Scalable Synthesis of BODIPY-Naphthalimide Porphyrin Derivatives for Advanced Optoelectronic Applications
Scalable Synthesis of BODIPY-Naphthalimide Porphyrin Derivatives for Advanced Optoelectronic Applications
In the rapidly evolving landscape of organic electronics and biomimetic energy systems, the demand for highly efficient donor-acceptor molecular architectures has never been more critical. Patent CN115304628A introduces a groundbreaking BODIPY-naphthalimide alkynyl long-chain alkoxy porphyrin derivative, designated as Compound (V), which represents a significant leap forward in the design of light-harvesting antennas. This sophisticated molecule integrates three distinct fluorescent moieties—BODIPY, naphthalimide, and porphyrin—into a single conjugated system, enabling multi-pathway efficient energy transfer that mimics natural photosynthesis. For research and development teams focusing on next-generation OLED materials and artificial photosynthetic systems, this technology offers a robust platform for achieving superior photostability and broad-spectrum absorption. The strategic incorporation of long-chain alkoxy groups not only enhances solubility in common organic solvents but also mitigates aggregation issues that often plague large planar aromatic systems during device fabrication.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of multi-chromophore porphyrin derivatives has been fraught with significant challenges that hinder their commercial viability and practical application. Traditional routes often require cumbersome multi-step functionalization procedures where introducing multiple distinct functional groups onto the porphyrin macrocycle leads to complex mixtures of regioisomers that are notoriously difficult to separate. Furthermore, conventional methods frequently suffer from poor solubility of the intermediate species, necessitating the use of exotic solvents or extreme reaction conditions that degrade the delicate porphyrin core. The lack of effective conjugation linkers in older designs often results in substantial steric hindrance between the donor and acceptor units, which severely limits the efficiency of intramolecular energy transfer. Consequently, the resulting materials often exhibit suboptimal quantum yields and narrow absorption bands, failing to meet the rigorous performance standards required for high-efficiency organic light-emitting diodes or advanced solar energy conversion devices.
The Novel Approach
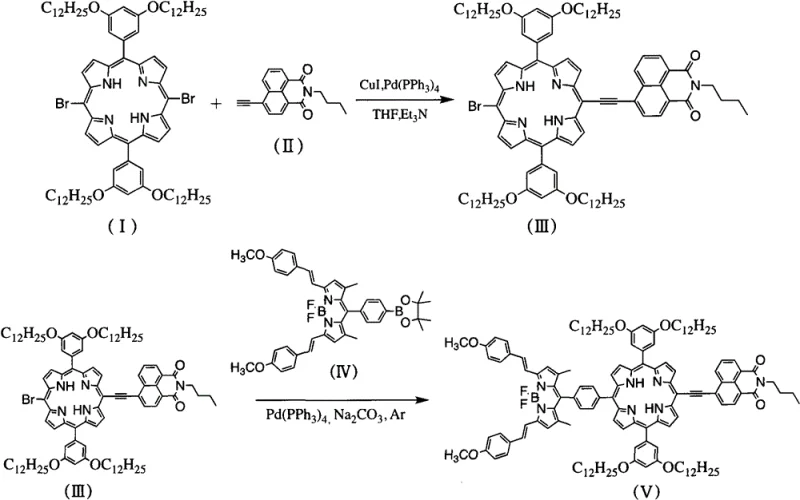
The methodology disclosed in the patent overcomes these historical bottlenecks through a streamlined, two-step catalytic strategy that ensures high regioselectivity and structural integrity. By employing a sequential Sonogashira coupling followed by a Suzuki coupling, the synthesis allows for the precise installation of the naphthalimide and BODIPY units at the 5 and 15 positions of the porphyrin ring. This modular approach drastically simplifies the purification process, as the intermediates are well-defined and can be isolated with high purity using standard silica gel chromatography. The use of an alkynyl linker is particularly ingenious, as it acts as a rigid conjugated bridge that effectively reduces steric repulsion between the bulky fluorophores while maximizing orbital overlap. This structural optimization leads to a significant red-shift in the absorption spectrum and enhances the planarity of the entire molecular system, thereby facilitating the rapid and efficient migration of excitation energy across the molecule.
Mechanistic Insights into Pd-Catalyzed Cross-Coupling Reactions
The core of this synthetic innovation lies in the precise orchestration of palladium-catalyzed cross-coupling mechanisms, which are fundamental to modern organic synthesis yet require meticulous control to achieve the desired complexity. In the first step, the Sonogashira coupling utilizes a dual-catalyst system comprising tetrakis(triphenylphosphine)palladium and cuprous iodide to activate the terminal alkyne of the naphthalimide derivative. The copper co-catalyst forms a copper-acetylide intermediate which then undergoes transmetallation with the oxidative addition complex of the palladium and the dibromo-porphyrin substrate. This mechanism is highly sensitive to oxygen and moisture, necessitating the strict anhydrous and anaerobic conditions described in the patent, typically maintained under an argon atmosphere. The reaction temperature is carefully controlled between 70°C and 80°C to ensure complete conversion of the dibromo precursor to the mono-substituted intermediate without promoting homocoupling side reactions that could compromise the yield.
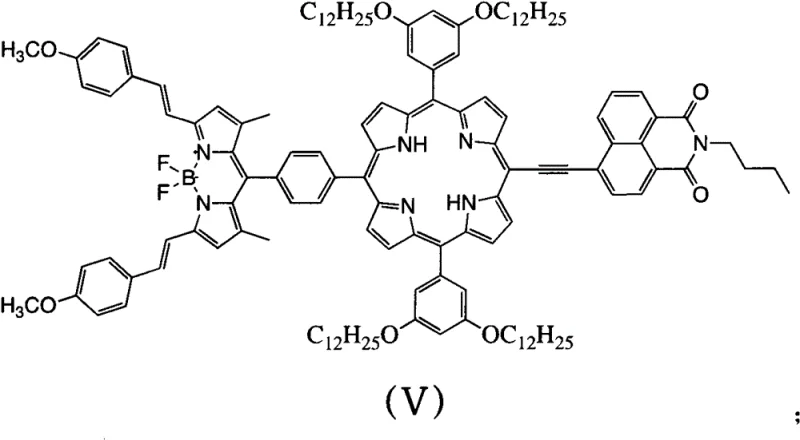
Following the initial coupling, the second stage employs a Suzuki-Miyaura coupling to attach the borate phenyl BODIPY derivative, a reaction renowned for its tolerance of various functional groups and mild conditions. In this phase, the palladium catalyst activates the remaining bromine atom on the porphyrin intermediate, allowing for the transmetallation of the organoboron species in the presence of a base such as sodium carbonate. The choice of a mixed solvent system containing tetrahydrofuran, methanol, and water is critical here, as it ensures the solubility of both the hydrophobic organic substrates and the inorganic base, creating a homogeneous reaction environment that accelerates the catalytic cycle. The resulting molecular architecture, as depicted in the structural analysis, features a highly conjugated pi-system where the electron-rich porphyrin core effectively communicates with the electron-deficient naphthalimide and the highly fluorescent BODIPY unit. This electronic communication is the driving force behind the observed Fluorescence Resonance Energy Transfer (FRET), where energy absorbed by the peripheral groups is funneled efficiently to the BODIPY emitter.
How to Synthesize BODIPY-Naphthalimide Porphyrin Derivative Efficiently
The successful replication of this high-value compound requires strict adherence to the optimized reaction parameters outlined in the patent to ensure consistent quality and yield. The process begins with the preparation of the mono-substituted intermediate under inert gas protection, followed by a rigorous purification step to remove any unreacted starting materials or homocoupled byproducts before proceeding to the final coupling. Detailed operational guidelines regarding stoichiometry, solvent ratios, and workup procedures are essential for maintaining the integrity of the sensitive fluorophores throughout the synthesis. For a comprehensive breakdown of the standardized operating procedures, including specific molar ratios and purification techniques, please refer to the technical guide below.
- Perform a Sonogashira coupling reaction between 5,15-dibromo long-chain alkoxy porphyrin and 6-alkynyl naphthalimide using Pd(PPh3)4 and CuI catalysts at 70-80°C to obtain the mono-substituted intermediate.
- Purify the resulting 5-bromo-15-naphthalimide intermediate via silica gel column chromatography using dichloromethane and petroleum ether eluents.
- Execute a Suzuki coupling reaction between the purified intermediate and borate phenyl BODIPY derivative using Pd(PPh3)4 and sodium carbonate at 65°C to yield the final target compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this synthetic route offers tangible benefits that directly impact the bottom line and operational reliability of chemical manufacturing. The reliance on widely available commodity chemicals such as tetrahydrofuran, dichloromethane, and standard palladium catalysts means that raw material sourcing is straightforward and less susceptible to geopolitical supply disruptions compared to processes requiring exotic reagents. Furthermore, the mild reaction temperatures ranging from 65°C to 80°C significantly reduce energy consumption during the production phase, aligning with modern sustainability goals and lowering utility costs for large-scale manufacturing facilities. The robustness of the two-step sequence also implies a shorter overall production cycle time, allowing for faster turnaround on custom orders and improved responsiveness to market demands for specialized optoelectronic materials.
- Cost Reduction in Manufacturing: The streamlined nature of this two-step synthesis eliminates the need for complex protecting group strategies and extensive purification sequences that are typical of traditional porphyrin functionalization. By utilizing highly selective cross-coupling reactions, the process minimizes the formation of difficult-to-separate impurities, thereby reducing the volume of solvents and silica gel required for purification. This efficiency translates into substantial cost savings in terms of both raw material consumption and waste disposal, making the commercial production of these advanced fluorescent materials economically viable for the first time.
- Enhanced Supply Chain Reliability: The starting materials, including 5,15-dibromo porphyrins and alkynyl naphthalimides, are established intermediates with stable supply chains, reducing the risk of production delays due to raw material shortages. The use of standard catalytic systems like Pd(PPh3)4 ensures that the process can be easily transferred between different manufacturing sites without the need for specialized equipment or unique reactor configurations. This flexibility allows for diversified sourcing strategies and the ability to scale production up or down rapidly in response to fluctuating customer requirements in the OLED and sensor markets.
- Scalability and Environmental Compliance: The reaction conditions are inherently safer and more environmentally friendly than many alternative high-energy synthetic routes, as they avoid the use of strong acids or bases and operate at moderate temperatures. The solvents used are common industrial chemicals with well-established recovery and recycling protocols, facilitating compliance with stringent environmental regulations regarding volatile organic compound emissions. The high selectivity of the coupling reactions also results in a cleaner crude product profile, which simplifies downstream processing and reduces the environmental footprint associated with waste treatment and disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this novel porphyrin derivative. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the material's performance characteristics and manufacturing feasibility. Understanding these details is crucial for stakeholders evaluating the integration of this technology into existing product lines or research pipelines.
Q: What are the key optical advantages of this BODIPY-porphyrin derivative?
A: The compound exhibits a widest absorption wavelength of 477nm and a maximum fluorescence emission at 678nm. It facilitates efficient Fluorescence Resonance Energy Transfer (FRET) from the naphthalimide or porphyrin groups to the BODIPY fluorophore, making it ideal for light-harvesting antenna systems.
Q: How does the synthetic route improve upon conventional porphyrin modifications?
A: Unlike traditional methods that often involve complex, multi-step functionalization with poor solubility, this patent utilizes a modular two-step cross-coupling strategy. This approach significantly reduces steric hindrance through direct alkyne linkage and ensures better product solubility due to the long-chain alkoxy groups.
Q: Is this material suitable for commercial scale-up in organic electronics?
A: Yes, the reaction conditions are mild (65-80°C) and utilize standard palladium catalysts and common organic solvents like THF and dichloromethane. The process avoids extreme temperatures or pressures, facilitating easier transition from laboratory synthesis to industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable BODIPY-Naphthalimide Porphyrin Derivative Supplier
As the global demand for high-performance organic electronic materials continues to surge, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures access to cutting-edge synthesis capabilities and rigorous quality assurance. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of BODIPY-naphthalimide porphyrin derivative meets the exacting standards required for OLED and photonics applications.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthetic route can be adapted to your specific manufacturing needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of adopting this streamlined process for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that will demonstrate our commitment to delivering high-purity electronic chemicals with unmatched reliability and speed.
