Advancing Pharmaceutical Intermediate Production via Visible-Light Mediated Guanylhydrazone Synthesis
The pharmaceutical and fine chemical industries are constantly seeking more efficient and sustainable pathways to construct carbon-nitrogen bonds, which are fundamental motifs in bioactive molecules. Patent CN112812040B introduces a groundbreaking preparation method for amidine acylhydrazone compounds, specifically guanylhydrazone derivatives, utilizing visible-light-mediated photoredox catalysis. This technology represents a significant leap forward from traditional thermal methods, enabling the direct addition reaction between 3-oxo-nitrile compounds and 1,1-disubstituted hydrazine compounds under exceptionally mild conditions. By leveraging the unique ability of photocatalysts to induce single electron transfer processes, this invention achieves high conversion rates and exceptional product purity, reaching up to 99.9 percent, without the need for harsh oxidants or elevated temperatures. The versatility of this approach is demonstrated by the successful synthesis of a broad library of derivatives, showcasing its potential as a robust platform for generating high-purity pharmaceutical intermediates.
Traditionally, the construction of C-N bonds, particularly in the synthesis of guanylhydrazone scaffolds, has relied on methodologies that impose significant operational and safety burdens on manufacturing facilities. Conventional routes often necessitate the use of strong oxidizing agents, stoichiometric amounts of bases, and high-temperature conditions to drive the reaction to completion. Furthermore, many established protocols depend on transition metal catalysts, which introduce the risk of heavy metal contamination in the final active pharmaceutical ingredient (API), necessitating costly and time-consuming purification steps to meet regulatory limits. These harsh conditions can also lead to poor functional group tolerance, limiting the scope of substrates that can be utilized and resulting in lower overall yields due to side reactions or decomposition of sensitive intermediates. The reliance on thermal energy and aggressive reagents inherently increases the energy footprint and safety risks associated with large-scale production.
In stark contrast, the novel approach detailed in CN112812040B circumvents these limitations by employing visible light as a clean, renewable energy source to drive the transformation. This photocatalytic strategy operates at ambient temperatures ranging from 25°C to 35°C, effectively eliminating the thermal stress on reactants and allowing for the preservation of sensitive functional groups. The use of organic photocatalysts, such as Rose Bengal, Eosin Y, or metal complexes like tris(2,2'-bipyridine)ruthenium dichloride, facilitates the reaction through a radical mechanism that does not require external oxidants or strong bases. This shift not only simplifies the reaction workup but also significantly enhances the safety profile of the process by removing hazardous reagents from the workflow. The result is a streamlined synthetic route that delivers target guanylhydrazone compounds with superior purity profiles, directly addressing the stringent quality requirements of modern drug development.
Mechanistic Insights into Visible-Light Mediated Photoredox Catalysis
The core innovation of this technology lies in the activation of unactivated C-H bonds through a photoredox catalytic cycle. Upon irradiation with light in the range of 200nm to 1000nm, preferably around 470nm, the photocatalyst absorbs photons and transitions to an excited state. This excited species possesses sufficient redox potential to engage in single electron transfer (SET) processes with the substrate mixture. Specifically, the interaction between the excited photocatalyst and the 3-oxo-nitrile compound generates a radical intermediate, which subsequently undergoes addition with the 1,1-disubstituted hydrazine compound. This mechanism bypasses the high energy barriers associated with thermal activation, allowing the reaction to proceed smoothly at room temperature. The precise control over the electronic excitation ensures that the reaction pathway is highly selective, minimizing the formation of by-products that typically plague thermal condensation reactions.
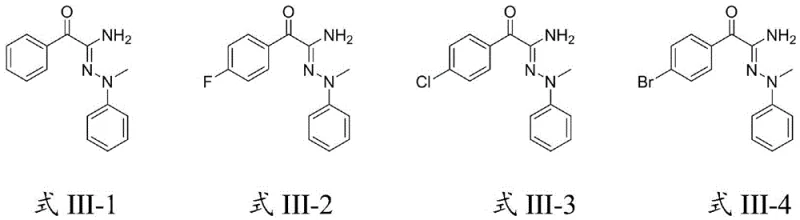
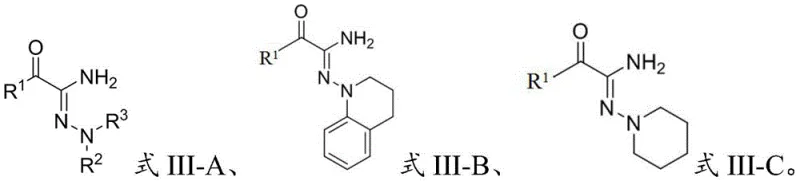
Furthermore, the mild nature of this photocatalytic system contributes significantly to impurity control, a critical parameter for R&D directors focused on regulatory compliance. By avoiding high temperatures and strong acids or bases, the process prevents common degradation pathways such as hydrolysis of the nitrile group or rearrangement of the hydrazone moiety. The reaction demonstrates excellent tolerance to various substituents on the aromatic rings, including halogens, alkyl groups, and alkoxy groups, as evidenced by the successful synthesis of diverse derivatives shown in the patent examples. This broad substrate scope implies that the electronic and steric properties of the reactants do not significantly hinder the catalytic cycle, ensuring consistent performance across a wide range of molecular architectures. The resulting products, characterized by the general structure of Formula III, exhibit high structural integrity and purity, facilitating downstream processing and formulation.
How to Synthesize Guanylhydrazone Derivatives Efficiently
The implementation of this photocatalytic protocol offers a straightforward and reproducible pathway for laboratory and pilot-scale synthesis. The process begins with the preparation of a reaction feed liquid by mixing the 3-oxo-nitrile starting material, the 1,1-disubstituted hydrazine derivative, and the chosen photocatalyst in a polar organic solvent such as ethyl acetate or acetonitrile. The molar ratio of the nitrile to the hydrazine is optimized between 1:2 and 1:3 to ensure complete consumption of the limiting reagent, while the photocatalyst loading is kept low, typically around 1 mol percent, reflecting its catalytic efficiency. Once the mixture is homogenized, it is subjected to irradiation under stirring, maintaining the temperature within the narrow window of 25°C to 35°C for a duration of 6 to 24 hours. Following the reaction, the target compounds are isolated through standard column chromatography using a petroleum ether and ethyl acetate gradient, yielding the final high-purity product ready for characterization.
- Mix 3-oxo-nitrile compounds, 1,1-disubstituted hydrazine compounds, and a photocatalyst such as Rose Bengal in a polar organic solvent.
- Irradiate the reaction mixture with a light source having a wavelength between 200nm and 1000nm, preferably around 470nm.
- Maintain the reaction temperature between 25°C and 35°C for 6 to 24 hours, followed by purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photocatalytic methodology presents compelling economic and logistical benefits that extend beyond simple yield improvements. The elimination of expensive transition metal catalysts and hazardous oxidants translates directly into substantial cost savings in raw material procurement and waste disposal. Traditional methods often incur hidden costs associated with the removal of heavy metal residues to meet ppm-level specifications, a step that is rendered unnecessary or significantly simplified by this metal-free or low-metal organic photocatalytic approach. Additionally, the operation at ambient temperature drastically reduces energy consumption compared to thermal processes requiring reflux or heating mantles, contributing to a lower overall cost of goods sold (COGS) and a smaller carbon footprint for the manufacturing facility.
From a supply chain reliability perspective, the use of readily available starting materials such as benzoylacetonitrile derivatives and substituted hydrazines ensures a stable and continuous supply of precursors. The robustness of the reaction conditions means that the process is less susceptible to fluctuations in utility availability, such as steam pressure variations, since it relies primarily on electrical power for LED light sources. This decoupling from thermal utilities enhances the resilience of the production schedule, reducing lead times for high-purity pharmaceutical intermediates. Furthermore, the mild reaction environment minimizes the risk of runaway reactions or thermal hazards, simplifying safety compliance and insurance requirements. The scalability of the process is inherently supported by the modular nature of photoreactors, allowing for seamless transition from gram-scale discovery to kilogram-scale commercial production without the need for extensive re-engineering of the process parameters.
Environmental compliance and sustainability are increasingly critical factors in vendor selection, and this technology aligns perfectly with green chemistry principles. By utilizing visible light as the primary energy input and avoiding toxic reagents, the process generates significantly less hazardous waste, easing the burden on wastewater treatment systems. The high purity of the crude product, often reaching 99.9 percent before final purification, reduces the solvent load required for recrystallization or chromatography, further enhancing the environmental profile. This combination of operational safety, cost efficiency, and environmental stewardship makes the photocatalytic synthesis of guanylhydrazone compounds an attractive option for long-term supply agreements. It offers a sustainable solution for the commercial scale-up of complex heterocyclic precursors, ensuring that supply chains remain agile and compliant with evolving global regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic synthesis technology. These insights are derived directly from the experimental data and claims presented in patent CN112812040B, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into existing manufacturing workflows.
Q: What are the key advantages of this photocatalytic method over traditional synthesis?
A: Unlike conventional methods requiring harsh oxidants, strong bases, or high temperatures, this patent describes a mild process using visible light and organic photocatalysts at room temperature, achieving purities up to 99.9%.
Q: Which photocatalysts are suitable for this guanylhydrazone synthesis?
A: The patent specifies several effective photocatalysts including tris(2,2'-bipyridine)ruthenium dichloride, tris(2-phenylpyridine)iridium, Eosin B, Eosin Y, and notably Rose Bengal.
Q: Is this process scalable for commercial production of pharmaceutical intermediates?
A: Yes, the reaction operates under mild conditions (25-35°C) without expensive transition metals or high-pressure equipment, making it highly suitable for safe and efficient commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Guanylhydrazone Supplier
The technological advancements described in CN112812040B highlight the immense potential of photocatalytic synthesis in modernizing the production of critical pharmaceutical intermediates. At NINGBO INNO PHARMCHEM, we recognize the value of such innovative pathways and have positioned ourselves as a leader in translating cutting-edge academic and patent research into viable commercial solutions. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory methods are successfully adapted for industrial throughput. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of guanylhydrazone derivatives meets the highest quality standards required by global regulatory bodies.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this efficient synthesis route for their drug development programs. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to reach out to request specific COA data for our available intermediates and to discuss route feasibility assessments for your target molecules. Together, we can accelerate the delivery of high-quality chemical building blocks, driving innovation and efficiency in the pharmaceutical supply chain.
