Advanced Biocatalytic Route for Methyl (S)-(+)-Mandelate: Scaling High-Purity Chiral Intermediates
The pharmaceutical and fine chemical industries are increasingly pivoting towards sustainable, high-precision manufacturing technologies to meet the rigorous demands of modern drug development. A prime example of this technological evolution is found in patent CN102719497B, which discloses a highly efficient method for preparing methyl (S)-(+)-mandelate through microbial transformation. This chiral intermediate is critical for the synthesis of various bioactive compounds, including vasodilators and ophthalmic agents. The patent outlines a sophisticated process utilizing Saccharomyces cerevisiae CGMCC No.2230, immobilized within a protective matrix, to perform asymmetric reduction in a dibutyl phthalate solvent system. This approach represents a significant leap forward from traditional chemical synthesis, offering a pathway to achieve exceptional optical purity and molar conversion rates under mild, environmentally benign conditions. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the nuances of this biocatalytic route is essential for optimizing supply chains and reducing the environmental footprint of chiral manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of chiral mandelic acid derivatives has relied heavily on chemical asymmetric reduction or resolution of racemates, processes that are often fraught with significant economic and technical inefficiencies. Chemical methods typically require expensive chiral catalysts, such as transition metal complexes with chiral ligands, which not only drive up raw material costs but also introduce the risk of heavy metal contamination in the final API. Furthermore, these reactions often necessitate harsh conditions, including extreme temperatures or pressures, and require complex downstream processing to remove toxic metal residues to meet regulatory standards. The resolution of racemates, another common strategy, is inherently inefficient with a maximum theoretical yield of only 50%, leading to substantial waste generation and increased disposal costs. These factors collectively contribute to a high cost of goods sold (COGS) and extended lead times, creating bottlenecks for companies aiming for cost reduction in chiral drug manufacturing.
The Novel Approach
In stark contrast, the biocatalytic method detailed in the patent leverages the innate stereoselectivity of whole-cell enzymes to achieve near-perfect chirality without the need for precious metal catalysts. By employing immobilized Saccharomyces cerevisiae cells, the process creates a robust micro-environment that protects the biocatalyst from the potentially denaturing effects of the organic substrate. The use of dibutyl phthalate as a reaction medium is particularly innovative; it serves as a reservoir that solubilizes the hydrophobic substrate, methyl benzoylformate, while maintaining a compatible interface for the aqueous-based enzymatic activity within the cells. This biphasic-like system mitigates substrate inhibition, allowing for higher substrate loading and improved volumetric productivity. Consequently, this novel approach delivers high-purity methyl (S)-(+)-mandelate with simplified purification steps, positioning it as a superior alternative for the commercial scale-up of complex chiral intermediates.
Mechanistic Insights into Immobilized Whole-Cell Biocatalysis
The core of this technology lies in the intricate interplay between the microbial carbonyl reductase enzymes and the immobilization matrix. The Saccharomyces cerevisiae strain CGMCC No.2230 contains specific reductases that selectively reduce the pro-chiral ketone group of methyl benzoylformate to the corresponding (S)-alcohol with high fidelity. When these cells are entrapped within calcium alginate beads, the diffusion of substrates and cofactors is carefully regulated, preventing cell lysis and enzyme leakage. The regeneration of necessary cofactors, such as NADPH, occurs naturally within the metabolic network of the living yeast cells, eliminating the need for external cofactor addition which is a common cost driver in isolated enzyme systems. This self-sustaining catalytic cycle ensures that the reduction proceeds efficiently over extended periods, maintaining the structural integrity of the biocatalyst throughout the reaction duration.
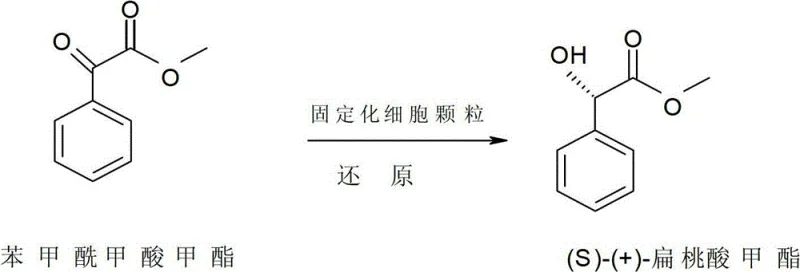
Furthermore, the choice of solvent and immobilization parameters critically influences the mass transfer kinetics and overall reaction rate. The patent data indicates that the diameter of the immobilized particles and the concentration of sodium alginate are key variables; optimal performance is achieved with 2mm particles and a 2% alginate concentration. This specific configuration maximizes the surface-area-to-volume ratio, facilitating rapid diffusion of the substrate into the bead while providing sufficient mechanical strength for repeated handling. The dibutyl phthalate solvent plays a dual role: it dissolves the organic substrate effectively and extracts the product, shifting the equilibrium towards product formation and reducing product inhibition on the enzyme active sites. This mechanistic understanding allows for precise process control, ensuring consistent quality and enabling the reducing lead time for high-purity pharmaceutical intermediates through predictable reaction kinetics.
How to Synthesize Methyl (S)-(+)-Mandelate Efficiently
Implementing this biocatalytic route requires strict adherence to the fermentation and immobilization protocols to ensure maximum catalyst activity. The process begins with the cultivation of the yeast strain to a specific cell density, followed by entrapment in alginate and a crucial proliferation step that enhances the enzymatic potential of the immobilized beads. The subsequent transformation in dibutyl phthalate must be monitored to balance substrate concentration against potential inhibition effects. While the general workflow is straightforward, the specific operational parameters regarding temperature, agitation, and reaction time are critical for achieving the reported >99.0% conversion and ee values. Detailed standardized synthesis steps are provided in the guide below to assist process engineers in replicating these results.
- Ferment Saccharomyces cerevisiae CGMCC No.2230 to obtain enzyme-containing cells, then immobilize them using sodium alginate and calcium chloride to form 2mm particles.
- Proliferate the immobilized cell particles in fermentation medium for 72 hours to enhance catalytic activity before use.
- Conduct the asymmetric reduction of methyl benzoylformate in dibutyl phthalate at 30°C for 72 hours, followed by filtration and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this biocatalytic methodology offers profound strategic advantages beyond mere technical performance. The elimination of expensive chiral metal catalysts and the ability to operate under ambient pressure and moderate temperatures significantly lowers the barrier to entry for production, reducing both capital expenditure (CAPEX) on specialized equipment and operational expenditure (OPEX) on energy and raw materials. The robustness of the immobilized catalyst means that a single batch of biocatalyst can be utilized for multiple production cycles, dramatically decreasing the frequency of catalyst procurement and preparation. This stability translates directly into a more predictable and resilient supply chain, minimizing the risks associated with raw material volatility and ensuring consistent availability of critical intermediates for downstream drug synthesis.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the substitution of costly chemical catalysts with renewable, fermentable biomass. Since the biocatalyst can be reused multiple times without significant loss of activity, the effective cost per kilogram of catalyst consumed is drastically reduced compared to single-use chemical reagents. Additionally, the mild reaction conditions eliminate the need for energy-intensive heating or cooling systems, further lowering utility costs. The simplified downstream processing, which avoids complex metal scavenging steps, reduces solvent usage and waste treatment expenses, contributing to substantial overall cost savings in the manufacturing budget.
- Enhanced Supply Chain Reliability: Relying on a biological system derived from a stable, preserved strain ensures a consistent and reproducible source of catalytic activity, unlike chemical catalysts which may suffer from batch-to-batch variability or supply shortages of precious metals. The immobilization technique extends the shelf-life and operational life of the catalyst, allowing manufacturers to maintain strategic stockpiles of biocatalyst beads that are ready for immediate deployment. This readiness significantly shortens the production lead time, enabling faster response to market demands and reducing the inventory holding costs for finished intermediates.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from laboratory flask experiments to industrial-scale bioreactors without fundamental changes to the reaction chemistry. The use of non-toxic, biodegradable yeast and the avoidance of heavy metals align perfectly with increasingly stringent global environmental regulations, such as REACH and EPA guidelines. This green chemistry profile simplifies the permitting process for new manufacturing facilities and enhances the corporate sustainability image, which is a growing requirement for partnerships with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this microbial transformation technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on performance metrics and operational feasibility. Understanding these details is crucial for stakeholders evaluating the integration of this route into their existing production portfolios.
Q: What is the optical purity of the methyl (S)-(+)-mandelate produced by this method?
A: The method described in patent CN102719497B achieves an enantiomeric excess (ee%) greater than 99.0%, ensuring high stereochemical integrity suitable for chiral drug synthesis.
Q: Can the biocatalyst be reused to lower production costs?
A: Yes, the immobilized cell particles can be recovered by simple filtration and reused for up to 12 cycles while maintaining significant catalytic activity, drastically reducing catalyst consumption costs.
Q: Why is dibutyl phthalate used as the reaction solvent?
A: Dibutyl phthalate acts as a biocompatible organic phase that dissolves high concentrations of substrate and product, reducing inhibition effects on the microbial cells and improving overall transformation efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methyl (S)-(+)-Mandelate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality chiral intermediates in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of methyl (S)-(+)-mandelate meets the highest international standards for optical purity and chemical identity. Our commitment to technical excellence allows us to deliver products that support your R&D timelines and commercial manufacturing goals with unwavering reliability.
We invite you to collaborate with us to optimize your supply chain for chiral building blocks. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced biocatalytic capabilities can drive value and efficiency in your production processes.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
