Industrial Scale Production of Fluorinated Methyl-Benzyl Alcohol via Advanced Catalytic Hydrogenolysis
Industrial Scale Production of Fluorinated Methyl-Benzyl Alcohol via Advanced Catalytic Hydrogenolysis
The global demand for high-performance agrochemical and pharmaceutical intermediates continues to drive innovation in synthetic methodology, particularly for fluorinated aromatic compounds which exhibit enhanced metabolic stability and biological activity. Patent CN1466560A introduces a transformative industrial process for the production of fluorinated methyl-benzyl alcohol, specifically addressing the limitations of traditional stoichiometric reduction methods. This technology leverages catalytic hydrogenolysis to selectively cleave one hydroxyl group from fluorinated benzenedimethanol precursors, offering a pathway that is not only chemically efficient but also economically superior for large-scale manufacturing. By shifting from hazardous hydride reagents to catalytic hydrogenation, this method aligns perfectly with modern green chemistry principles while ensuring the consistent supply of critical building blocks for insecticides and other organic syntheses.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fluorinated methyl-benzyl alcohols has relied heavily on stoichiometric reducing agents such as lithium aluminum hydride (LiAlH4) or sodium borohydride (NaBH4), as documented in prior art like JP-B-4-6694/1992. These conventional approaches present significant operational challenges for industrial procurement and safety teams, primarily due to the extreme sensitivity of reagents like LiAlH4 to moisture, which necessitates rigorous anhydrous conditions and specialized handling equipment. Furthermore, the use of Grignard reagents, such as methyl magnesium bromide reacting with pentafluorobenzyl alcohol, introduces complex waste streams containing magnesium salts that require costly disposal protocols. The reliance on these non-catalytic methods inherently limits scalability, as the exothermic nature of hydride reductions poses substantial safety risks when attempting to increase batch sizes beyond laboratory scales, thereby creating bottlenecks in the supply chain for high-volume agrochemical intermediates.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes catalytic hydrogenolysis, a method that fundamentally alters the economic and safety landscape of producing these valuable intermediates. By employing transition metal catalysts such as cobalt, nickel, or palladium under hydrogen pressure, the process achieves the selective reduction of one hydroxymethyl group to a methyl group without the need for stoichiometric quantities of expensive reducing agents. This shift allows for the use of common solvents like toluene, methanol, or dioxane, which are far more manageable from a logistics and storage perspective compared to the ether solvents often required for hydride chemistry. The ability to operate at moderate temperatures ranging from ambient to 250°C and pressures between 0.05 to 15 MPa provides process engineers with a flexible window to optimize throughput, ensuring that the production of high-purity fluorinated benzyl alcohol can be seamlessly integrated into existing hydrogenation infrastructure.
Mechanistic Insights into Catalytic Hydrogenolysis of Benzylic Alcohols
The core of this technological advancement lies in the selective activation of the benzylic carbon-oxygen bond on the fluorinated aromatic ring, a transformation that requires precise control to prevent over-reduction to the corresponding xylene derivatives. The reaction mechanism involves the adsorption of the fluorinated benzenedimethanol onto the active sites of the metal catalyst, where hydrogen molecules are dissociated into atomic hydrogen species. These active hydrogen species then attack the benzylic hydroxyl group, facilitating its removal as water while leaving the second hydroxymethyl group intact, thus yielding the desired fluorinated methyl-benzyl alcohol. The presence of electron-withdrawing fluorine atoms on the aromatic ring significantly influences the electronic density of the benzylic positions, necessitating the use of robust catalysts like sponge cobalt or supported rhenium that can maintain activity in the presence of these electronegative substituents.
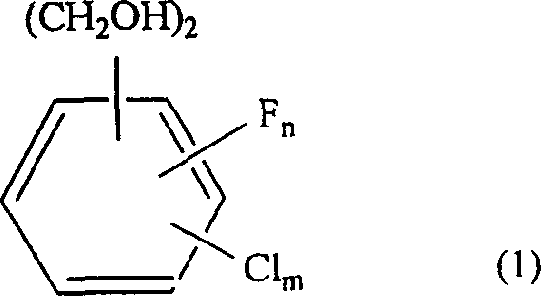
Impurity control is a critical aspect of this mechanism, as the competing reaction pathway involves the hydrogenolysis of both hydroxyl groups to form fluorinated xylenes, which are difficult to separate and represent a loss of yield. The patent data demonstrates that by carefully selecting the catalyst type—such as preferring sponge cobalt over certain supported variants—and optimizing the hydrogen partial pressure, the selectivity towards the mono-alcohol can be maximized. For instance, the use of sponge cobalt in toluene at 160°C has been shown to achieve conversion rates exceeding 94% with high selectivity, minimizing the formation of the fully reduced xylene byproduct. This level of control is essential for R&D directors who require intermediates with tight impurity profiles to ensure the efficacy and regulatory compliance of the final agrochemical or pharmaceutical products.
How to Synthesize 2,3,5,6-Tetrafluoro-4-methyl-benzyl Alcohol Efficiently
The synthesis of specific high-value derivatives, such as 2,3,5,6-tetrafluoro-4-methyl-benzyl alcohol, follows the general principles of the patented hydrogenolysis process but requires specific attention to catalyst loading and solvent choice to maximize yield. The procedure typically involves charging a high-pressure autoclave with the tetrafluorobenzenedimethanol starting material, a solvent system such as toluene or dioxane, and a predetermined amount of sponge cobalt or supported palladium catalyst. Following a thorough purge of the system to remove oxygen, hydrogen is introduced to reach the target pressure, and the mixture is heated to initiate the reaction, with progress monitored by hydrogen uptake. Detailed standardized synthesis steps see the guide below.
- Charge a high-pressure reactor with fluorinated benzenedimethanol, a suitable solvent such as toluene or dioxane, and a metal catalyst like sponge cobalt or supported palladium.
- Purge the system with inert gas followed by hydrogen, then pressurize to 0.05-15 MPa and heat to a temperature between ambient and 250°C.
- Maintain reaction conditions until hydrogen absorption ceases, then cool, filter the catalyst, and purify the product via distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this catalytic hydrogenolysis process represents a strategic opportunity to optimize the cost structure and reliability of the raw material supply base. The elimination of stoichiometric reducing agents like lithium aluminum hydride removes a major cost driver, as these reagents are not only expensive to purchase but also incur significant costs related to safe storage, transportation, and the disposal of hazardous waste byproducts. By switching to a catalytic system that utilizes hydrogen gas and reusable metal catalysts, manufacturers can achieve substantial cost savings in fine chemical manufacturing, allowing for more competitive pricing of the final fluorinated intermediates without compromising on quality or purity specifications.
- Cost Reduction in Manufacturing: The transition from stoichiometric reduction to catalytic hydrogenolysis drastically simplifies the bill of materials by removing the need for high-cost hydride reagents and their associated quenching agents. This process change effectively eliminates the generation of large volumes of inorganic salt waste, such as aluminum or boron salts, which significantly reduces waste treatment expenses and environmental compliance burdens. Furthermore, the catalysts employed, particularly base metal options like cobalt and nickel, are far more economical than precious metal alternatives or single-use chemical reducers, leading to a lower overall cost of goods sold for the produced fluorinated methyl-benzyl alcohol.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like hydrogen gas and common organic solvents such as toluene or methanol ensures a stable and resilient supply chain that is less susceptible to the market volatility often seen with specialized reducing agents. The robustness of the sponge metal catalysts allows for longer campaign runs and easier regeneration or replacement, minimizing unplanned downtime and ensuring consistent delivery schedules for downstream customers. This reliability is crucial for maintaining continuous production lines in the agrochemical sector, where interruptions in the supply of key intermediates can halt the manufacturing of finished pesticide formulations.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing standard high-pressure reactor technology that is widely available in chemical manufacturing facilities, thereby facilitating rapid scale-up from pilot plant to commercial production volumes. From an environmental perspective, the atom economy of hydrogenolysis is superior to stoichiometric reduction, as the only byproduct is water, aligning with increasingly stringent global regulations on industrial emissions and waste discharge. This green chemistry profile enhances the sustainability credentials of the supply chain, appealing to end-users who are prioritizing environmentally responsible sourcing strategies for their agricultural and pharmaceutical products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this hydrogenolysis technology, based on the specific embodiments and data provided in the patent literature. These insights are designed to clarify the operational parameters and benefits for technical decision-makers evaluating this route for potential adoption or outsourcing. Understanding these details is vital for assessing the feasibility of integrating this process into existing manufacturing workflows.
Q: What are the advantages of using catalytic hydrogenolysis over lithium aluminum hydride for this synthesis?
A: Catalytic hydrogenolysis eliminates the need for expensive, moisture-sensitive stoichiometric reducing agents like LiAlH4, significantly improving safety profiles and reducing waste disposal costs associated with aluminum salts.
Q: Which catalysts are most effective for producing tetrafluoro-methyl-benzyl alcohol?
A: Patent data indicates that sponge cobalt, modified sponge cobalt, and supported palladium catalysts provide high conversion rates and selectivity, with sponge cobalt showing particularly robust performance in toluene solvents.
Q: Can this process be scaled for commercial agrochemical production?
A: Yes, the process utilizes standard high-pressure hydrogenation equipment and common solvents like toluene, making it highly amenable to large-scale commercial manufacturing without requiring specialized cryogenic or anhydrous conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluorinated Methyl-Benzyl Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality fluorinated intermediates play in the development of next-generation agrochemicals and pharmaceuticals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of fluorinated methyl-benzyl alcohol meets the exacting standards required for sensitive synthetic applications, providing our partners with the confidence they need to advance their own R&D pipelines.
We invite you to contact our technical procurement team to discuss how we can support your specific project requirements with a Customized Cost-Saving Analysis tailored to your volume needs. By leveraging our expertise in catalytic hydrogenation and fluorine chemistry, we can help you optimize your supply chain for cost and reliability. Please reach out to request specific COA data and route feasibility assessments to determine the best path forward for your fluorinated intermediate sourcing strategy.
