Advanced One-Pot Synthesis of Dipyrrolebisanthene for High-Performance Electronic Materials Manufacturing
Advanced One-Pot Synthesis of Dipyrrolebisanthene for High-Performance Electronic Materials Manufacturing
The rapidly evolving landscape of organic optoelectronics demands materials that combine exceptional stability with tunable electronic properties, a challenge that traditional polycyclic aromatic hydrocarbons often fail to meet due to inherent photo-instability. Patent CN114853654A introduces a groundbreaking preparation method for Dipyrrolebisanthene, a novel nitrogen-doped bisanthene derivative that overcomes these limitations through a sophisticated molecular design. This technology represents a significant leap forward for any reliable OLED material supplier seeking to offer next-generation components for organic conductors and magnetic devices. By utilizing a bishalogen substituted bisanthene derivative, specifically BA-2Br, the process achieves a seamless integration of coupling and intramolecular dehydrogenation ring-closing reactions in a single vessel. The resulting molecule not only retains the desirable planar pi-conjugated system of bisanthene but also incorporates pyrrole rings that enhance solubility and stability, making it an ideal candidate for high-purity organic semiconductors used in advanced display technologies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fused-ring aromatic hydrocarbons like bisanthene has been plagued by severe practical drawbacks that hinder their widespread adoption in cost reduction in electronic chemical manufacturing initiatives. Traditional routes often require harsh reaction conditions, multiple discrete synthetic steps, and complex purification protocols to remove stubborn impurities that degrade device performance. Furthermore, the parent bisanthene molecule suffers from poor solubility in common organic solvents and significant photo-instability, which complicates its handling and limits its utility in long-lifespan applications such as solar cells or organic light-emitting diodes. These factors collectively increase the lead time for high-purity organic semiconductors and drive up production costs due to low overall yields and the necessity for expensive recycling of unreacted intermediates. Consequently, manufacturers have struggled to find a scalable pathway that delivers both the electronic performance of graphene fragments and the processability required for industrial coating or printing techniques.
The Novel Approach
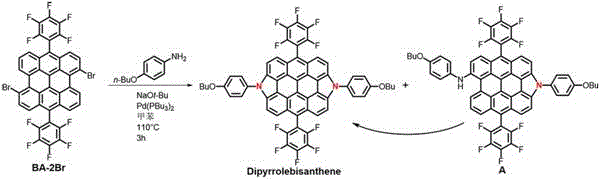
In stark contrast to these legacy challenges, the novel approach detailed in the patent utilizes a strategic one-pot methodology that dramatically simplifies the synthetic workflow while enhancing the final product quality. By reacting BA-2Br with 4-butoxyaniline in the presence of sodium tert-butoxide and a palladium catalyst, the process efficiently constructs the nitrogen-doped framework under relatively mild conditions at 110°C. The most transformative aspect of this innovation is the discovery that the reaction byproduct, a monopyrrole derivative known as Compound A, is not waste but a latent precursor that automatically converts into the target Dipyrrolebisanthene upon exposure to neutral silica gel. This serendipitous transformation eliminates the need for separate cyclization steps or the disposal of valuable intermediate material, thereby streamlining the entire production line. For procurement teams, this translates to a more robust supply chain where raw material utilization is maximized, and the complexity of waste management is substantially reduced without compromising the structural integrity of the final optoelectronic material.
Mechanistic Insights into Pd-Catalyzed Coupling and Dehydrocyclization
The core of this synthetic breakthrough lies in the precise orchestration of palladium-catalyzed C-N bond formation followed by an oxidative dehydrogenation event that closes the pyrrole rings. The reaction initiates with the coordination of the palladium catalyst, specifically bis(tri-tert-butylphosphine)palladium, to the bromine sites on the BA-2Br substrate, facilitating a Buchwald-Hartwig type amination with the 4-butoxyaniline nucleophile. Under the basic conditions provided by sodium tert-butoxide in anhydrous toluene, the system promotes the elimination of hydrogen bromide, driving the equilibrium towards the formation of the C-N bonds necessary for the pyrrole fusion. This mechanistic pathway is critical for R&D directors focused on purity and杂质谱 control, as the use of bulky phosphine ligands on the palladium center helps prevent unwanted side reactions such as homocoupling or over-oxidation, ensuring a cleaner reaction profile. The subsequent intramolecular dehydrogenation is thermodynamically favored at the elevated reaction temperature of 110°C, locking the molecule into its rigid, planar conformation which is essential for efficient charge transport in electronic devices.
Furthermore, the unique behavior of the intermediate Compound A during the purification phase offers a fascinating glimpse into surface-mediated catalysis that can be leveraged for process optimization. While Compound A is initially formed as a partially cyclized species, the acidic sites present on the surface of neutral silica gel act as a mild heterogeneous catalyst that promotes the final ring-closing step to form Dipyrrolebisanthene with an impressive conversion efficiency. This phenomenon suggests that the energy barrier for the final cyclization is low enough to be overcome by the interaction with the stationary phase, effectively turning the purification column into a continuation of the reactor. Understanding this mechanism allows process chemists to fine-tune the silica gel specifications, such as mesh size and pH, to maximize the in-situ conversion rate, thereby achieving higher isolated yields without the need for additional reagents or thermal treatment. This level of control over the reaction trajectory is invaluable for scaling up complex optoelectronic materials where consistency and batch-to-batch reproducibility are paramount.
How to Synthesize Dipyrrolebisanthene Efficiently
Executing this synthesis requires careful attention to atmospheric conditions and reagent stoichiometry to ensure the palladium catalyst remains active throughout the extended heating period. The standard protocol involves charging a reaction vessel with BA-2Br and 4-butoxyaniline in a molar ratio that favors complete consumption of the dibromo precursor, typically using a slight excess of the aniline derivative to drive the double coupling event. The mixture is subjected to freeze-pump-thaw cycles to remove dissolved oxygen, which could otherwise oxidize the phosphine ligands or interfere with the dehydrogenation step, before being heated to 110°C for approximately 3 hours. Following the reaction, the crude mixture is processed through a standard aqueous workup and then loaded directly onto a column packed with neutral silica gel, where the magic of the byproduct conversion takes place over a period of roughly 12 hours.
- Combine BA-2Br, 4-butoxyaniline, Pd(PtBu3)2 catalyst, and NaOtBu base in anhydrous toluene under inert atmosphere.
- Heat the reaction mixture to 110°C and stir for 3 hours to facilitate coupling and initial cyclization.
- Purify via column chromatography, allowing the monopyrrole byproduct to convert to the target product on neutral silica gel.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement managers, the implementation of this patented methodology offers a compelling value proposition centered around operational efficiency and resource optimization. The consolidation of coupling and cyclization into a single pot drastically reduces the number of unit operations required, which directly correlates to lower energy consumption and reduced solvent usage per kilogram of finished product. Moreover, the ability to convert what would traditionally be considered a waste byproduct into saleable inventory significantly improves the atom economy of the process, providing a buffer against fluctuations in raw material pricing. This inherent efficiency supports a strategy of cost reduction in electronic chemical manufacturing by minimizing the need for expensive recycling loops or complex multi-step purifications that often bottleneck production capacity. Additionally, the use of commodity chemicals like toluene and sodium tert-butoxide ensures that the supply chain remains resilient and less susceptible to the volatility associated with specialized reagents.
- Cost Reduction in Manufacturing: The elimination of distinct cyclization reagents and the reduction in processing time lead to substantial cost savings in the overall manufacturing budget. By leveraging the silica gel conversion effect, the process avoids the capital expenditure associated with additional reactors or heating mantles for secondary reaction steps, allowing facilities to produce more batches within the same timeframe. The high selectivity of the palladium catalyst also means that less precious metal is lost to side products, further optimizing the cost structure of the catalyst system which is often a significant portion of the variable costs in fine chemical synthesis.
- Enhanced Supply Chain Reliability: Sourcing strategies benefit from the use of widely available starting materials such as 4-butoxyaniline and standard halogenated aromatics, which reduces the risk of supply disruptions common with exotic intermediates. The robustness of the reaction conditions, which tolerate standard laboratory glassware and do not require extreme pressures or cryogenic temperatures, simplifies the technology transfer to contract manufacturing organizations. This ease of replication ensures that production can be scaled across multiple sites if necessary, providing a safety net for maintaining continuous supply to downstream electronics manufacturers who rely on just-in-time delivery models.
- Scalability and Environmental Compliance: The simplified workflow inherently generates less chemical waste, aligning with increasingly stringent environmental regulations and corporate sustainability goals. The one-pot nature reduces the volume of solvent required for intermediate isolations, lowering the burden on solvent recovery systems and reducing the facility's carbon footprint. Furthermore, the high stability of the final Dipyrrolebisanthene product minimizes degradation during storage and transport, reducing the incidence of rejected shipments and ensuring that the material arrives at the customer's site with the specified purity and performance characteristics intact.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this advanced organic material. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear picture of what partners can expect when adopting this technology. Understanding these details is crucial for making informed decisions about integrating Dipyrrolebisanthene into your product development pipeline.
Q: What is the key advantage of this Dipyrrolebisanthene synthesis method?
A: The primary advantage is the unique ability of the monopyrrole byproduct (Compound A) to automatically convert into the target Dipyrrolebisanthene upon contact with neutral silica gel during purification, significantly boosting overall yield without additional synthetic steps.
Q: How does nitrogen doping affect the properties of Bisanthene?
A: Introducing nitrogen atoms into the bisanthene bay area creates Dipyrrolebisanthene, which exhibits significantly improved material stability, high fluorescence quantum yield, and multiple reversible redox properties compared to the photo-unstable parent bisanthene.
Q: Is this process suitable for large-scale production of organic semiconductors?
A: Yes, the one-pot methodology utilizing standard palladium catalysis and common solvents like toluene, combined with a simplified purification workflow, makes it highly amenable to commercial scale-up for electronic chemical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dipyrrolebisanthene Supplier
As a premier CDMO partner, NINGBO INNO PHARMCHEM possesses the technical expertise and infrastructure to translate this innovative patent into commercial reality for your organization. We understand that moving from milligram-scale laboratory synthesis to industrial production requires more than just a recipe; it demands a deep understanding of process safety, thermodynamics, and quality control. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the delicate balance of the palladium catalysis and silica gel conversion is maintained even at massive volumes. We operate stringent purity specifications and utilize rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of Dipyrrolebisanthene meets the exacting standards required for high-performance electronic applications.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis route to your specific volume and purity requirements. By partnering with us, you gain access to a Customized Cost-Saving Analysis that evaluates the total cost of ownership for this material compared to your current supply options. We encourage you to request specific COA data and route feasibility assessments to verify that our capabilities align with your project timelines. Let us help you secure a stable supply of this cutting-edge material, enabling you to focus on developing the next generation of organic electronic devices with confidence.
