Advanced Phthalocyanine Synthesis: A Strategic Breakthrough for Functional Material Manufacturing
Advanced Phthalocyanine Synthesis: A Strategic Breakthrough for Functional Material Manufacturing
The chemical industry is constantly seeking more efficient pathways to synthesize high-performance functional materials, and Patent CN100378102C presents a transformative approach to producing phthalocyanine compounds. This intellectual property outlines a sophisticated multi-step synthesis that circumvents the traditional reliance on nitrophthalonitrile intermediates, which are often associated with complex processing requirements and safety concerns. By utilizing a novel sequence involving nucleophilic substitution of phthalimides followed by controlled oxidation and cyclization, this technology enables the precise installation of solubilizing groups. This capability is critical for manufacturers of organic photoconductive materials, optical recording media, and advanced pigments who require consistent quality and tailored physicochemical properties. As a reliable phthalocyanine intermediates supplier, understanding these mechanistic nuances is essential for evaluating the scalability and commercial viability of next-generation functional dyes.
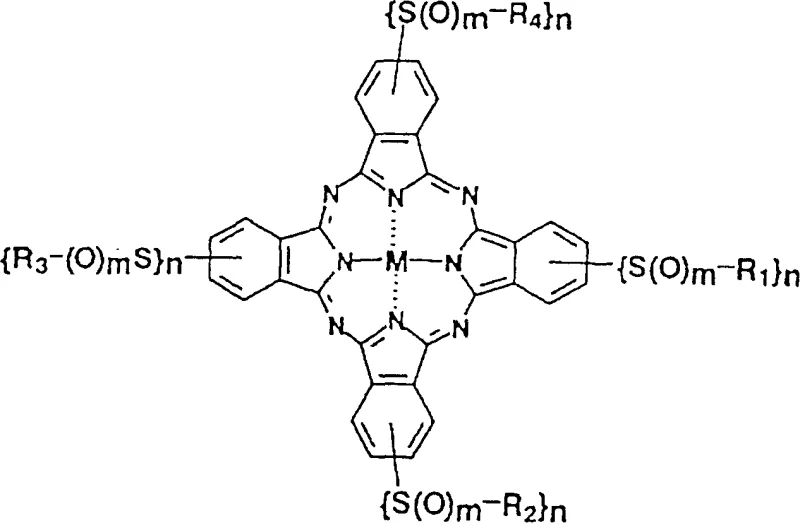
The core innovation lies in the structural flexibility offered by General Formula (I), where substituents R1 through R4 can be independently modified to include alkyl, aryl, or heterocyclic groups bearing ionic hydrophilic functionalities. This modularity allows for the fine-tuning of oxidation potentials and solubility profiles without compromising the stability of the macrocyclic core. The patent specifically highlights the utility of lithium salts of sulfo groups to enhance water solubility, a feature highly desirable for inkjet printing applications and aqueous-based coating formulations. Furthermore, the ability to control the number and position of these substituents ensures a narrow impurity profile, addressing a key pain point for R&D directors focused on color reproducibility and batch-to-batch consistency in high-end electronic materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of substituted phthalocyanines has heavily depended on the use of 4-nitrophthalonitrile or 3-nitrophthalonitrile as key starting materials. While effective for laboratory-scale synthesis, these nitro-based routes present significant hurdles when scaled to commercial production levels. The handling of nitro compounds often necessitates specialized equipment to manage exothermic risks and potential toxicity, leading to increased capital expenditure and operational complexity. Moreover, the introduction of solubilizing groups via nitro intermediates can result in mixed isomer distributions, complicating downstream purification and reducing overall yield. For procurement managers, these inefficiencies translate into higher raw material costs and longer lead times, as the supply chain for high-purity nitrophthalonitriles can be volatile and subject to regulatory scrutiny regarding environmental impact and waste disposal.
The Novel Approach
In stark contrast, the methodology disclosed in CN100378102C leverages a thiol-substitution strategy on phthalimide precursors, effectively bypassing the need for hazardous nitro intermediates. This route begins with the reaction of a mercapto compound with a substituted phthalimide, creating a stable thioether linkage that serves as a robust anchor for subsequent functionalization. The process allows for the introduction of diverse side chains, including those with ionic hydrophilic groups, at an early stage of the synthesis. This early-stage modification ensures that the solubility characteristics are built into the molecular framework before the formation of the rigid phthalocyanine ring. For supply chain heads, this represents a substantial opportunity for cost reduction in functional dye manufacturing, as it utilizes more abundant and safer starting materials while simplifying the overall process flow and minimizing the generation of hazardous byproducts.
Mechanistic Insights into Thiol-Substitution and Oxidative Cyclization
The reaction mechanism proceeds through a meticulously designed five-step sequence that maximizes atom economy and minimizes side reactions. Initially, a nucleophilic substitution occurs between a metal salt of a mercapto compound (General Formula II) and a leaving-group-substituted phthalimide (General Formula III), typically in a polar aprotic solvent like DMF or DMSO. This step is crucial for establishing the carbon-sulfur bond that will eventually become the solubilizing tail. Following this, an oxidation step converts the sulfide moiety into a sulfoxide or sulfone using oxidants such as hydrogen peroxide, often catalyzed by tungstate salts. This oxidation not only stabilizes the sulfur linkage against further unwanted reactions but also modifies the electronic nature of the substituent, potentially enhancing the electron-withdrawing character of the group attached to the aromatic ring.
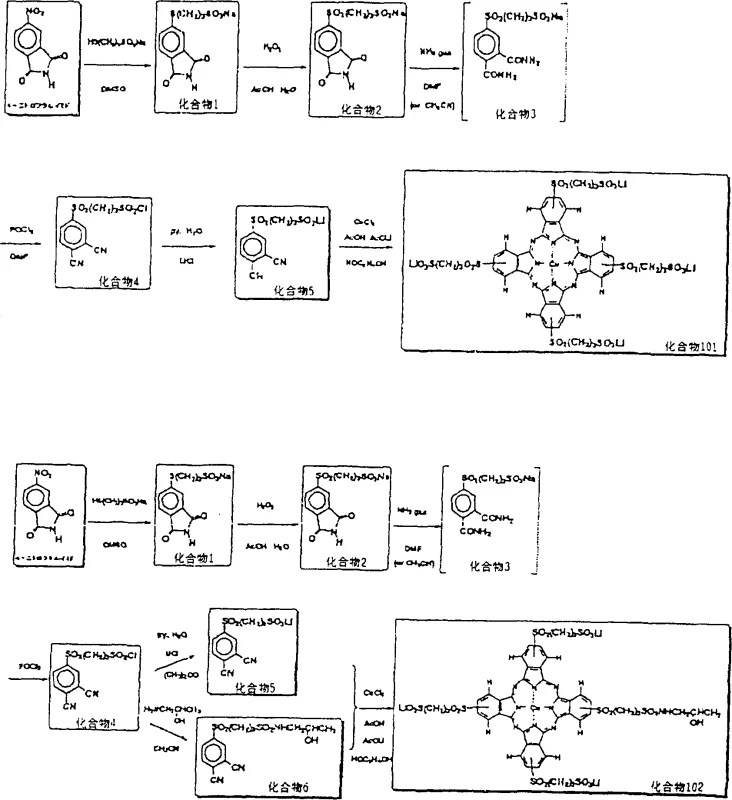
Subsequent steps involve the conversion of the phthalimide to a phthalamide via ammonolysis, followed by dehydration to form the critical phthalonitrile intermediate. This nitrile species then undergoes cyclotetramerization in the presence of a metal derivative, such as copper chloride, at elevated temperatures. The use of buffer systems containing organic acids with specific pKa values during the cyclization step is a notable refinement, as it suppresses the decomposition of the forming phthalocyanine macrocycle. This level of control over the reaction environment is vital for achieving high purity, as it prevents the formation of metal-free phthalocyanines or partially metallated species that could act as impurities. For technical teams, understanding this buffer effect is key to optimizing reaction conditions for maximum yield and minimal downstream purification burden.
How to Synthesize Substituted Phthalocyanine Efficiently
The synthesis protocol detailed in the patent provides a robust framework for producing high-quality phthalocyanine derivatives suitable for demanding applications. The process emphasizes the importance of solvent selection, temperature control, and stoichiometric precision at each stage to ensure the integrity of the functional groups. Detailed standardized synthesis steps are provided in the guide below to assist process engineers in replicating these results.
- React substituted phthalimide with mercapto compounds to form thioether intermediates.
- Oxidize the sulfide group to sulfone or sulfoxide using hydrogen peroxide.
- Convert imide to diamide via ammonolysis, followed by dehydration to phthalonitrile.
- Perform cyclotetramerization with metal salts in high-boiling solvents to yield the final phthalocyanine.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers distinct advantages that align with the strategic goals of cost efficiency and supply chain resilience. By eliminating the dependency on nitrophthalonitrile, manufacturers can mitigate risks associated with the sourcing of hazardous intermediates, which are often subject to strict transportation and storage regulations. The use of common reagents like mercapto compounds and hydrogen peroxide simplifies the procurement landscape, allowing for greater flexibility in vendor selection and potentially lower raw material costs due to the commoditized nature of these chemicals. Additionally, the improved solubility of the final products reduces the need for extensive milling or dispersion aids in downstream formulation, further driving down the total cost of ownership for end-users in the ink and coating industries.
- Cost Reduction in Manufacturing: The streamlined synthetic route significantly reduces the number of unit operations required compared to traditional methods. By avoiding the complex purification steps often needed to remove nitro-containing byproducts, the process lowers energy consumption and solvent usage. The ability to perform certain steps, such as oxidation and amidation, without isolating intermediates (telescoped synthesis) further enhances operational efficiency. This reduction in processing time and resource intensity translates directly into improved margins, making the production of high-performance functional materials more economically viable without compromising on quality standards.
- Enhanced Supply Chain Reliability: Diversifying the raw material base away from specialized nitro compounds towards widely available thiols and phthalimides strengthens supply chain security. This shift reduces vulnerability to supply disruptions caused by regulatory changes or production issues at specific nitro-chemical facilities. Furthermore, the robustness of the reaction conditions, which tolerate a range of temperatures and solvent systems, allows for greater flexibility in manufacturing scheduling. For supply chain planners, this means more predictable lead times and the ability to scale production volumes rapidly in response to market demand fluctuations without encountering bottlenecks related to rare precursor availability.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing reaction conditions that are easily transferable from pilot plant to full-scale commercial production. The avoidance of heavy metal catalysts in the early stages and the use of aqueous workups for salt formation align with green chemistry principles. This environmental compatibility simplifies waste treatment protocols and reduces the regulatory burden associated with effluent discharge. For organizations committed to sustainability goals, adopting this technology demonstrates a proactive approach to reducing the environmental footprint of chemical manufacturing while maintaining high product performance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential adopters.
Q: Why is avoiding nitrophthalonitrile significant for industrial production?
A: Traditional methods relying on nitrophthalonitrile often require complex equipment and harsh conditions. This new route uses readily available phthalimides and thiols, simplifying the process and reducing safety risks associated with nitro compounds.
Q: How does this process improve the solubility of the final pigment?
A: By introducing ionic hydrophilic groups such as sulfo or carboxyl groups at specific positions on the benzene rings prior to cyclization, the resulting phthalocyanine exhibits superior water solubility and dispersion stability.
Q: What metals can be incorporated into the phthalocyanine core using this method?
A: The process is versatile and supports the incorporation of various metals including Copper (Cu), Nickel (Ni), Zinc (Zn), and Aluminum (Al), allowing for tuning of electronic and optical properties for specific applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phthalocyanine Intermediates Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing advanced synthesis technologies to maintain a competitive edge in the global market. Our team of experts possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative processes like the one described in CN100378102C can be successfully translated into industrial reality. We are equipped with rigorous QC labs and adhere to stringent purity specifications to deliver intermediates that meet the exacting demands of the pharmaceutical and electronic materials sectors. Our commitment to quality assurance guarantees that every batch of phthalocyanine precursor we supply performs consistently in your downstream applications.
We invite you to collaborate with us to explore how this novel synthesis route can optimize your production costs and enhance your product portfolio. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to support your decision-making process and help you secure a stable supply of high-quality functional materials for your future projects.
