Advanced Copper-Catalyzed Synthesis of N-Phenylcarbazole for Commercial OLED Production
Advanced Copper-Catalyzed Synthesis of N-Phenylcarbazole for Commercial OLED Production
The rapid expansion of the organic electronics sector has placed immense pressure on supply chains to deliver high-performance intermediates with both economic efficiency and exceptional purity. Patent CN111777543B introduces a transformative methodology for the preparation of N-phenylcarbazole, a critical building block for organic photoelectric materials, utilizing a ligand-free copper-catalyzed C-N coupling strategy. This innovation addresses the longstanding dichotomy in the industry between costly mild processes and harsh, energy-intensive high-yield routes. By leveraging a specifically engineered mixed solvent system and optimizing the copper catalyst loading, the disclosed method achieves reaction yields exceeding 90% under significantly milder thermal conditions than previously possible. For R&D directors and procurement strategists alike, this patent represents a pivotal shift towards sustainable and cost-effective manufacturing of OLED precursors, eliminating the need for expensive ligands while maintaining rigorous quality standards essential for downstream device performance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of N-phenylcarbazole has been constrained by significant economic and operational bottlenecks inherent in traditional Ullmann-type coupling reactions. One prevalent route utilizes iodobenzene as the electrophilic partner; while this method proceeds under relatively mild conditions, the prohibitive cost of iodobenzene compared to other halobenzenes severely impacts the overall production economics, rendering it less viable for large-scale commodity chemical manufacturing. Alternatively, processes employing the more economical bromobenzene have typically demanded extreme reaction conditions to drive the coupling to completion. As illustrated in the prior art reaction scheme below, these legacy methods often require temperatures soaring to 240°C to 250°C and necessitate the addition of complex organic ligands such as 18-crown ether-6 to facilitate the catalytic cycle.
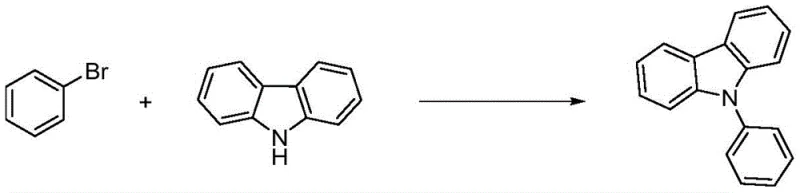
The reliance on such harsh thermal environments not only escalates energy consumption and safety risks but also complicates the purification landscape. The presence of auxiliary ligands introduces additional impurities that are notoriously difficult to separate from the final product, often requiring extensive chromatographic or recrystallization steps that erode overall yield. Furthermore, the use of high-boiling polar aprotic solvents like DMPU in these high-temperature regimes poses challenges for solvent recovery and waste management, creating a substantial environmental footprint that modern green chemistry initiatives strive to eliminate. Consequently, there has been a persistent demand for a process that combines the low raw material cost of bromobenzene with the operational simplicity and mildness previously associated only with more expensive iodine-based routes.
The Novel Approach
The methodology detailed in patent CN111777543B fundamentally reengineers the reaction landscape by introducing a robust ligand-free copper catalytic system operating within a tailored mixed solvent environment. This novel approach successfully activates bromobenzene for C-N coupling with carbazole at temperatures ranging strictly from 110°C to 150°C, a drastic reduction from the >240°C required by previous state-of-the-art methods. The elimination of expensive and separation-challenging ligands is a key differentiator, streamlining the downstream processing workflow and significantly enhancing the purity profile of the crude product. The reaction scheme depicted below highlights the simplicity of this transformation, where bromobenzene and carbazole converge under the influence of a copper salt and base to form the target N-phenylcarbazole efficiently.
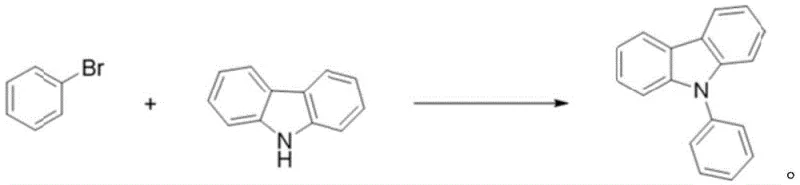
Central to this breakthrough is the strategic utilization of a mixed solvent system comprising a solvent A (such as ethylene glycol or glycerol) and a solvent B (such as N-methylpyrrolidone or DMF). This binary solvent architecture plays a dual role: it ensures the adequate solubility of the inorganic base and organic substrates while potentially stabilizing the active copper species in the absence of external ligands. The result is a process that delivers yields of greater than or equal to 90% and product content exceeding 99%, matching or surpassing the performance of costly alternatives while utilizing the abundant and inexpensive bromobenzene feedstock. This combination of mild operating parameters, ligand-free conditions, and high efficiency positions the technology as a superior choice for the commercial scale-up of complex organic intermediates destined for high-value electronic applications.
Mechanistic Insights into Ligand-Free Copper-Catalyzed C-N Coupling
The success of this ligand-free protocol hinges on a nuanced understanding of the copper catalytic cycle and the specific solvation effects provided by the mixed solvent system. In traditional Ullmann couplings, bidentate ligands are often employed to stabilize the Cu(I) species and facilitate the oxidative addition of the aryl halide. However, in this optimized system, the hydroxyl-rich components of the solvent mixture, such as ethylene glycol or glycerol, likely act as weak coordinating agents that sufficiently stabilize the copper center without forming overly stable complexes that would inhibit turnover. The reaction initiates with the formation of a copper-amide species through the deprotonation of carbazole by the inorganic base, followed by the oxidative addition of bromobenzene to the copper center. The subsequent reductive elimination step releases the N-phenylcarbazole product and regenerates the active copper catalyst, completing the cycle with high fidelity.
From an impurity control perspective, the absence of exogenous ligands is paramount for achieving the high purity levels required for OLED materials. In ligand-mediated processes, residual ligand or ligand-metal complexes can persist through standard workups, acting as deep-level traps in electronic devices that degrade efficiency and lifespan. By removing this variable entirely, the patented process inherently reduces the complexity of the impurity spectrum. Furthermore, the moderate reaction temperature of 110°C to 150°C minimizes thermal degradation pathways and side reactions such as homocoupling of the aryl halide or over-arylation of the carbazole nitrogen, which are more prevalent at the extreme temperatures of 250°C seen in prior art. This precise control over the reaction environment ensures that the resulting high-purity N-phenylcarbazole meets the stringent specifications necessary for integration into sensitive optoelectronic architectures without requiring burdensome purification protocols.
How to Synthesize N-Phenylcarbazole Efficiently
Implementing this synthesis route in a pilot or production setting requires careful attention to the stoichiometry of the copper catalyst and the composition of the mixed solvent system to maximize yield and minimize cycle time. The patent outlines a versatile range of copper salts, including cuprous chloride, cuprous iodide, and cupric acetate, allowing manufacturers to select the most cost-effective variant based on local supply chain availability. The process is designed to be operationally simple, involving a single-pot reaction under nitrogen protection followed by a straightforward aqueous workup and crystallization. Detailed below are the standardized operational parameters derived from the patent examples, which serve as a robust starting point for process engineers aiming to establish a reliable OLED material supplier pipeline.
- Charge bromobenzene and carbazole into a reactor with a copper catalyst, inorganic base, and a mixed solvent system under nitrogen protection.
- Heat the reaction mixture to a temperature range of 110-150°C and maintain stirring for 6 to 24 hours to ensure complete conversion.
- Perform aqueous workup followed by extraction with an organic solvent, desolventization, and recrystallization to isolate high-purity N-phenylcarbazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this ligand-free copper-catalyzed process offers tangible strategic benefits that extend beyond simple raw material substitution. The primary value driver is the substantial reduction in manufacturing costs achieved by replacing expensive iodobenzene with commodity-grade bromobenzene and eliminating the need for specialized ligands like 18-crown ether-6. This shift not only lowers the direct bill of materials but also simplifies the supply chain by reducing dependency on niche chemical suppliers. Additionally, the milder reaction conditions translate directly into lower energy utility costs and reduced wear on reactor equipment, contributing to a more sustainable and economically resilient production model that aligns with modern corporate sustainability goals.
- Cost Reduction in Manufacturing: The economic argument for this process is compelling, driven principally by the substitution of high-cost inputs with widely available commodities. By utilizing bromobenzene instead of iodobenzene, manufacturers can access a raw material market that is significantly larger and more price-stable, insulating production costs from the volatility often seen in the iodine market. Furthermore, the complete removal of organic ligands eliminates a costly line item from the batch sheet and removes the associated expenses of ligand recovery or disposal. The simplified downstream processing, necessitated by the cleaner reaction profile, reduces solvent usage and labor hours during purification, cumulatively driving down the cost per kilogram of the final API intermediate or electronic material.
- Enhanced Supply Chain Reliability: Supply chain continuity is critically dependent on the availability of key reagents and the robustness of the manufacturing process. Bromobenzene is a bulk chemical produced globally in massive quantities, ensuring that supply disruptions are highly unlikely compared to specialized ligands or iodobenzene. The robustness of the catalytic system, which tolerates a variety of copper salts and bases, provides procurement teams with the flexibility to switch suppliers or grades of raw materials without requiring extensive re-validation of the process. This flexibility is a crucial risk mitigation strategy, ensuring that production schedules for high-purity OLED precursors remain uninterrupted even in the face of regional supply fluctuations or logistical challenges.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to multi-ton production often reveals hidden complexities, particularly regarding heat management and waste treatment. The moderate temperature range of 110°C to 150°C allows for the use of standard heating media such as steam or thermal oil, avoiding the need for specialized high-temperature equipment required for 250°C operations. From an environmental standpoint, the ligand-free nature of the reaction simplifies wastewater treatment and reduces the load of organic contaminants in the effluent. The ability to achieve high yields with minimal byproducts supports cost reduction in organic photoelectric material manufacturing by lowering waste disposal fees and ensuring compliance with increasingly strict environmental regulations governing chemical production facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copper-catalyzed synthesis route. These insights are derived directly from the experimental data and comparative analysis presented in patent CN111777543B, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this method for their specific production requirements and quality targets.
Q: Why is the copper-catalyzed bromobenzene route preferred over iodobenzene methods?
A: While iodobenzene routes offer mild conditions, the raw material cost is significantly higher. The patented bromobenzene route achieves comparable yields (>90%) and purity (>99%) at a fraction of the raw material cost, making it economically superior for large-scale production.
Q: How does this method improve upon previous high-temperature bromobenzene processes?
A: Previous methods required extreme temperatures (240-250°C) and expensive ligands like 18-crown ether-6. This novel approach operates at mild temperatures (110-150°C) without ligands, drastically simplifying purification and reducing energy consumption.
Q: What purity levels can be achieved with this synthesis method?
A: The process consistently delivers N-phenylcarbazole with a content of greater than or equal to 99%, meeting the stringent specifications required for organic photoelectric applications such as OLEDs and OFETs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Phenylcarbazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic methodologies requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the ligand-free copper-catalyzed process are fully realized in a commercial setting. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of N-phenylcarbazole meets the exacting standards required for organic light-emitting diodes and other high-performance electronic applications. Our commitment to quality assurance ensures that the impurity profiles are tightly controlled, safeguarding the performance of your final devices.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your supply chain and reduce overall manufacturing expenditures. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic impact of switching to this bromobenzene-based protocol. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your volume requirements, allowing us to demonstrate our capability as a trusted provider of high-value electronic chemical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
