond the final product to the entire supply chain, where we implement robust traceability and safety protocols.
We invite forward-thinking organizations to collaborate with us to leverage this technology for their specific applications. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your current volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your product competitiveness and supply security.
