Scalable Synthesis of Triphenylene-Pyrene Discotic Liquid Crystals for Next-Gen Optoelectronics
Scalable Synthesis of Triphenylene-Pyrene Discotic Liquid Crystals for Next-Gen Optoelectronics
The rapidly evolving landscape of organic electronics demands materials that seamlessly integrate high charge mobility with robust luminescent properties. A groundbreaking development in this sector is detailed in patent CN112979434A, which discloses a sophisticated preparation method for a triphenylene-pyrene-triphenylene discotic liquid crystal triad compound. This specific class of materials is engineered to self-assemble into highly ordered hexagonal columnar and nematic phases, making them ideal candidates for molecular electronics and photovoltaic applications. The core innovation lies in the strategic fusion of a rigid, planar aromatic core with flexible aliphatic side chains, creating a unique microenvironment that facilitates one-dimensional charge carrier transport. As a reliable electronic chemical supplier, understanding the nuances of such complex molecular architectures is critical for delivering high-purity OLED material to the global market.
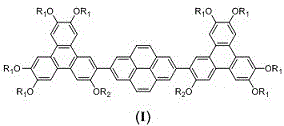
The structural integrity of these compounds is paramount for their function as organic semiconductors. The general formula (I) presented in the patent highlights a symmetric architecture where a central pyrene unit is flanked by two triphenylene moieties, each decorated with multiple alkoxy chains. These alkoxy chains, ranging from C6 to C10 alkyl groups, are not merely passive spectators; they play an active role in regulating the inter-columnar distance and ensuring the fluidity of the liquid crystalline phase. The ability of these molecules to form stable mesophases over a wide temperature range is a direct result of the delicate balance between the pi-pi stacking interactions of the aromatic cores and the steric bulk of the peripheral chains. For R&D directors focusing on purity and杂质谱 (impurity profiles), the precise control over these alkyl chain lengths is essential to tune the phase transition temperatures and optimize device performance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of large pi-conjugated systems containing both liquid crystalline and luminescent units has been fraught with challenges. Traditional routes often rely on harsh halogenation conditions to activate the aromatic rings for coupling, which can lead to unwanted side reactions and difficult-to-remove halogenated impurities. Furthermore, achieving the specific 2,7-substitution pattern on the pyrene core typically requires multi-step protection and deprotection strategies that drastically reduce overall yield and increase waste generation. The formation of excimers in pyrene derivatives, while useful for sensing, can be detrimental to the desired charge transport properties if not carefully controlled through precise molecular design. Conventional methods often struggle to maintain the structural rigidity required for effective columnar stacking while introducing the necessary solubility via alkoxy chains, resulting in materials with poor processability or unstable liquid crystal phases.
The Novel Approach
The methodology outlined in the patent offers a transformative solution by utilizing a modular Suzuki-Miyaura coupling strategy. Instead of relying on difficult halogenation, the process employs perfluorobutanesulfonyl fluoride to activate the triphenylene hydroxyl groups under remarkably mild conditions. This activation step, conducted at -50°C, generates a highly reactive sulfonate intermediate that serves as an excellent electrophile for the subsequent cross-coupling. This approach significantly simplifies the synthetic route, reducing the number of steps and avoiding the use of toxic or corrosive halogenating agents. By decoupling the synthesis of the triphenylene arms and the pyrene core, chemists can independently optimize the substitution patterns on each fragment before assembling the final triad. This modularity allows for cost reduction in display material manufacturing by enabling the use of diverse alkyl chain precursors without redesigning the entire synthetic pathway.
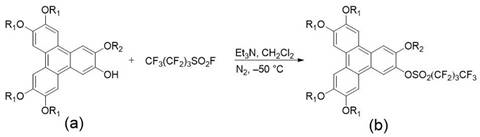
Mechanistic Insights into Suzuki-Miyaura Cross-Coupling
The heart of this synthesis lies in the palladium-catalyzed cross-coupling reaction that joins the triphenylene and pyrene fragments. The mechanism initiates with the oxidative addition of the zero-valent palladium catalyst, Pd(PPh3)4, into the carbon-oxygen bond of the triphenylene perfluorobutyl sulfonate. Although carbon-oxygen bonds are traditionally stronger and less reactive than carbon-halogen bonds, the electron-withdrawing nature of the perfluorobutyl sulfonyl group significantly weakens this bond, facilitating oxidative addition. Following this, the transmetallation step occurs, where the boronate species derived from the pyrene core transfers its aryl group to the palladium center. This step is facilitated by the presence of a base, typically potassium carbonate (K2CO3), which activates the boron atom by forming a more nucleophilic boronate complex. The final reductive elimination releases the coupled product and regenerates the active palladium catalyst, completing the cycle.
Impurity control in this reaction is achieved through the careful selection of ligands and reaction conditions. The use of triphenylphosphine ligands stabilizes the palladium species, preventing the formation of palladium black and minimizing homocoupling side reactions where two pyrene or two triphenylene units might couple together. The reaction is performed in a mixed solvent system of THF and water at 70°C, which ensures adequate solubility of both the organic substrates and the inorganic base. This biphasic condition helps to sequester inorganic salts and byproducts, simplifying the downstream purification process. For procurement managers, this high level of selectivity translates to reduced raw material consumption and lower costs associated with chromatographic purification, as the crude product contains fewer structurally similar impurities that are hard to separate.
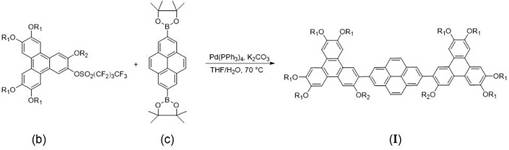
How to Synthesize Triphenylene-Pyrene Triad Efficiently
The synthesis of this advanced discotic liquid crystal involves a streamlined three-step sequence that balances reactivity with operational simplicity. The process begins with the activation of the triphenylene precursor, followed by the independent preparation of the pyrene core via iridium-catalyzed borylation. The final assembly brings these two key intermediates together through a robust palladium-catalyzed coupling. Detailed standard operating procedures for each stage, including precise molar ratios and temperature controls, are essential for reproducibility. The following guide outlines the critical parameters extracted from the patent data to ensure successful execution of this complex transformation.
- Activate the triphenylene hydroxyl group by reacting with perfluorobutanesulfonyl fluoride at -50°C to form the sulfonate intermediate.
- Perform iridium-catalyzed C-H borylation on pyrene using bis-pinacolato diboron to generate the 2,7-bis(boronate) pyrene core.
- Execute the final Suzuki-Miyaura cross-coupling between the triphenylene sulfonate and pyrene boronate using Pd(PPh3)4 and K2CO3 at 70°C.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this synthetic route offers substantial strategic benefits beyond mere technical feasibility. The reliance on commercially available starting materials, such as pyrene and monohydroxytriphenylene derivatives, mitigates the risk of raw material shortages that often plague the specialty chemical sector. The reaction conditions are notably mild, with the most energy-intensive step occurring at a moderate 70°C, which significantly lowers the thermal load on manufacturing infrastructure. This energy efficiency, combined with the use of standard solvents like dichloromethane and cyclohexane, ensures that the process can be easily integrated into existing multipurpose reactors without requiring exotic high-pressure or cryogenic equipment. Such operational flexibility is crucial for maintaining supply continuity in a volatile global market.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous halogenating reagents in favor of perfluorobutanesulfonyl fluoride represents a significant shift in cost structure. While the activator itself is a specialized reagent, its high efficiency and the resulting high yields (reported up to 75% for intermediates) offset the initial material cost. Furthermore, the simplified purification workflow, which relies on standard silica gel chromatography and recrystallization rather than preparative HPLC, drastically reduces processing time and solvent consumption. This streamlined downstream processing directly contributes to a lower cost of goods sold (COGS), making high-performance organic semiconductors more accessible for mass-market applications.
- Enhanced Supply Chain Reliability: The modular nature of the synthesis allows for the stocking of key intermediates, such as the boronated pyrene and the activated triphenylene sulfonate. This decoupling means that production of the final API or material can continue even if there are temporary disruptions in the supply of one specific precursor. Additionally, the robustness of the Suzuki coupling reaction tolerates minor variations in reagent quality, reducing the rejection rate of batches due to raw material specification deviations. This resilience is vital for long-term contracts with major display manufacturers who require guaranteed delivery schedules and consistent quality.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram quantities is facilitated by the homogeneous nature of the catalytic systems and the absence of gas-evolving steps that complicate reactor design. The use of nitrogen protection is standard practice in fine chemical manufacturing and poses no significant barrier to scale-up. From an environmental standpoint, the avoidance of heavy metal halides and the potential for solvent recovery in the extraction steps align with increasingly stringent green chemistry regulations. The ability to recycle the aqueous phase containing the inorganic base further minimizes waste generation, supporting corporate sustainability goals and reducing disposal costs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these discotic liquid crystal materials. The answers are derived directly from the experimental data and theoretical framework provided in the patent documentation, ensuring accuracy and relevance for industry stakeholders. Understanding these details is key to evaluating the feasibility of integrating this technology into your current product development pipeline.
Q: What are the key advantages of the triphenylene-pyrene triad structure?
A: The combination of the rigid triphenylene liquid crystal unit and the luminescent pyrene unit creates a large pi-conjugated system. This structure self-assembles into stable hexagonal columnar and nematic phases over a wide temperature range, enhancing charge carrier mobility and fluorescence properties for organic semiconductor applications.
Q: Why is perfluorobutanesulfonyl fluoride used in the synthesis?
A: Perfluorobutanesulfonyl fluoride acts as a highly efficient activating agent for the phenolic hydroxyl group on the triphenylene ring. It converts the hydroxyl group into a superior leaving group (perfluorobutyl sulfonate) under mild conditions (-50°C to room temperature), facilitating the subsequent palladium-catalyzed cross-coupling reaction with higher yields compared to traditional halogenation methods.
Q: Is this synthesis method suitable for large-scale production?
A: Yes, the patent describes a route with simple operations, mild reaction conditions (70°C for the final step), and commercially available catalysts like Pd(PPh3)4. The use of standard solvents like THF and dichloromethane, along with straightforward purification via silica gel chromatography and recrystallization, indicates strong potential for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triphenylene-Pyrene Triad Supplier
The synthesis of complex discotic liquid crystals like the triphenylene-pyrene triad requires a partner with deep expertise in organometallic catalysis and process optimization. NINGBO INNO PHARMCHEM stands at the forefront of this field, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the sensitive air- and moisture-free conditions required for iridium and palladium catalysis, ensuring that every batch meets stringent purity specifications. With rigorous QC labs employing advanced analytical techniques, we guarantee the structural integrity and phase behavior of our electronic chemicals, providing you with a reliable foundation for your next-generation optoelectronic devices.
We invite you to collaborate with us to unlock the full potential of this patented technology. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a supply chain that prioritizes quality, consistency, and innovation. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you accelerate your path to market with high-performance organic semiconductor materials.
