Advanced Manufacturing of 4-Pyridylaldehyde via Efficient N-Oxidation and Rearrangement Technology
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and cost-effective pathways for critical intermediates like 4-pyridylaldehyde, a key building block for drugs such as Donepezil hydrochloride. A recent technological breakthrough detailed in patent CN114380737A introduces a highly efficient preparation method that addresses long-standing challenges in yield, safety, and environmental impact. This novel approach utilizes 4-methylpyridine (4-picoline) as a readily available raw material, subjecting it to a sophisticated two-step sequence involving nitrogen oxidation and subsequent rearrangement. By shifting away from harsh gas-phase oxidations or toxic heavy metal reagents, this technology offers a compelling value proposition for manufacturers aiming to optimize their supply chains for high-purity pharmaceutical intermediates. The method not only simplifies the operational workflow but also drastically reduces the generation of hazardous three wastes, aligning perfectly with modern green chemistry principles and stringent regulatory requirements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-pyridylaldehyde has been plagued by significant technical and economic hurdles that hinder efficient commercial scale-up. Traditional catalytic oxidation methods often rely on gas-phase reactions over Vanadium-Molybdenum (V-Mo) catalysts at extreme temperatures approaching 400°C, necessitating massive capital investment in specialized high-temperature reactors and posing severe safety risks. Alternative liquid-phase routes, such as the oxidation of 4-pyridinemethanol using Pyridinium Chlorochromate (PCC), introduce different sets of problems, including the generation of substantial quantities of toxic chromium-containing solid waste and difficulties in product separation due to adsorption issues.
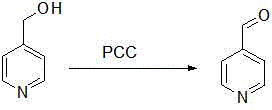
Furthermore, other documented pathways involving 4-cyanopyridine require high-pressure hydrogenation equipment resistant to strong acids, while routes utilizing hydrazine hydrate carry inherent explosion hazards and involve multi-step sequences that erode overall yield. These conventional methodologies collectively result in higher production costs, complex waste treatment protocols, and inconsistent product quality, creating a pressing need for a more streamlined and sustainable manufacturing solution.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN114380737A presents a streamlined, two-step liquid-phase process that operates under mild conditions, typically between 30°C and 80°C. This innovative route begins with the selective N-oxidation of 4-methylpyridine using hydrogen peroxide and a heteropoly acid catalyst, followed by a rearrangement reaction mediated by cesium fluoride. The elegance of this chemistry lies in its ability to bypass high-energy barriers and toxic reagents, directly converting a commodity chemical into a high-value aldehyde with exceptional efficiency.
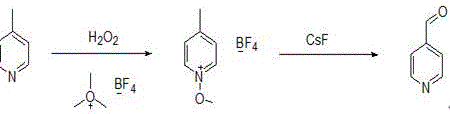
By employing this strategy, manufacturers can achieve high yields without the need for expensive high-pressure infrastructure or the handling of explosive intermediates. The process is designed for industrial viability, offering a clear pathway to cost reduction in pharmaceutical intermediates manufacturing through simplified unit operations and reduced raw material consumption. This represents a paradigm shift from brute-force oxidation to precise, catalytic molecular transformation.
Mechanistic Insights into N-Oxidation and Fluoride-Induced Rearrangement
The core of this technological advancement relies on a precise mechanistic sequence that ensures high selectivity and minimizes byproduct formation. The first stage involves the activation of the pyridine nitrogen atom through oxidation with hydrogen peroxide, facilitated by catalysts such as phosphomolybdic acid or phosphotungstic acid. This N-oxidation step is critical as it electronically activates the methyl group at the 4-position, making it susceptible to subsequent functionalization. The use of heteropoly acids provides a robust acidic environment that promotes oxidation while remaining stable under the reaction conditions, ensuring that the pyridine ring remains intact and preventing over-oxidation to the corresponding carboxylic acid.
Following the isolation of the N-oxide intermediate, the second stage employs a silylating agent, specifically identified in the patent context as reacting with trimethylsilyl derivatives (noted as trimethyl tartar tetrafluoroborate in the text, functionally acting as a silyl source), to protect and activate the oxygen. The subsequent addition of cesium fluoride acts as a potent fluoride source that triggers a rearrangement. The fluoride ion attacks the silicon center, facilitating the migration of the oxygen functionality and the eventual elimination to form the aldehyde group. This mechanism allows for the direct conversion of the methyl group to an aldehyde without passing through unstable alcohol or halide intermediates that often degrade or polymerize. The inclusion of a recrystallization step for the intermediate using ethyl acetate serves as a powerful purification checkpoint, removing trace impurities before the final rearrangement, thereby guaranteeing the high purity of the final distilled product.
How to Synthesize 4-Pyridylaldehyde Efficiently
To implement this advanced synthesis route effectively, operators must adhere to strict control over reaction temperatures and stoichiometry to maximize the formation of the N-oxide intermediate while minimizing side reactions. The process begins with the careful addition of hydrogen peroxide to the 4-methylpyridine solution containing the catalyst, maintaining a temperature range of 30-80°C to ensure complete conversion. Following the isolation and drying of the intermediate, the rearrangement step requires precise temperature control between 10-60°C during the addition of the fluoride source to manage the exotherm and ensure smooth conversion to the aldehyde. For a comprehensive guide on the specific molar ratios, solvent choices, and workup procedures validated by experimental data, please refer to the standardized protocol below.
- Oxidize 4-methylpyridine with hydrogen peroxide and a heteropoly acid catalyst to form the N-oxide intermediate.
- React the N-oxide with a silylating agent (trimethylsilyl tetrafluoroborate) to generate the silylated intermediate.
- Treat the intermediate with cesium fluoride to induce rearrangement and release the final 4-pyridylaldehyde product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis method translates into tangible strategic benefits that extend beyond simple chemical yield. By eliminating the need for specialized high-temperature gas-phase reactors or high-pressure hydrogenation vessels, the capital expenditure required for setting up production lines is significantly reduced. Furthermore, the reliance on commodity chemicals like 4-methylpyridine and hydrogen peroxide, rather than expensive or hazardous reagents like PCC or hydrazine hydrate, stabilizes the raw material supply chain and mitigates the risk of price volatility associated with specialty reagents. This stability is crucial for maintaining consistent production schedules and meeting the rigorous delivery timelines demanded by global pharmaceutical clients.
- Cost Reduction in Manufacturing: The economic model of this process is driven by the elimination of costly waste treatment and expensive reagents. By avoiding the use of heavy metal oxidants like chromium, the facility avoids the substantial costs associated with hazardous waste disposal and environmental compliance remediation. Additionally, the high yield reported in the examples, reaching over 90% for the intermediate and nearly 80% for the final product after distillation, means that less raw material is wasted per kilogram of output. This efficiency directly lowers the cost of goods sold (COGS), allowing for more competitive pricing in the market for reliable pharmaceutical intermediates suppliers.
- Enhanced Supply Chain Reliability: The simplicity of the two-step liquid-phase process enhances operational reliability and reduces the likelihood of unplanned downtime. Unlike multi-step routes that involve isolating unstable intermediates or handling explosive substances, this method uses stable reagents and straightforward workup procedures involving filtration and crystallization. This robustness ensures a continuous flow of production, reducing lead time for high-purity pharmaceutical intermediates and enabling manufacturers to respond quickly to fluctuations in market demand without compromising on safety or quality standards.
- Scalability and Environmental Compliance: From a sustainability perspective, this route offers a distinct advantage by generating significantly less three wastes compared to traditional methods. The absence of heavy metal sludge and the use of aqueous hydrogen peroxide, which decomposes into water and oxygen, align with increasingly strict global environmental regulations. This green profile facilitates easier permitting for new production capacity and reduces the long-term liability associated with environmental contamination. The process is inherently scalable from pilot plant to commercial tonnage, supporting the commercial scale-up of complex pharmaceutical intermediates with a minimal environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aimed at clarifying the operational benefits and safety profiles for potential partners and technical stakeholders.
Q: How does this new method compare to traditional catalytic oxidation in terms of safety?
A: Unlike traditional gas-phase oxidation which requires temperatures around 400°C and specialized equipment, this liquid-phase method operates at mild temperatures (30-80°C), significantly reducing thermal risks and equipment investment.
Q: What are the purity advantages of the crystallization step in this process?
A: The process includes a recrystallization step for Intermediate I using ethyl acetate, which effectively removes unreacted starting materials and side products, ensuring the final distillation yields a high-purity product suitable for sensitive API synthesis.
Q: Why is this route considered more environmentally friendly than the PCC oxidation method?
A: This route eliminates the use of Pyridinium Chlorochromate (PCC), a toxic heavy metal oxidant that generates large amounts of hazardous solid waste, replacing it with safer hydrogen peroxide and recoverable catalysts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Pyridylaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of downstream drug development and manufacturing. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering 4-pyridylaldehyde that meets stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify identity and assay. Our capability to adapt and optimize processes like the one described in CN114380737A allows us to offer a product that balances cost-efficiency with the highest standards of quality assurance.
We invite global partners to collaborate with us to leverage this advanced technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with a reliable, cost-effective, and sustainable source of this vital chemical building block.
