Advanced Biocatalytic Synthesis of Pregabalin Intermediates for Commercial Scale Manufacturing
Advanced Biocatalytic Synthesis of Pregabalin Intermediates for Commercial Scale Manufacturing
The pharmaceutical industry is constantly seeking more efficient and sustainable pathways for the production of critical active pharmaceutical ingredients (APIs) and their precursors. A significant breakthrough in this domain is documented in Chinese patent CN102382785A, which discloses a novel microbial transformation method for preparing (S)-2-carboxyethyl-3-cyano-5-methylhexanoic acid. This compound serves as a pivotal chiral intermediate in the synthesis of Pregabalin, a widely prescribed medication for neuropathic pain and epilepsy. The patent introduces a newly isolated strain, Morganella morganii ZJB09203, which offers superior stability and stereoselectivity compared to previously available biocatalysts. By leveraging this specific microbial strain, manufacturers can achieve high optical purity exceeding 95% under mild aqueous conditions, representing a substantial advancement over traditional chemical resolution methods that often rely on toxic solvents and expensive chiral auxiliaries.
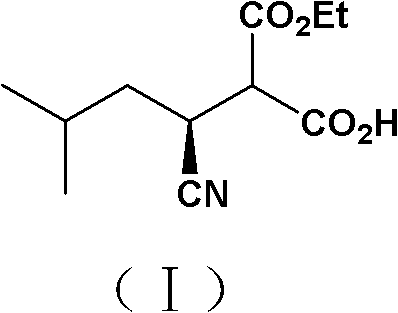
This technological development is particularly relevant for global supply chains aiming to reduce their environmental footprint while maintaining rigorous quality standards. The ability to utilize a robust bacterial strain that tolerates high substrate concentrations allows for more concentrated reaction mixtures, thereby improving volumetric productivity. For procurement specialists and R&D directors alike, understanding the nuances of this biocatalytic route is essential for evaluating potential suppliers who can offer cost-effective and reliable sources of high-purity pharmaceutical intermediates. The shift towards such biological processes aligns with broader industry trends favoring green chemistry principles without compromising on the stringent purity specifications required for regulatory approval in major markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral intermediates like (S)-2-carboxyethyl-3-cyano-5-methylhexanoic acid has relied heavily on chemical resolution or early-generation enzymatic processes that present significant operational challenges. Traditional chemical routes often necessitate the use of large volumes of organic solvents and stoichiometric amounts of chiral resolving agents, such as mandelic acid, which drives up raw material costs and generates substantial hazardous waste. Furthermore, earlier enzymatic approaches utilizing commercial lipases, while an improvement, still faced limitations regarding enzyme cost and the difficulty of recycling unreacted substrates. Some prior art methods involving Pseudomonas species required complex downstream processing to handle racemization issues, leading to lower overall yields and increased production complexity. These inefficiencies create bottlenecks in the supply chain, resulting in higher lead times and volatility in pricing for the final API manufacturers who depend on these critical building blocks.
The Novel Approach
In contrast, the methodology outlined in patent CN102382785A utilizes the novel Morganella morganii ZJB09203 strain to catalyze the hydrolysis of ethyl 2-carboxyethyl-3-cyano-5-methylhexanoate directly in an aqueous environment. This approach eliminates the need for expensive organic solvents and reduces the reliance on costly commercial enzyme preparations by using whole-cell biocatalysts that can be produced in-house via fermentation. The process operates effectively within a broad temperature range of 20 to 45°C and a pH window of 6.5 to 9.0, offering remarkable flexibility for industrial scale-up. By achieving high stereoselectivity with strict control over the chiral center, this method minimizes the formation of unwanted enantiomers, simplifying the purification steps and enhancing the overall yield of the desired (S)-isomer. This represents a paradigm shift towards more sustainable and economically viable manufacturing practices for high-value pharmaceutical intermediates.
Mechanistic Insights into Biocatalytic Hydrolysis
The core of this innovation lies in the specific enzymatic activity inherent to the Morganella morganii ZJB09203 strain, which facilitates the highly selective hydrolysis of the ester bond in the substrate. The biocatalytic mechanism involves the interaction of the enzyme's active site with the pro-chiral substrate, preferentially recognizing and cleaving the ester group of one enantiomer while leaving the other largely untouched or converting it at a significantly slower rate. This kinetic resolution is driven by the precise three-dimensional arrangement of amino acids within the enzyme, which creates a chiral environment that discriminates between the (R) and (S) forms of the molecule. The patent data indicates that this selectivity is maintained even at high substrate loadings, suggesting that the enzyme possesses a robust binding affinity and resistance to substrate inhibition, which is critical for maintaining high reaction rates in industrial reactors.
Furthermore, the control of impurities is intrinsically linked to the high stereoselectivity of the biological system. In chemical synthesis, side reactions such as non-selective hydrolysis or racemization can lead to complex impurity profiles that are difficult and costly to remove. However, the biological specificity of the ZJB09203 strain ensures that the primary reaction pathway leads almost exclusively to the desired (S)-2-carboxyethyl-3-cyano-5-methylhexanoic acid with an enantiomeric excess (ee) value consistently above 95%. The subsequent decarboxylation step to form the next intermediate, (S)-3-cyano-5-methylhexanoic acid ethyl ester, proceeds cleanly from this high-purity precursor. This inherent purity reduces the burden on downstream purification units, such as chromatography or crystallization, thereby lowering energy consumption and solvent usage in the overall manufacturing process.
How to Synthesize (S)-2-carboxyethyl-3-cyano-5-methylhexanoic acid Efficiently
Implementing this biocatalytic route requires a systematic approach to fermentation and bioconversion to maximize yield and optical purity. The process begins with the cultivation of the Morganella morganii ZJB09203 strain under optimized conditions to generate a high-density cell suspension capable of efficient catalysis. Operators must carefully monitor parameters such as pH and temperature during the conversion phase to ensure the enzyme remains active and selective throughout the reaction duration. The following guide outlines the standardized synthetic steps derived from the patent examples, providing a clear roadmap for technical teams looking to adopt this methodology for pilot or commercial production.
- Prepare the enzyme source by cultivating Morganella morganii ZJB09203 in a fermentation medium containing glucose, yeast powder, and peptone at 20-40°C for 48-72 hours to obtain a cell suspension.
- Suspend the wet bacterial cells in pure water and add the substrate ethyl 2-carboxyethyl-3-cyano-5-methylhexanoate to achieve a concentration of 10-300 g/L.
- Maintain the reaction at 20-45°C and pH 6.5-9.0 for 4-48 hours, then separate the product via centrifugation and purification to achieve >95% optical purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this biocatalytic technology offers tangible strategic benefits that extend beyond simple technical feasibility. The transition from solvent-intensive chemical processes to aqueous biocatalysis fundamentally alters the cost structure of manufacturing these intermediates. By removing the need for vast quantities of organic solvents and expensive chiral resolving agents, the variable costs associated with raw materials are significantly reduced. Additionally, the use of a stable, self-replicating bacterial strain means that the catalyst itself can be produced at a fraction of the cost of purchasing commercial immobilized enzymes, leading to substantial long-term savings in operating expenses without compromising on quality or consistency.
- Cost Reduction in Manufacturing: The elimination of hazardous organic solvents and the reduction in downstream purification requirements directly translate to lower waste disposal costs and reduced energy consumption for solvent recovery. Since the process utilizes water as the primary medium and relies on a robust bacterial strain that can be cultured using standard fermentation nutrients, the input costs are inherently lower than those associated with precious metal catalysts or specialized chemical reagents. This economic efficiency allows suppliers to offer more competitive pricing structures while maintaining healthy margins, providing a buffer against raw material price fluctuations in the global market.
- Enhanced Supply Chain Reliability: The stability of the Morganella morganii ZJB09203 strain ensures consistent batch-to-batch performance, which is crucial for maintaining uninterrupted supply lines to API manufacturers. Unlike chemical processes that might suffer from variability due to reagent quality or environmental conditions, this biological system is highly reproducible when controlled within the specified pH and temperature ranges. This reliability reduces the risk of production delays and quality deviations, ensuring that procurement teams can secure long-term contracts with confidence knowing that the supplier has a resilient and scalable production platform capable of meeting demand surges.
- Scalability and Environmental Compliance: Scaling biocatalytic processes is often more straightforward than scaling complex chemical syntheses involving hazardous reagents, as the reaction conditions are mild and safe. The aqueous nature of the reaction mixture simplifies containment and handling, reducing the regulatory burden associated with volatile organic compound (VOC) emissions. This alignment with green chemistry principles not only facilitates easier permitting and compliance with increasingly strict environmental regulations but also enhances the corporate social responsibility profile of the supply chain, appealing to end-users who prioritize sustainability in their sourcing decisions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biocatalytic process. These insights are derived directly from the experimental data and claims presented in the patent documentation, offering clarity on the capabilities and limitations of the technology. Understanding these details is vital for stakeholders evaluating the feasibility of integrating this intermediate into their existing production workflows or sourcing strategies.
Q: What is the optical purity achieved by the Morganella morganii ZJB09203 strain?
A: According to patent CN102382785A, the new strain demonstrates strict stereoselectivity, achieving an optical purity (ee value) of greater than 95% when the conversion rate approaches 50%.
Q: How does this biocatalytic method compare to traditional chemical synthesis?
A: Unlike traditional chemical methods that often require harsh conditions and expensive chiral resolving agents, this biocatalytic process operates in water at mild temperatures (20-45°C), significantly reducing organic solvent usage and environmental impact.
Q: Is the Morganella morganii ZJB09203 strain stable for industrial production?
A: Yes, the patent highlights that the ZJB09203 strain exhibits excellent stability and can tolerate high concentrations of both substrate and product, making it highly suitable for scalable industrial fermentation processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-2-carboxyethyl-3-cyano-5-methylhexanoic acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the pharmaceutical value chain. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global API manufacturers. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify identity and optical purity. Our capability to implement advanced biocatalytic technologies like the one described in CN102382785A positions us as a forward-thinking partner capable of driving innovation and efficiency in your supply chain.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to our biocatalytically produced intermediates. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete technical evidence and our proven track record of excellence in the fine chemical industry.
