Advanced Synthesis of Fluorinated Taxane Compounds for Overcoming Multidrug Resistance in Oncology
Introduction to Next-Generation Fluorinated Taxane Technology
The landscape of oncology therapeutics is constantly evolving to address the critical challenge of multidrug resistance (MDR), a phenomenon that severely limits the efficacy of first-line chemotherapies like Paclitaxel and Docetaxel. Patent CN110143934B introduces a groundbreaking series of fluorine-containing taxane compounds designed specifically to overcome these resistance mechanisms while maintaining potent antitumor activity. Unlike traditional taxanes that struggle with P-glycoprotein (P-gp) mediated efflux and poor water solubility requiring toxic solubilizers like Cremophor EL, these novel derivatives incorporate strategic fluorine substitutions at the C6 and C7 positions of the baccatin III core. 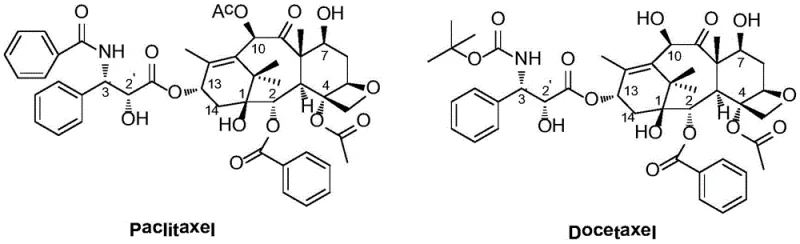 This structural modification not only enhances metabolic stability but also significantly improves cellular uptake in resistant cancer lines. The general structural formula, designated as Formula I in the patent, encompasses a range of derivatives (JY-01 to JY-06) where the C10 position is modified with various acyl or alkyl groups, offering a versatile platform for optimizing pharmacokinetic profiles.
This structural modification not only enhances metabolic stability but also significantly improves cellular uptake in resistant cancer lines. The general structural formula, designated as Formula I in the patent, encompasses a range of derivatives (JY-01 to JY-06) where the C10 position is modified with various acyl or alkyl groups, offering a versatile platform for optimizing pharmacokinetic profiles. 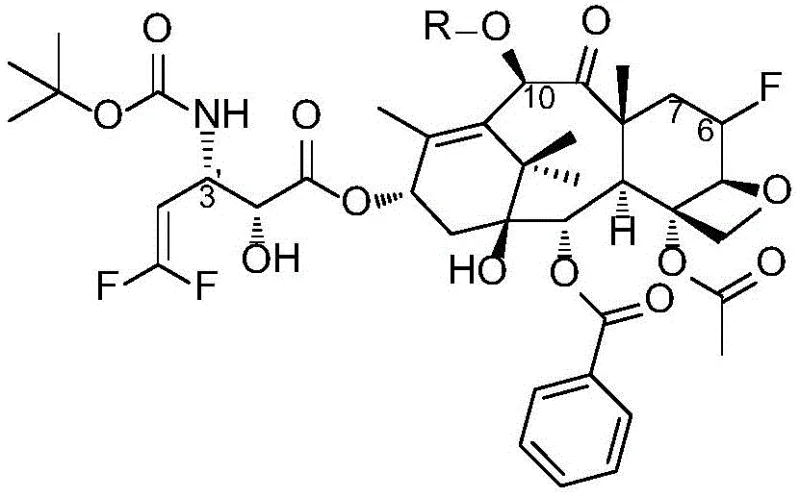 For pharmaceutical developers seeking a reliable fluorinated taxane intermediates supplier, this technology represents a pivotal shift towards more effective, next-generation anticancer agents capable of treating refractory tumors.
For pharmaceutical developers seeking a reliable fluorinated taxane intermediates supplier, this technology represents a pivotal shift towards more effective, next-generation anticancer agents capable of treating refractory tumors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional taxane chemotherapy has long been hampered by intrinsic and acquired resistance mechanisms, primarily driven by the overexpression of efflux transporters such as P-glycoprotein in tumor cells. When patients are treated with standard Paclitaxel, the drug is often pumped out of the cancer cells before it can stabilize microtubules and induce apoptosis, leading to treatment failure. Furthermore, the clinical formulation of Paclitaxel relies heavily on polyoxyethylene castor oil (Cremophor EL) and ethanol due to the drug's extreme hydrophobicity, which frequently causes severe hypersensitivity reactions and necessitates premedication with steroids and antihistamines. From a manufacturing perspective, the semi-synthetic production of traditional taxanes often involves complex protection and deprotection sequences that can suffer from low overall yields and difficult purification steps, particularly when introducing functional groups at the sterically hindered C6 and C7 positions of the taxane ring system. These limitations create a significant bottleneck in cost reduction in anticancer API manufacturing, as extensive processing is required to achieve pharmaceutical-grade purity without compromising the delicate stereochemistry of the molecule.
The Novel Approach
The innovative methodology described in the patent circumvents these issues by introducing a fluorine atom at the C6 position and removing the C7 hydroxyl group, creating a 6-fluoro-7-deoxy taxane scaffold that demonstrates exceptional resilience against MDR mechanisms. This novel approach utilizes a convergent synthesis strategy where a highly functionalized fluorinated baccatin III core is coupled with a specialized difluoro-vinyl beta-lactam side chain. 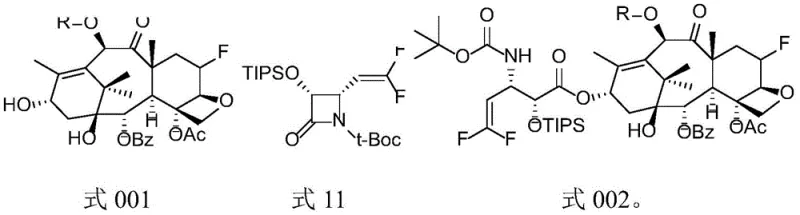 The key to this success lies in the 'Hilton docking reaction,' a base-mediated coupling that efficiently joins the C13 hydroxyl of the core with the beta-lactam nitrogen of the side chain under mild conditions (0°C to -40°C). This method avoids the harsh conditions often associated with traditional esterification at the C13 position, thereby preserving the integrity of the sensitive oxetane ring and other functional groups. By streamlining the synthesis into distinct modules for the core and the side chain, the process offers enhanced supply chain reliability and simplifies the scale-up of complex fluorinated taxane intermediates, ensuring consistent quality and batch-to-batch reproducibility essential for clinical applications.
The key to this success lies in the 'Hilton docking reaction,' a base-mediated coupling that efficiently joins the C13 hydroxyl of the core with the beta-lactam nitrogen of the side chain under mild conditions (0°C to -40°C). This method avoids the harsh conditions often associated with traditional esterification at the C13 position, thereby preserving the integrity of the sensitive oxetane ring and other functional groups. By streamlining the synthesis into distinct modules for the core and the side chain, the process offers enhanced supply chain reliability and simplifies the scale-up of complex fluorinated taxane intermediates, ensuring consistent quality and batch-to-batch reproducibility essential for clinical applications.
Mechanistic Insights into Fluorination and Beta-Lactam Coupling
The synthesis of the fluorinated taxane core begins with 10-deacetyl baccatin III (10-DAB), a readily available natural precursor. The mechanistic pathway involves a precise sequence of protection, elimination, and hydrofluorination. Initially, the C7 hydroxyl is protected with a triethylsilyl (TES) group, followed by acylation at the C10 position using cerium trichloride (CeCl3) catalysis to ensure regioselectivity. Subsequent removal of the C7 silyl group and conversion to a triflate leaving group sets the stage for an elimination reaction using DBU at elevated temperatures (100°C), generating a C6-C7 double bond. The critical fluorination step then employs hydrogen fluoride (HF) complexes to add fluorine across the double bond, yielding the 6-fluoro-7-deoxy configuration. 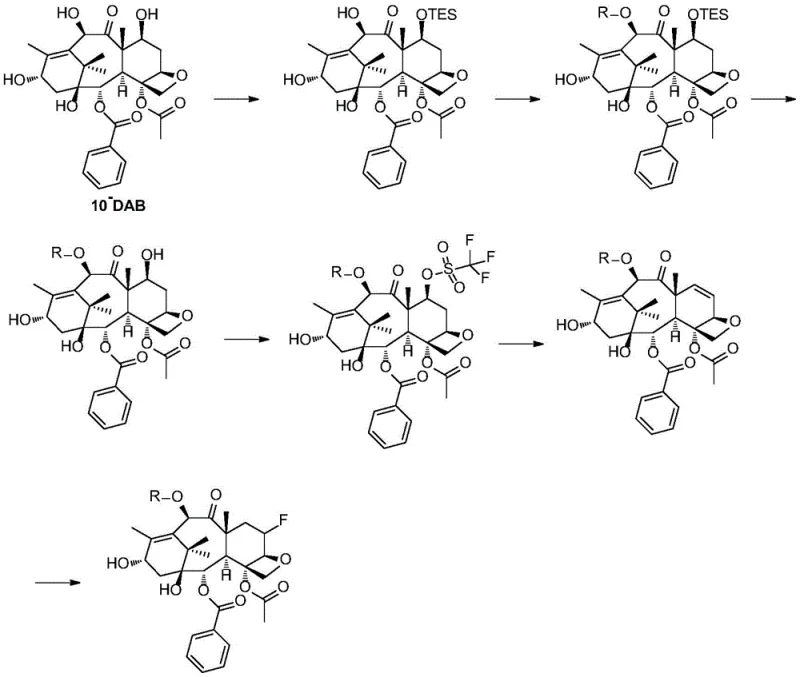 Parallel to this, the side chain precursor (Compound 11) is constructed via a Staudinger-type cyclization to form the beta-lactam ring, followed by ozonolysis and fluorination to install the difluoro-vinyl moiety. The final coupling mechanism relies on the deprotonation of the beta-lactam nitrogen by lithium hexamethyldisilazide (LHMDS), generating a nucleophilic species that attacks the C13 ester of the core. This mechanism is highly sensitive to temperature and base strength, requiring strict control to prevent epimerization at the C2' and C3' positions, which are critical for biological activity. The robustness of this mechanistic design ensures high-purity fluorinated taxane intermediates with minimal impurity profiles.
Parallel to this, the side chain precursor (Compound 11) is constructed via a Staudinger-type cyclization to form the beta-lactam ring, followed by ozonolysis and fluorination to install the difluoro-vinyl moiety. The final coupling mechanism relies on the deprotonation of the beta-lactam nitrogen by lithium hexamethyldisilazide (LHMDS), generating a nucleophilic species that attacks the C13 ester of the core. This mechanism is highly sensitive to temperature and base strength, requiring strict control to prevent epimerization at the C2' and C3' positions, which are critical for biological activity. The robustness of this mechanistic design ensures high-purity fluorinated taxane intermediates with minimal impurity profiles.
Impurity control is paramount in the production of complex oncology intermediates, and this patent outlines specific strategies to mitigate common side reactions. For instance, the use of HF/Pyridine complexes for the final deprotection of the silyl groups is conducted at controlled temperatures (0°C to room temperature) to prevent acid-catalyzed degradation of the oxetane ring or the ester linkages. Additionally, the purification protocols involve multiple extraction and chromatography steps using standard solvents like ethyl acetate and dichloromethane, which effectively remove unreacted starting materials and side products. The detailed NMR data provided in the patent confirms the stereochemical integrity of the final products, such as JY-01, demonstrating that the synthesis maintains the crucial (2'R, 3'S) configuration of the side chain. This level of mechanistic understanding allows manufacturers to implement rigorous QC labs and stringent purity specifications, ensuring that the final API intermediates meet the demanding requirements of global regulatory bodies.
How to Synthesize Fluorinated Taxane Compound JY-01 Efficiently
The efficient synthesis of JY-01 serves as a representative example of the broader platform technology described in the patent, demonstrating the feasibility of producing these high-value intermediates on a commercial scale. The process is divided into three main stages: the preparation of the fluorinated core, the synthesis of the beta-lactam side chain, and the final coupling and deprotection. Each stage utilizes commercially available reagents and standard laboratory equipment, facilitating easy technology transfer from R&D to pilot and production scales. The detailed standardized synthesis steps see the guide below, which outlines the specific stoichiometry, solvent systems, and reaction times required to achieve optimal yields and purity levels exceeding 95%. 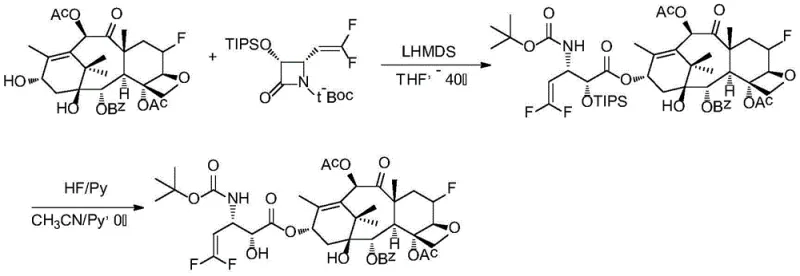 This streamlined approach minimizes the number of isolation steps and reduces solvent consumption, aligning with modern green chemistry principles while maintaining the high quality necessary for pharmaceutical applications.
This streamlined approach minimizes the number of isolation steps and reduces solvent consumption, aligning with modern green chemistry principles while maintaining the high quality necessary for pharmaceutical applications.
- Synthesize the 6-fluoro-7-deoxy-10-acyl-baccatin III core starting from 10-DAB via silylation, acylation, elimination, and hydrofluorination.
- Prepare the difluoro-vinyl beta-lactam side chain precursor (Compound 11) through a series of protection, condensation, and fluorination steps.
- Couple the core and side chain using LHMDS at -40°C in THF, followed by acidic deprotection with HF/Py to yield the final product with >95% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this fluorinated taxane synthesis route offers substantial strategic benefits beyond mere technical novelty. The primary advantage lies in the significant cost optimization achieved through the use of robust, scalable chemistry that avoids reliance on scarce or prohibitively expensive transition metal catalysts. By utilizing common reagents like LHMDS, HF/Py, and standard acid anhydrides, the manufacturing process reduces the complexity of raw material sourcing and mitigates the risk of supply disruptions associated with specialized catalysts. Furthermore, the convergent nature of the synthesis allows for the parallel production of the core and side chain, drastically simplifying inventory management and reducing lead time for high-purity fluorinated taxane intermediates. This modularity ensures that production bottlenecks in one branch of the synthesis do not halt the entire operation, thereby enhancing overall supply chain resilience and continuity.
- Cost Reduction in Manufacturing: The synthetic route eliminates the need for costly enzymatic resolutions or rare earth metal catalysts often found in older taxane semi-synthesis methods. Instead, it relies on efficient chemical transformations with high atom economy, such as the direct fluorination of the C6-C7 double bond. This simplification of the reaction sequence translates directly into lower operational expenditures (OPEX) and reduced waste disposal costs, providing a clear pathway for cost reduction in anticancer API manufacturing without compromising on the therapeutic potency of the final drug substance.
- Enhanced Supply Chain Reliability: The starting material, 10-deacetyl baccatin III (10-DAB), is a well-established commodity in the pharmaceutical industry with a stable global supply base. By building upon this reliable foundation and adding value through chemical modification rather than total synthesis, the process leverages existing supply chains while creating a differentiated, high-margin product. The use of standard organic solvents like THF and DCM further ensures that solvent availability is never a constraint, allowing for flexible production scheduling and rapid response to market demand fluctuations for these critical oncology intermediates.
- Scalability and Environmental Compliance: The reaction conditions described, such as the coupling at -40°C and deprotection at 0°C, are readily achievable in standard stainless steel reactors equipped with cryogenic cooling systems, making the commercial scale-up of complex fluorinated taxane intermediates straightforward. Moreover, the process generates manageable waste streams that can be treated using conventional methods, avoiding the generation of heavy metal-contaminated waste that requires specialized disposal. This alignment with environmental compliance standards reduces regulatory hurdles and facilitates faster approval for commercial production facilities, ensuring a sustainable and long-term supply of these vital therapeutic agents.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these novel fluorinated taxane compounds. The answers are derived directly from the experimental data and technical specifications outlined in patent CN110143934B, providing clarity for R&D teams and procurement specialists evaluating this technology for their pipelines. Understanding these details is crucial for assessing the feasibility of integrating these intermediates into existing drug development programs. These insights underscore the maturity of the technology and its readiness for industrial adoption.
Q: What is the primary advantage of these fluorinated taxanes over Paclitaxel?
A: According to patent CN110143934B, these compounds exhibit superior cytotoxicity against multidrug-resistant cell lines (MCF-7/Adr, NCI/Adr) and tumor stem cell-overexpressing lines (HCT-116++), overcoming the P-gp efflux mechanism that limits Paclitaxel.
Q: What are the critical reaction conditions for the key coupling step?
A: The Hilton docking reaction between the taxane core and the beta-lactam side chain requires strict temperature control between 0°C and -40°C using LHMDS as the base in THF solvent to ensure high stereochemical fidelity.
Q: Is the synthesis process scalable for commercial manufacturing?
A: Yes, the process utilizes standard organic solvents like THF, DCM, and acetonitrile, and avoids exotic transition metal catalysts, making it highly suitable for scale-up from kilogram to multi-ton production in GMP facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluorinated Taxane Intermediates Supplier
As the demand for next-generation oncology therapeutics grows, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM becomes essential for navigating the complexities of advanced intermediate synthesis. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from clinical trials to full-scale market supply. Our state-of-the-art facilities are equipped to handle the specific requirements of fluorinated chemistry, including specialized corrosion-resistant equipment for HF handling and precise temperature control systems for cryogenic reactions. With our rigorous QC labs and commitment to stringent purity specifications, we guarantee that every batch of fluorinated taxane intermediates meets the highest international standards for safety and efficacy.
We invite you to contact our technical procurement team to discuss your specific requirements and explore how our manufacturing capabilities can support your drug development goals. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to our optimized synthesis route. We are ready to provide specific COA data and route feasibility assessments tailored to your project timeline, ensuring that you have all the necessary information to make informed decisions. Let us be your partner in bringing these life-saving fluorinated taxane therapies to patients worldwide.
