Revolutionizing L-Glufosinate Production: Advanced Enzymatic Deracemization for Commercial Scale
The global agrochemical industry is currently witnessing a paradigm shift towards sustainable and high-efficiency manufacturing processes, particularly for critical herbicides like glufosinate-ammonium. Patent CN111321193A introduces a groundbreaking biocatalytic strategy for the asymmetric preparation of L-glufosinate, addressing the long-standing inefficiencies associated with racemic mixtures. This technology leverages a sophisticated multi-enzyme coupling system involving engineered D-amino acid oxidase mutants and transaminases to achieve dynamic kinetic resolution. By transforming the inactive D-isomer directly into the bioactive L-form, this method bypasses the theoretical 50% yield limitation inherent in traditional resolution techniques. The implications for commercial production are profound, offering a pathway to significantly reduce raw material waste and environmental footprint while ensuring superior optical purity.
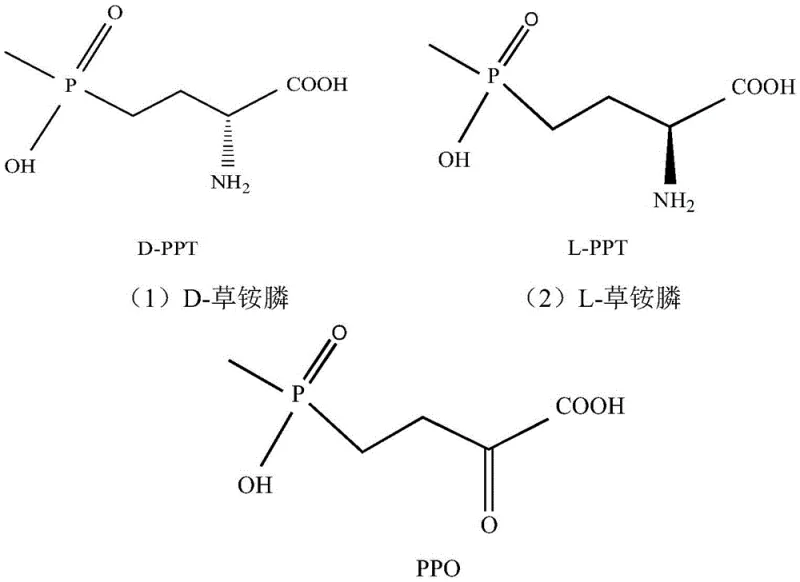
For R&D directors and process chemists, the structural elegance of this approach lies in its ability to utilize readily available racemic D,L-glufosinate as a starting substrate. The core innovation involves a cascade reaction where a specifically mutated D-amino acid oxidase selectively oxidizes D-glufosinate into the keto-acid intermediate, 2-carbonyl-4-[hydroxy(methyl)phosphono]butyric acid (PPO). Crucially, the L-isomer remains untouched during this oxidation phase. Subsequently, a transaminase catalyzes the stereoselective amination of the PPO intermediate back into L-glufosinate. This tandem mechanism effectively creates a thermodynamic sink that drives the equilibrium towards the exclusive formation of the L-enantiomer, demonstrating a level of atomic economy that chemical synthesis struggles to match without complex protecting group strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically pure L-glufosinate has been plagued by significant economic and technical bottlenecks. Traditional chiral resolution methods, which rely on separating enantiomers from a racemic mixture, suffer from an intrinsic maximum theoretical yield of only 50%. This means that half of the synthesized material, the D-isomer, is essentially waste unless subjected to energy-intensive and costly racemization processes for recycling. Furthermore, chemical asymmetric synthesis routes often require expensive chiral auxiliaries, harsh reaction conditions involving extreme temperatures or pressures, and generate substantial amounts of hazardous organic waste. These factors collectively drive up the cost of goods sold (COGS) and complicate regulatory compliance regarding environmental discharge, making conventional methods less attractive for modern, sustainability-focused supply chains.
The Novel Approach
In stark contrast, the biocatalytic method disclosed in the patent offers a streamlined, one-pot solution that elegantly circumvents these limitations. By employing a coupled enzyme system, the process achieves in-situ deracemization, converting the entire racemic feedstock into the desired product with yields approaching 95% or higher under optimized conditions. The use of whole-cell biocatalysts or crude enzyme extracts eliminates the need for expensive cofactor regeneration systems typically required in vitro, as the cellular machinery supports necessary metabolic functions. Moreover, the reaction proceeds under mild physiological conditions (30°C, pH 8.0), drastically reducing energy consumption and safety risks associated with high-pressure chemical reactors. This biological route not only enhances yield but also simplifies downstream processing, as the specificity of enzymes minimizes the formation of side-products and impurities.
Mechanistic Insights into Multi-Enzyme Coupled Deracemization
The heart of this technological advancement lies in the protein engineering of the D-amino acid oxidase (DAAO). The patent details a series of site-directed mutations derived from the wild-type Rhodotorula taiwanensis enzyme, specifically targeting residues such as M213, N54, F58, D207, and S60. These mutations are not random; they are strategically designed to reshape the enzyme's active site pocket to better accommodate the bulky phosphinothricin substrate. For instance, the M213S mutation likely alters the hydrophobicity and steric environment near the catalytic center, enhancing substrate binding affinity. When combined with additional mutations like N54V and F58E, the enzyme exhibits a synergistic increase in catalytic efficiency (kcat/Km), allowing for rapid turnover of the D-isomer even at high substrate concentrations. This engineered robustness is critical for industrial applications where substrate loading must be maximized to ensure economic viability.
Furthermore, the integration of catalase into the reaction system addresses a common pitfall of oxidase-catalyzed reactions: the accumulation of hydrogen peroxide. As the DAAO oxidizes the D-amino acid, it generates hydrogen peroxide as a byproduct, which can irreversibly inactivate sensitive enzymes like transaminases through oxidative damage. The inclusion of catalase ensures the immediate decomposition of hydrogen peroxide into water and oxygen, thereby protecting the catalytic integrity of the entire system. This protective measure extends the operational lifetime of the biocatalysts, reducing the frequency of enzyme replenishment and ensuring consistent reaction kinetics throughout the batch cycle. The result is a highly stable and reliable process capable of maintaining high enantiomeric excess (ee > 99%) throughout the conversion.
How to Synthesize L-Glufosinate Efficiently
Implementing this biocatalytic route requires precise control over fermentation parameters and reaction conditions to maximize the expression of the engineered enzymes. The process begins with the cultivation of recombinant E. coli strains harboring the plasmids for both the mutant DAAO and the transaminase. Induction with IPTG at optimal cell density ensures high intracellular enzyme titers. Following cell harvest and lysis, the crude enzyme preparation is introduced into a buffered reaction vessel containing the racemic substrate. The synergy between the oxidation and amination steps must be balanced by adjusting the ratio of the two biocatalysts to prevent the accumulation of the keto-acid intermediate. Detailed standard operating procedures for strain construction, fermentation, and biotransformation are essential for technology transfer.
- Construct genetically engineered E. coli strains expressing specific D-amino acid oxidase mutants (e.g., M213S-N54V-F58E-D207A-S60T) and transaminase.
- Prepare a one-pot reaction system containing racemic D,L-glufosinate, engineered bacterial cells, catalase, PLP cofactor, and an amino donor.
- Maintain reaction at 30°C and pH 8.0 for 10 hours, followed by ion exchange purification and crystallization to isolate high-purity L-glufosinate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this enzymatic technology represents a strategic opportunity to optimize cost structures and secure supply continuity. The elimination of expensive chiral resolving agents and the drastic reduction in solvent usage directly translate to lower variable costs per kilogram of finished product. Additionally, the reliance on fermentation-derived biocatalysts decouples production from the volatility of petrochemical feedstock prices, offering a more stable cost baseline. The high specificity of the enzymatic process also reduces the burden on purification units, lowering utility costs associated with distillation and chromatography. These factors combine to create a compelling economic case for adopting this green chemistry approach in large-scale manufacturing facilities.
- Cost Reduction in Manufacturing: The most significant financial benefit arises from the near-quantitative yield of the deracemization process. Unlike resolution methods that discard half the material, this method utilizes the entire racemic input, effectively doubling the output from the same amount of starting material without proportional increases in raw material costs. Furthermore, the mild reaction conditions eliminate the need for specialized high-pressure equipment and extensive safety infrastructure, leading to substantial capital expenditure savings. The reduction in waste disposal costs, driven by the aqueous nature of the biocatalytic medium, further enhances the overall profit margin.
- Enhanced Supply Chain Reliability: Biological manufacturing offers distinct advantages in terms of supply security. The genetic sequences for the enzymes can be stored indefinitely and revived rapidly, ensuring that production capacity can be scaled up quickly in response to market demand spikes. Unlike chemical synthesis which may rely on scarce metal catalysts or complex intermediates subject to geopolitical supply risks, the raw materials for this fermentation process (sugars, nitrogen sources) are globally abundant and commoditized. This resilience makes the supply chain less vulnerable to external disruptions, ensuring consistent delivery schedules for downstream formulators.
- Scalability and Environmental Compliance: Scaling biocatalytic processes is generally more straightforward than scaling complex chemical syntheses involving hazardous reagents. The reaction operates at ambient pressure and moderate temperatures, simplifying reactor design and heat management. From an environmental perspective, the process aligns perfectly with increasingly stringent global regulations on volatile organic compounds (VOCs) and heavy metal residues. The biodegradable nature of the enzyme catalysts and the aqueous waste stream simplifies wastewater treatment, reducing the risk of regulatory fines and enhancing the corporate sustainability profile of the manufacturer.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this patent is crucial for stakeholders evaluating its implementation potential. Common inquiries often revolve around the stability of the enzymes, the purity of the final product, and the feasibility of retrofitting existing facilities. The following answers address these key concerns based on the data provided in the patent documentation, offering clarity on the operational realities of this advanced biocatalytic platform.
Q: What is the primary advantage of this enzymatic method over traditional chemical resolution?
A: Unlike traditional resolution which theoretically caps yield at 50% and requires expensive chiral reagents, this multi-enzyme coupling method achieves near-theoretical yields (up to 95%) by recycling the unwanted D-isomer into the desired L-product.
Q: How does the D-amino acid oxidase mutant improve process efficiency?
A: The specific quintuple mutation (M213S-N54V-F58E-D207A-S60T) significantly enhances catalytic activity compared to the wild-type enzyme, allowing for faster conversion rates and reduced enzyme loading requirements.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process utilizes robust E. coli expression systems and mild reaction conditions (30°C, atmospheric pressure), making it highly scalable and environmentally friendly compared to harsh chemical synthesis routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Glufosinate Supplier
As the agrochemical sector demands higher purity and greener manufacturing standards, NINGBO INNO PHARMCHEM stands ready to facilitate the commercialization of this cutting-edge technology. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory successes are seamlessly translated into industrial reality. We understand the critical importance of stringent purity specifications and rigorous QC labs in maintaining product consistency for global registration. Our state-of-the-art facilities are equipped to handle complex biocatalytic processes, offering a secure and compliant environment for the manufacture of high-value intermediates like L-glufosinate.
We invite potential partners to engage with our technical procurement team to discuss how this enzymatic route can be tailored to your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential ROI of switching to this biocatalytic method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your procurement strategy is built on the foundation of the most advanced and efficient chemical technologies available.
