Advanced Manufacturing of Chiral Tetrahydroisoquinoline Intermediates for Pharmaceutical Applications
Advanced Manufacturing of Chiral Tetrahydroisoquinoline Intermediates for Pharmaceutical Applications
The pharmaceutical industry's relentless pursuit of potent enzyme inhibitors and peptidomimetic drugs has placed a premium on high-purity chiral building blocks, specifically tetrahydroisoquinoline derivatives. Patent CN112724084B discloses a groundbreaking preparation method for synthesizing (S)-5-bromo-1,2,3,4-tetrahydro-N-Boc-isoquinoline-1-carboxylic acid, a critical intermediate for cardiovascular medicaments and bioactive alkaloids. This technical disclosure addresses a significant gap in the prior art, where effective synthesis methods for this specific molecular architecture were previously nonexistent or inefficient. By leveraging a sophisticated multi-step sequence involving protective group strategies and dynamic kinetic resolution, this process offers a robust pathway for producing complex heterocyclic compounds. For R&D directors and procurement specialists alike, understanding the nuances of this patented methodology is essential for securing a reliable supply chain for next-generation therapeutic agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral tetrahydroisoquinoline derivatives has been plagued by significant challenges regarding enantioselectivity and process stability. Traditional approaches often attempt direct resolution of the racemic mixture using various chiral amines while the molecule remains under Cbz protection. However, empirical data suggests that such direct resolution strategies frequently fail to achieve adequate stereochemical control, often resulting in enantiomeric excess (ee) values lower than 50% after a single resolution attempt. Furthermore, many existing routes rely on harsh reaction conditions or expensive, difficult-to-source chiral catalysts that complicate the purification process and introduce heavy metal impurities. These inefficiencies not only drive up the cost of goods sold (COGS) but also create bottlenecks in the supply chain, making it difficult for pharmaceutical manufacturers to scale production without compromising on purity specifications or facing extended lead times.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a strategic sequence that maximizes both yield and optical purity through a clever manipulation of reaction equilibria. The process begins with the protection of o-bromophenylethylamine, followed by a highly efficient cyclization with glyoxylic acid under specific acid catalysis. The true innovation lies in the final resolution step, where the removal of the Cbz group precedes a resolution mediated by L-tartaric acid in the presence of a catalytic amount of aldehyde. This unique combination facilitates the formation of an imine intermediate, allowing for the dynamic interconversion of chiral centers and driving the equilibrium toward the desired (S)-enantiomer. As illustrated in the overall reaction scheme below, this method transforms a potentially low-yielding resolution into a high-efficiency conversion process.
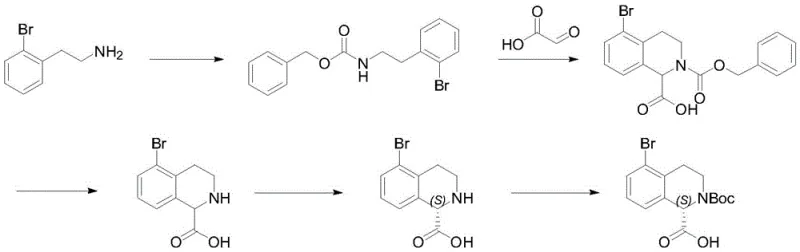
This streamlined pathway not only simplifies the operational complexity but also ensures that the final product meets the stringent purity requirements demanded by modern regulatory bodies. By avoiding the pitfalls of direct resolution on the protected species, the novel approach achieves exceptional stereochemical outcomes that were previously unattainable with standard techniques.
Mechanistic Insights into Acid-Catalyzed Cyclization and Chiral Resolution
The core of this synthesis relies on two critical mechanistic events: the construction of the isoquinoline core and the subsequent establishment of chirality. The cyclization step involves the reaction of the protected phenylethylamine with glyoxylic acid, catalyzed by either a strong Brønsted acid system (such as sulfuric acid/acetic acid) or a Lewis acid like tris(pentafluorophenyl)borane. This Pictet-Spengler-type cyclization is highly sensitive to reaction conditions; the patent specifies temperatures ranging from 30-35°C for the Brønsted acid method or 50-55°C for the Lewis acid variant. The choice of catalyst influences the electronic activation of the glyoxylic acid carbonyl, facilitating the electrophilic attack on the aromatic ring to close the tetrahydroisoquinoline ring system with high regioselectivity. This step is crucial for establishing the scaffold upon which the chiral center will later be defined, and the high yields reported (over 90% in some examples) demonstrate the robustness of this cyclization protocol.
Following the cyclization and subsequent deprotection via catalytic hydrogenation, the process enters its most sophisticated phase: the chiral resolution. Unlike static resolution methods that simply separate enantiomers, this method employs a dynamic kinetic resolution strategy. By introducing an aldehyde (such as n-butyraldehyde or isobutyraldehyde) alongside L-tartaric acid in an organic solvent like acetic acid or formic acid, an imine structure is transiently formed with the amine group. Under these acidic conditions, the chiral center at the C-1 position (connected to the carboxyl group) becomes labile, allowing the two configurations to mutually convert. The L-tartaric acid then selectively crystallizes or complexes with the desired (S)-configuration, effectively pulling the equilibrium and converting the entire racemic pool into the target enantiomer. The reaction scheme for this critical resolution and protection step is depicted below, highlighting the integration of resolution and Boc protection.
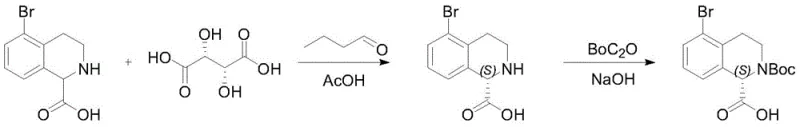
This mechanism explains the exceptionally high enantiomeric excess values (up to 99.6% ee) reported in the patent examples. The ability to racemize the unwanted enantiomer in situ and convert it into the desired product significantly improves the theoretical maximum yield from 50% to nearly 100% relative to the racemate, representing a massive efficiency gain for commercial manufacturing.
How to Synthesize (S)-5-Bromo-Isoquinoline Carboxylic Acid Efficiently
The synthesis of this high-value intermediate is structured into four distinct operational stages, each optimized for safety and scalability. The process begins with the formation of the carbamate protecting group, proceeds through the ring-closing cyclization, follows with the removal of the benzyl group via hydrogenation, and concludes with the chiral resolution and final Boc protection. Each step has been rigorously tested to ensure that impurities are minimized and that the reaction can be performed in standard glass-lined or stainless steel reactors. The detailed standardized synthesis steps, including precise stoichiometric ratios, temperature controls, and workup procedures, are outlined in the guide below to assist process chemists in replicating this high-yielding route.
- React o-bromophenylethylamine with benzyl chloroformate in the presence of an organic base to form the Cbz-protected intermediate.
- Perform a ring-closure reaction with glyoxylic acid under Lewis acid or Brønsted acid catalysis to form the tetrahydroisoquinoline core.
- Execute catalytic hydrogenation using Pd/C to remove the benzyl protecting group, yielding the free amine acid.
- Conduct chiral resolution using L-tartaric acid and an aldehyde additive, followed by Boc protection to obtain the final chiral product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from laboratory curiosity to commercial viability is often hindered by cost volatility and raw material scarcity. This patented process directly addresses these pain points by utilizing commodity chemicals as starting materials. The primary feedstock, o-bromophenylethylamine, and the cyclization agent, glyoxylic acid, are widely available in the global chemical market, reducing the risk of supply disruptions. Furthermore, the elimination of exotic chiral catalysts in favor of inexpensive L-tartaric acid and simple aldehydes drastically reduces the raw material cost profile. The process avoids the use of transition metals in the resolution step, which simplifies the downstream purification and eliminates the need for expensive heavy metal scavenging resins, leading to substantial cost savings in the overall manufacturing budget.
- Cost Reduction in Manufacturing: The economic viability of this route is underpinned by the high atom economy of the cyclization step and the near-quantitative yield of the dynamic kinetic resolution. By converting the entire racemic mixture into the desired chiral product rather than discarding half of the material, the effective cost per kilogram of the active chiral intermediate is significantly reduced. Additionally, the use of common solvents like dichloromethane, ethanol, and ethyl acetate allows for efficient solvent recovery and recycling systems, further lowering the environmental and financial burden of waste disposal. The avoidance of cryogenic conditions (most reactions proceed between 20-35°C) also results in lower energy consumption for heating and cooling utilities compared to processes requiring extreme temperatures.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the simplicity of the reagent list. Since the process does not rely on proprietary or single-source chiral ligands, manufacturers are not vulnerable to the monopolistic pricing or availability issues often associated with specialized catalysts. The robustness of the reaction conditions means that the process can be easily transferred between different manufacturing sites or contract development and manufacturing organizations (CDMOs) without extensive re-optimization. This flexibility ensures a continuous supply of high-purity pharmaceutical intermediates, mitigating the risk of production delays that could impact the launch timelines of downstream drug products.
- Scalability and Environmental Compliance: From an environmental and safety perspective, the process is designed with green chemistry principles in mind. The hydrogenation step uses standard palladium on carbon catalysts which can be filtered and recycled, minimizing heavy metal waste. The resolution step generates tartaric acid salts as byproducts, which are biodegradable and non-toxic, simplifying wastewater treatment protocols. The mild reaction temperatures and atmospheric or low-pressure hydrogenation conditions (0.1-0.3 MPa) reduce the operational hazards associated with high-pressure reactors, making the scale-up from pilot plant to multi-ton commercial production safer and more straightforward for engineering teams.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this synthesis method, we have compiled a list of frequently asked questions based on the patent specifications. These inquiries address common concerns regarding the scalability of the resolution step, the handling of chiral impurities, and the compatibility of this intermediate with downstream peptide coupling reactions. Understanding these details is vital for technical teams evaluating this route for inclusion in their regulatory filings or process validation dossiers.
Q: Why is an aldehyde additive used during the chiral resolution step?
A: The aldehyde facilitates the formation of an imine structure with the amine under acidic conditions. This allows for the mutual conversion of the two configurations at the chiral carbon center connected to the carboxyl group, enabling dynamic kinetic resolution where the racemate is completely converted into the desired (S)-enantiomer.
Q: What represents the key advantage of this synthesis route over conventional methods?
A: Conventional direct resolution methods often yield poor enantioselectivity (lower than 50% ee). This novel method utilizes a specific sequence of Cbz protection, efficient cyclization, and a tartaric acid-mediated resolution with aldehyde assistance, achieving high optical purity (up to 99.6% ee) and high chemical yield suitable for industrial scale-up.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is designed for scalability. It utilizes cheap and easily obtained raw materials like glyoxylic acid and o-bromophenylethylamine. The reaction conditions are mild (20-35°C for key steps), and the solvents used (DCM, ethanol, ethyl acetate) are standard industrial solvents, ensuring safety and cost-effectiveness for tonnage production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-5-Bromo-Isoquinoline Carboxylic Acid Supplier
The successful implementation of this sophisticated synthesis route requires a partner with deep expertise in chiral chemistry and process optimization. NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of this process, including low-temperature reactions and catalytic hydrogenation, while maintaining stringent purity specifications through our rigorous QC labs. We understand that the consistency of chiral intermediates is paramount for the efficacy of the final drug product, and our quality management systems are designed to ensure batch-to-batch reproducibility that exceeds industry standards.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this advanced technology for their drug development programs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this route can optimize your bill of materials. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples to accelerate your project timelines.
