Advanced Green Synthesis of 4-Chloro-3-Trifluoromethyl Phenylisocyanate for Scalable API Production
Advanced Green Synthesis of 4-Chloro-3-Trifluoromethyl Phenylisocyanate for Scalable API Production
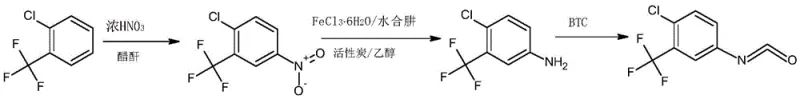
The global demand for high-performance kinase inhibitors continues to surge, driving the need for robust and environmentally sustainable supply chains for critical pharmaceutical intermediates. Patent CN110885298B introduces a transformative synthesis method for 4-chloro-3-(trifluoromethyl) phenylisocyanate, a pivotal building block in the manufacture of Sorafenib, a first-in-class oral multi-kinase inhibitor used for treating advanced renal cell carcinoma. This technical breakthrough addresses long-standing inefficiencies in traditional manufacturing by replacing hazardous mixed-acid nitration and sludge-generating reduction processes with cleaner, catalytic alternatives. For R&D directors and procurement strategists, this patent represents a significant opportunity to optimize cost structures while enhancing the safety profile of API production. By leveraging acetyl nitrate for low-temperature nitration and a novel iron-free reduction system, the process achieves superior purity profiles exceeding 99.8%, ensuring that downstream drug synthesis meets the rigorous standards required by global regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 4-chloro-3-(trifluoromethyl) phenylisocyanate has relied on archaic and hazardous chemical protocols that pose significant operational and environmental risks. The traditional nitration step typically employs a concentrated sulfuric acid and nitric acid mixed system, which necessitates elevated reaction temperatures to drive the conversion. These harsh conditions not only increase the potential for thermal runaway incidents but also promote the formation of unwanted isomer impurities and multi-nitrated byproducts, complicating downstream purification. Furthermore, the subsequent reduction of the nitro intermediate traditionally utilizes an iron powder and ethanol system. While effective chemically, this method generates substantial quantities of iron mud waste residue, a solid sludge that is notoriously difficult and expensive to treat and dispose of in compliance with modern environmental regulations. Additionally, the evolution of hydrogen gas during the iron powder reduction introduces inherent safety hazards regarding explosion risks in large-scale reactors.
The Novel Approach
In stark contrast to these legacy methods, the innovative pathway detailed in the patent utilizes a sophisticated acetic anhydride and concentrated nitric acid system for the initial nitration. This modification allows the reaction to proceed efficiently at significantly lower temperatures, typically between 10-15°C, by harnessing the potent nitrating capability of in-situ generated acetyl nitrate. This mild condition drastically suppresses the formation of poly-nitrated impurities and enhances process safety. Following nitration, the process employs a groundbreaking reduction system comprising ferric chloride hexahydrate (FeCl3·6H2O), activated carbon, and hydrazine hydrate. This catalytic approach completely eliminates the generation of iron mud, thereby resolving the major waste disposal bottleneck associated with traditional iron powder reduction. The seamless integration of these green chemistry principles results in a streamlined workflow that is not only safer but also economically superior due to reduced waste treatment costs and higher overall yields.
Mechanistic Insights into FeCl3-Catalyzed Reduction and Acetyl Nitrate Nitration
The core chemical innovation lies in the mechanistic efficiency of the acetic anhydride-mediated nitration and the subsequent catalytic reduction cycle. In the nitration phase, acetic anhydride reacts with concentrated nitric acid to form acetyl nitrate, a highly reactive nitrating agent that functions effectively under mild thermal conditions. This mechanism ensures high regioselectivity for the 4-nitro position on the chlorobenzene ring, minimizing the energy input required and preventing the degradation of the sensitive trifluoromethyl group. The reaction mixture is subsequently neutralized with a sodium hydroxide solution, allowing for clean phase separation and isolation of the nitro intermediate with minimal acidic waste. This contrasts sharply with the sulfonation side reactions often observed in sulfuric acid-based systems, which can lead to complex impurity profiles that are difficult to remove.
Regarding the reduction mechanism, the FeCl3·6H2O/activated carbon/hydrazine hydrate system operates through a catalytic hydrogen transfer pathway rather than stoichiometric metal consumption. The activated carbon serves as a support that enhances the dispersion of the ferric catalyst, facilitating the efficient transfer of electrons from hydrazine to the nitro group. This catalytic cycle reduces the nitro group to an amine without consuming the iron species in a way that generates solid precipitates. Consequently, the reaction mixture remains homogeneous or easily filterable, avoiding the formation of the voluminous iron oxide sludge characteristic of Bechamp reductions. This mechanistic elegance translates directly to operational simplicity, as the workup involves simple hot filtration and solvent extraction, yielding a high-purity amine ready for the final phosgenation step with triphosgene.
How to Synthesize 4-Chloro-3-(trifluoromethyl) Phenylisocyanate Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize the benefits of the novel catalytic systems. The process begins with the careful addition of concentrated nitric acid to a mixture of o-chlorotrifluoromethyl benzene and acetic anhydride, maintaining strict temperature control to leverage the acetyl nitrate mechanism. Following the isolation of the nitro intermediate, the reduction step utilizes refluxing ethanol as the solvent, with hydrazine hydrate added dropwise to manage the exotherm and ensure complete conversion. The final conversion to the isocyanate involves reacting the purified amine with triphosgene in the presence of a catalyst such as DMAP or pyridine. Detailed standardized operating procedures for each stage, including specific mass ratios and distillation parameters, are essential for reproducible high-yield manufacturing.
- Nitration of o-chlorotrifluoromethyl benzene using acetic anhydride and concentrated nitric acid at 10-15°C.
- Reduction of the nitro intermediate using FeCl3·6H2O, activated carbon, and hydrazine hydrate in ethanol.
- Reaction of the resulting aniline with triphosgene and a catalyst to form the final isocyanate product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented synthesis method offers profound strategic advantages beyond mere technical novelty. The elimination of the traditional iron powder reduction step fundamentally alters the cost structure of manufacturing by removing the need for extensive solid waste handling and disposal infrastructure. Traditional iron mud disposal is a significant line item in chemical manufacturing budgets, and its removal leads to substantial cost savings and a reduced environmental footprint. Furthermore, the use of milder nitration conditions reduces the energy consumption associated with heating and cooling large-scale reactors, contributing to lower utility costs and a more sustainable production profile. These efficiencies make the supply of this critical intermediate more resilient and cost-effective in the long term.
- Cost Reduction in Manufacturing: The transition to a catalytic reduction system eliminates the purchase of stoichiometric amounts of iron powder and the subsequent costs associated with filtering and disposing of tons of iron sludge. By replacing a waste-generating stoichiometric reagent with a catalytic amount of ferric chloride, the process significantly lowers raw material consumption and waste treatment expenses. Additionally, the higher selectivity of the acetic anhydride nitration reduces the loss of valuable starting materials to byproduct formation, further optimizing the overall material balance and reducing the cost per kilogram of the final API intermediate.
- Enhanced Supply Chain Reliability: The simplified workup procedures, particularly the absence of difficult-to-filter iron mud, streamline the production timeline and reduce the risk of batch delays caused by equipment fouling or waste handling bottlenecks. The use of readily available reagents like acetic anhydride and hydrazine hydrate ensures that the supply chain is not dependent on specialized or volatile raw materials that might face market shortages. This robustness ensures a consistent and reliable flow of high-purity intermediates to downstream API manufacturers, safeguarding against production interruptions.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, with safety features such as low-temperature nitration and the absence of hydrogen gas evolution making it suitable for large-scale commercial reactors. The drastic reduction in hazardous waste generation aligns perfectly with increasingly stringent global environmental regulations, future-proofing the manufacturing site against regulatory tightening. This compliance advantage minimizes the risk of fines or shutdowns, ensuring long-term operational continuity for the supply of this vital oncology drug precursor.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthesis pathway. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this method for commercial production. The answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a factual basis for decision-making.
Q: How does the new nitration method improve safety compared to traditional mixed acid systems?
A: The patented method replaces the traditional sulfuric/nitric acid mixed system with an acetic anhydride/nitric acid system. This utilizes acetyl nitrate for strong nitration at lower temperatures (10-15°C), significantly reducing the risk of thermal runaway and minimizing the formation of multi-nitration impurities.
Q: What are the environmental benefits of the FeCl3/hydrazine reduction system?
A: Unlike traditional iron powder reduction which generates massive amounts of difficult-to-treat iron mud waste, this catalytic system uses FeCl3·6H2O and activated carbon. This eliminates solid sludge waste, drastically reducing environmental pressure and waste disposal costs.
Q: What purity levels can be achieved with this synthesis route?
A: The process is optimized to achieve a total molar yield of over 80% and a final product purity exceeding 99.8%, making it highly suitable for the stringent quality requirements of oncology drug manufacturing like Sorafenib.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Chloro-3-(trifluoromethyl) Phenylisocyanate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development and commercialization of life-saving oncology therapies. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of materials regardless of their development stage. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 4-chloro-3-(trifluoromethyl) phenylisocyanate meets the exacting standards required for Sorafenib synthesis. We are committed to leveraging advanced green chemistry techniques to deliver products that are not only pure but also produced with a commitment to sustainability and safety.
We invite pharmaceutical partners to collaborate with us to optimize their supply chains and reduce manufacturing costs through the adoption of this superior synthesis technology. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your project timelines while ensuring the highest standards of quality and reliability.
