Scalable Bronsted Acid Catalysis for Third-Generation EGFR Inhibitor Intermediates
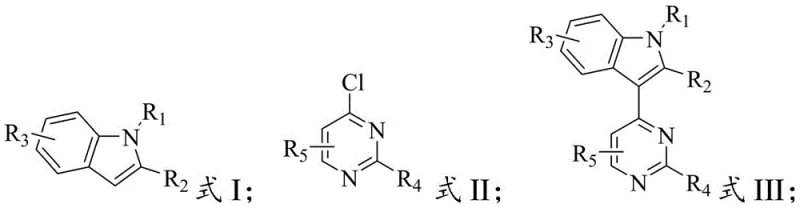
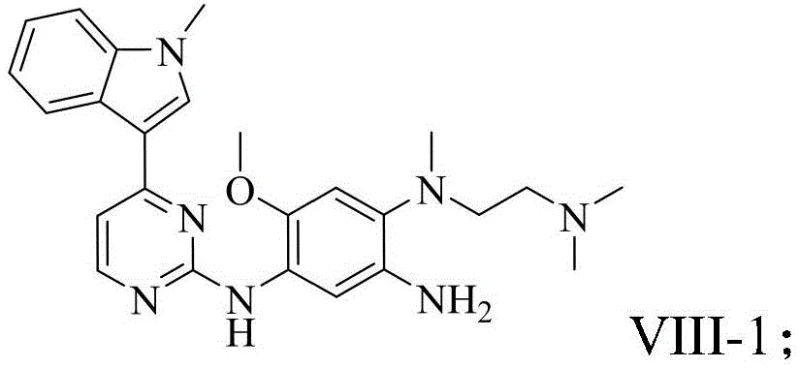
The pharmaceutical industry is constantly seeking more efficient and sustainable pathways for synthesizing complex heterocyclic scaffolds, particularly those serving as critical backbones for oncology therapeutics. Patent CN116283922A introduces a significant advancement in the preparation of 4-indolyl pyrimidine biaryl compounds, which are indispensable intermediates for third-generation EGFR inhibitors such as Osimertinib, Alflutinib, and Mobocertinib. This innovation addresses long-standing challenges in organic synthesis by replacing harsh transition metal catalysts with a milder Bronsted acid catalytic system. For R&D directors and procurement specialists, this shift represents not just a chemical optimization but a strategic opportunity to enhance supply chain resilience and reduce the environmental footprint of API manufacturing. The method demonstrates exceptional versatility, accommodating various substituents on both the indole and pyrimidine rings, thereby supporting the rapid development of next-generation kinase inhibitors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 4-indolyl pyrimidine biaryl motif has relied heavily on transition metal-catalyzed C-H arylation or Grignard reagent protocols. Conventional approaches utilizing catalysts like aluminum chloride (AlCl3) or iron(III) chloride (FeCl3) often struggle with moderate to low yields, typically ranging between 37% and 46% in comparative studies. Beyond the issue of efficiency, these metal-mediated reactions introduce significant downstream processing burdens. The removal of residual heavy metals to meet stringent pharmaceutical purity standards requires complex purification steps, such as extensive column chromatography or specialized scavenging resins, which drastically increase production costs and waste generation. Furthermore, the use of Grignard reagents necessitates strictly anhydrous conditions and cryogenic temperatures, posing safety risks and limiting the feasibility of scaling these reactions to multi-ton quantities required for commercial drug supply.
The Novel Approach
In contrast, the methodology disclosed in CN116283922A utilizes a Bronsted acid catalyst, such as bistrifluoromethanesulfonimide, to drive the C-H arylation reaction under significantly milder conditions. This metal-free approach eliminates the risk of heavy metal contamination at the source, streamlining the purification process to simple washing and filtration steps. The reaction operates effectively in polar solvents like hexafluoroisopropanol (HFIP) at temperatures between 30°C and 150°C, achieving yields of ≥74%, which is a substantial improvement over prior art. By avoiding the use of expensive and toxic transition metals, this novel route offers a cleaner, more cost-effective pathway that aligns perfectly with green chemistry principles. The robustness of this method allows for the efficient synthesis of diverse derivatives, making it an ideal candidate for the reliable supply of high-purity pharmaceutical intermediates.
Mechanistic Insights into Bronsted Acid-Catalyzed C-H Arylation
The core of this technological breakthrough lies in the activation of the 4-chloropyrimidine electrophile through protonation by the strong Bronsted acid. In traditional Lewis acid catalysis, the metal coordinates with the nitrogen atoms of the pyrimidine ring to enhance electrophilicity; however, this often leads to stable metal-complex byproducts that are difficult to separate. In this new protocol, the Bronsted acid protonates the pyrimidine nitrogen, generating a highly reactive cationic species that is susceptible to nucleophilic attack by the electron-rich C3 position of the indole ring. The choice of solvent plays a pivotal role in this mechanism; fluorinated alcohols like HFIP possess high ionizing power and hydrogen-bond donating ability, which stabilizes the developing positive charge in the transition state and facilitates the departure of the chloride leaving group. This synergistic effect between the acid catalyst and the specialized solvent environment lowers the activation energy of the reaction, allowing it to proceed efficiently without the need for external heating beyond moderate ranges.
From an impurity control perspective, the absence of transition metals fundamentally alters the impurity profile of the final product. Metal-catalyzed reactions often generate homocoupling byproducts or metal-inserted side products that co-elute with the desired compound, requiring rigorous analytical monitoring and repeated recrystallization. The Bronsted acid method minimizes these specific side reactions, resulting in a cleaner crude reaction mixture. The workup procedure described involves simply removing the solvent and washing the residue with a mixture of acetonitrile and water. This aqueous workup effectively removes the acid catalyst and polar byproducts, leaving the hydrophobic biaryl product as a high-purity solid. For quality control teams, this translates to a more predictable and consistent impurity spectrum, reducing the burden on QC labs and accelerating the release of batches for subsequent coupling steps in the API synthesis.
How to Synthesize 4-Indolyl Pyrimidine Biaryl Compounds Efficiently
The synthesis protocol outlined in the patent provides a straightforward and scalable procedure for producing these critical intermediates. The process begins with the precise mixing of the substituted indole and the 4-chloropyrimidine starting materials in a reaction vessel, followed by the addition of the Bronsted acid catalyst and the polar solvent. The reaction mixture is then heated to the specified temperature range and maintained for a duration sufficient to ensure complete conversion, typically between 6 to 48 hours depending on the specific substrates used. Upon completion, the solvent is removed under reduced pressure, and the resulting crude solid is subjected to a washing procedure using acetonitrile and water to remove residual catalyst and impurities. The detailed standardized synthesis steps for implementing this protocol in a GMP environment are provided below.
- Mix substituted indole compound (Formula I) and 4-chloropyrimidine compound (Formula II) with a Bronsted acid catalyst in a polar solvent.
- Heat the reaction mixture to 30-150°C and maintain for 6-48 hours to facilitate C-H arylation.
- Remove solvent, wash the crude product with acetonitrile and water, then filter and dry to obtain the pure biaryl compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Bronsted acid-catalyzed process offers tangible strategic benefits that extend beyond simple yield improvements. The elimination of transition metal catalysts removes a major cost driver associated with both the purchase of expensive metal salts and the disposal of hazardous metal waste. This shift significantly reduces the overall cost of goods sold (COGS) for the intermediate, allowing for more competitive pricing in the global market. Additionally, the simplified workup procedure, which relies on filtration and washing rather than complex chromatographic separation, shortens the production cycle time. This efficiency gain enhances the responsiveness of the supply chain, enabling manufacturers to react more quickly to fluctuations in demand for downstream EGFR inhibitors without compromising on quality or regulatory compliance.
- Cost Reduction in Manufacturing: The removal of transition metals like iron or aluminum from the process eliminates the need for costly metal scavengers and extensive purification steps. This simplification directly lowers operational expenditures related to raw materials and waste treatment. Furthermore, the ability to recover and recycle polar solvents like HFIP contributes to long-term sustainability and cost stability. By streamlining the synthesis into fewer unit operations, manufacturers can achieve substantial cost savings in labor and equipment utilization, making the production of high-purity pharmaceutical intermediates more economically viable.
- Enhanced Supply Chain Reliability: The starting materials required for this synthesis, such as substituted indoles and chloropyrimidines, are commercially available and widely sourced, reducing the risk of supply bottlenecks. The robustness of the reaction conditions, which tolerate a range of temperatures and do not require strict anhydrous environments like Grignard reactions, ensures consistent batch-to-batch performance. This reliability is crucial for maintaining continuous API production schedules and meeting the rigorous delivery timelines expected by multinational pharmaceutical partners. A stable and predictable supply of key intermediates mitigates the risk of drug shortages and supports uninterrupted patient access to life-saving therapies.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, operating at moderate temperatures and pressures that are compatible with standard stainless steel reactor infrastructure. The absence of pyrophoric reagents and toxic heavy metals simplifies safety protocols and reduces the regulatory burden associated with environmental discharge permits. This alignment with green chemistry principles not only improves the corporate sustainability profile but also future-proofs the manufacturing site against increasingly stringent environmental regulations. The ability to scale from kilogram to multi-ton quantities while maintaining high purity standards ensures that the supply chain can grow seamlessly alongside the commercial success of the final drug product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis method. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on how this technology can be integrated into existing manufacturing workflows. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their specific pipeline projects.
Q: How does this Bronsted acid method improve upon traditional transition metal catalysis?
A: Traditional methods using FeCl3 or Grignard reagents often suffer from low yields (37-46%) and difficult metal removal. This new method achieves yields ≥74% without transition metals, simplifying purification and reducing heavy metal impurity risks.
Q: What specific solvents are recommended for optimal reaction efficiency?
A: The patent highlights hexafluoroisopropanol (HFIP) as the preferred polar solvent due to its ability to stabilize cationic intermediates, though trifluoroethanol, methanol, and isopropanol are also viable alternatives depending on cost and availability.
Q: Is this process suitable for large-scale manufacturing of EGFR inhibitors?
A: Yes, the process is designed for industrial scalability. It utilizes commercially available starting materials, operates at moderate temperatures (30-150°C), and allows for solvent recovery, making it economically viable for producing intermediates like those for Osimertinib and Alflutinib.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Indolyl Pyrimidine Biaryl Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthetic routes in the development of modern oncology therapeutics. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of high-quality intermediates. We are fully equipped to implement the Bronsted acid-catalyzed methodology described in CN116283922A, leveraging our state-of-the-art facilities and rigorous QC labs to meet stringent purity specifications. Our commitment to excellence extends beyond mere synthesis; we partner with our clients to optimize every step of the process, ensuring that the final product meets the highest standards required for GMP API manufacturing.
We invite you to discuss how our advanced capabilities can support your project goals and drive efficiency in your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can deliver value to your organization. Let us be your trusted partner in bringing next-generation EGFR inhibitors from the lab to the market.
