Revolutionizing BAHPFP Production: A Safer, Scalable Route for High-Performance Polyimide Monomers
Revolutionizing BAHPFP Production: A Safer, Scalable Route for High-Performance Polyimide Monomers
The landscape of electronic material manufacturing is constantly evolving, driven by the need for higher performance polymers and safer, more sustainable production methodologies. A pivotal advancement in this sector is detailed in patent CN112707827B, which discloses a novel synthesis method for 2,2-bis(3-amino-4-hydroxyphenyl) hexafluoropropane (BAHPFP). This compound serves as a critical monomer for polyimides, which are indispensable in the fabrication of heat-resistant polymers and advanced electronic components such as computer chips. The patented technology represents a paradigm shift away from traditional, hazardous synthetic pathways, offering a streamlined process that begins with the acetylation of o-aminophenol, followed by condensation with hexafluoroacetone trihydrate, and concludes with a deacetylation step. For R&D directors and procurement specialists alike, this innovation signals a move towards processes that are not only chemically elegant but also commercially robust, eliminating the need for high-risk catalytic hydrogenation and environmentally burdensome nitration reactions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of BAHPFP has been plagued by significant safety and environmental challenges inherent to the prevailing synthetic routes. The dominant methodology involves the catalytic hydrogenation of 2,2-bis(3-nitro-4-hydroxyphenyl) hexafluoropropane, a process that necessitates the use of expensive noble metal catalysts and operates under high-risk conditions associated with hydrogen gas handling. Furthermore, the precursor synthesis often relies on nitration reactions, which are notoriously dangerous and generate substantial quantities of waste acid, imposing heavy burdens on wastewater treatment facilities and escalating production costs. Alternative routes, such as the halogen benzene method, suffer from excessive reaction steps and suboptimal yields, with some variations reporting yields as low as 70.7% across three steps. These inefficiencies create bottlenecks in the supply chain, leading to volatility in the availability of high-purity electronic chemical intermediates and complicating the cost structures for downstream polymer manufacturers.
The Novel Approach
In stark contrast to these legacy methods, the technology outlined in CN112707827B introduces a fundamentally safer and more efficient pathway that bypasses both nitration and hydrogenation entirely. By utilizing o-aminophenol as the starting material and employing a strategic acetylation protection group, the process enables a direct condensation with hexafluoroacetone trihydrate. This approach not only mitigates the safety risks associated with high-pressure hydrogenation but also drastically reduces the environmental footprint by avoiding the generation of nitration waste acids. The result is a synthesis route that maintains yields comparable to existing methods while offering superior operational safety and environmental friendliness, making it exceptionally well-suited for the rigorous demands of industrial mass production. This shift allows reliable electronic chemical suppliers to offer more consistent quality and pricing stability to their clients.
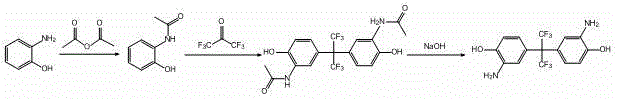
Mechanistic Insights into Acetylation-Protection and Condensation Strategy
The core chemical ingenuity of this patent lies in the strategic use of the acetyl group to modulate the reactivity of the amino functionality during the critical condensation step. In the first stage, o-aminophenol undergoes acetylation, typically using acetic anhydride in an ethyl acetate solvent at mild temperatures ranging from 15-30°C. This transformation converts the highly nucleophilic amine into an acetamide, which serves a dual purpose: it prevents unwanted side reactions at the nitrogen center during the subsequent acidic condensation and directs the electrophilic attack of the hexafluoroacetone species to the desired position on the aromatic ring. The condensation reaction itself is conducted in the presence of hydrofluoric acid, utilizing hexafluoroacetone trihydrate rather than the gaseous form, which significantly simplifies handling and dosing precision. Operating at temperatures between 100-110°C and pressures of 0.5-1.0 MPa, this step efficiently constructs the hexafluoroisopropyl bridge, a structural motif essential for the thermal stability and dielectric properties of the resulting polyimide.
Following the formation of the bis-acetamido intermediate, the final deacetylation step restores the free amine groups required for polymerization. The patent elucidates two viable pathways for this deprotection: alkaline deacetylation using sodium hydroxide in an ethanol-water mixture at 50-80°C, or acidic deacetylation using hydrochloric acid in 1,4-dioxane at 90-100°C. The alkaline route, in particular, has demonstrated exceptional efficiency, with experimental data showing yields reaching 93.7%. This flexibility in deprotection conditions allows process engineers to optimize the workflow based on available infrastructure and waste management capabilities. By carefully controlling the pH during the workup, specifically adjusting to a range of 7.5 ± 0.1, the process ensures the precipitation of high-purity product while minimizing the retention of inorganic salts, thereby addressing the critical purity specifications demanded by the semiconductor and advanced materials industries.
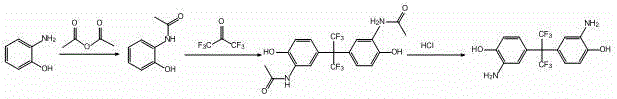
How to Synthesize 2,2-bis(3-amino-4-hydroxyphenyl) hexafluoropropane Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and purity, particularly during the exothermic acetylation and the high-pressure condensation phases. The process is designed to be scalable, moving seamlessly from laboratory validation to commercial production without the need for specialized high-pressure hydrogenation equipment. Operators must adhere to strict temperature protocols, such as maintaining the acetylation reaction between 20-25°C after the initial addition of reagents, and ensuring the condensation phase reaches the optimal 105°C to drive the reaction to completion. The detailed standardized synthesis steps, including specific molar ratios and workup procedures, are outlined below to guide technical teams in replicating this high-efficiency protocol.
- Perform acetylation of o-aminophenol using acetic anhydride in ethyl acetate at 15-30°C to form o-acetaminophenol.
- Condense o-acetaminophenol with hexafluoroacetone trihydrate in hydrofluoric acid at 100-110°C and 0.5-1.0 MPa pressure.
- Execute deacetylation using either alkaline conditions (NaOH/Ethanol/Water) at 50-80°C or acidic conditions (HCl/Dioxane) at 90-100°C to yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route translates into tangible strategic benefits that extend beyond simple unit cost metrics. By eliminating the dependency on noble metal catalysts required for hydrogenation, the process removes a significant variable cost component that is subject to global market fluctuations. Furthermore, the avoidance of nitration reactions means that facilities do not need to invest in extensive waste acid treatment infrastructure, leading to substantial reductions in environmental compliance costs and potential regulatory liabilities. This streamlined approach enhances the overall resilience of the supply chain, as it relies on readily available starting materials like o-aminophenol and avoids the logistical complexities associated with transporting and storing hazardous gases or managing high-risk hydrogenation units.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts and the reduction in waste treatment requirements directly contribute to a leaner cost structure. Without the need for high-pressure hydrogenation reactors, capital expenditure for new production lines is significantly lowered, and maintenance costs for existing infrastructure are reduced. The use of hexafluoroacetone trihydrate instead of gas further optimizes material handling costs, ensuring that cost reduction in polyimide monomer manufacturing is achieved through fundamental process simplification rather than mere economies of scale.
- Enhanced Supply Chain Reliability: By removing high-risk reaction steps that are prone to safety shutdowns or regulatory scrutiny, the continuity of supply is markedly improved. The process utilizes common solvents like ethyl acetate and ethanol, which are widely available in the global chemical market, reducing the risk of raw material shortages. This reliability is crucial for downstream manufacturers of electronic materials who require consistent, uninterrupted access to high-purity intermediates to maintain their own production schedules and meet delivery commitments to end-users.
- Scalability and Environmental Compliance: The synthetic route is inherently designed for industrial mass production, with reaction conditions that are easily manageable in standard stainless steel or Hastelloy reactors. The absence of nitration waste streams simplifies the environmental permitting process and reduces the long-term liability associated with hazardous waste disposal. This alignment with green chemistry principles not only future-proofs the manufacturing operation against tightening environmental regulations but also enhances the corporate sustainability profile, a factor of increasing importance to global stakeholders and investors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is this new synthesis route safer than conventional methods?
A: Conventional methods rely on catalytic hydrogenation and nitration, which involve high-risk reactions, expensive noble metal catalysts, and the generation of large amounts of waste acid. This novel route eliminates both nitration and hydrogenation steps, significantly enhancing operational safety and environmental compliance.
Q: What are the yield advantages of the acetylation-condensation-deacetylation pathway?
A: The patent demonstrates that this route achieves yields basically equivalent to existing methods while avoiding the low yields associated with the nitration of bisphenol AF or the multi-step halogen benzene method. Specifically, the acetylation step yields up to 97.3%, and the final deacetylation can reach 93.7% under optimized alkaline conditions.
Q: Is this process suitable for industrial scale-up of complex fluorinated intermediates?
A: Yes, the process is designed for industrial mass production. It utilizes readily available starting materials like o-aminophenol and avoids the handling of hazardous hexafluoroacetone gas directly by using the trihydrate form, simplifying the engineering controls required for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,2-bis(3-amino-4-hydroxyphenyl) hexafluoropropane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance monomers like BAHPFP play in the advancement of next-generation electronic materials. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of materials that meet stringent purity specifications. Our rigorous QC labs are equipped to verify the structural integrity and purity of every batch, guaranteeing that the fluorinated intermediates we deliver are perfectly suited for the demanding requirements of polyimide synthesis and semiconductor applications.
We invite you to collaborate with us to leverage this innovative synthesis technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this safer, more efficient route can optimize your bottom line. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to jointly engineer a supply solution that drives value and reliability for your organization.
