Advanced Mechanochemical Route for High-Purity N,N'-Disubstituted Benzimidazolium Derivatives Manufacturing
The landscape of fine chemical manufacturing is undergoing a paradigm shift towards greener, more efficient methodologies, as exemplified by the groundbreaking technology disclosed in patent CN111635363A. This intellectual property introduces a robust mechanochemical strategy for the synthesis of N,N'-disubstituted benzimidazolium derivatives, a class of compounds that serves as critical precursors for N-heterocyclic carbene (NHC) ligands and ionic liquids. Traditionally, the production of these valuable intermediates has been plagued by the heavy reliance on volatile organic solvents and energy-intensive thermal processes. However, this novel approach leverages the power of mechanical force in a solvent-free environment to drive chemical transformations at room temperature. For R&D directors and process chemists seeking to modernize their synthetic portfolios, this patent offers a compelling alternative that aligns with the principles of green chemistry while delivering exceptional reaction efficiency. The ability to generate high-purity products without the burden of solvent recovery represents a significant technological leap forward for the industry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N,N'-disubstituted benzimidazolium derivatives has relied heavily on traditional solution-phase chemistry, which presents numerous operational and environmental challenges for large-scale manufacturing. Conventional protocols typically necessitate prolonged heating under reflux conditions, often requiring reaction times extending from 8 hours to as long as 50 hours depending on the specific substrate reactivity. These extended thermal exposures not only consume substantial amounts of energy but also increase the risk of thermal degradation for sensitive functional groups. Furthermore, the reliance on solvents such as acetonitrile, ethanol, or dioxane creates a significant downstream burden regarding solvent recovery, waste disposal, and environmental compliance. The purification processes associated with these solution methods are frequently cumbersome, involving multiple extraction and chromatography steps to remove unreacted starting materials and side products, which ultimately erodes the overall process yield and economic viability.
The Novel Approach
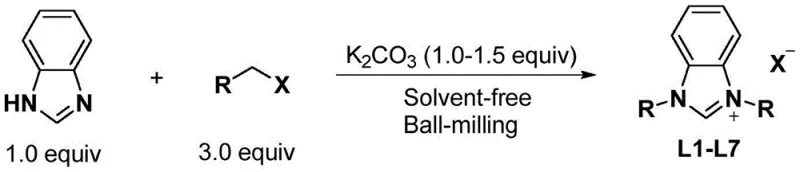
In stark contrast to these legacy methods, the mechanochemical synthesis described in the patent utilizes a planetary ball mill to facilitate the reaction through direct mechanical energy transfer rather than thermal activation. By grinding benzimidazole, alkyl or aryl substituents, and potassium carbonate together at a frequency of 37.5 Hz, the reaction reaches completion in merely 2 hours at room temperature. This drastic reduction in processing time from days to hours significantly enhances throughput capacity without compromising product quality. The absence of bulk solvents fundamentally alters the reaction kinetics, often leading to unique selectivity profiles and higher conversion rates that are unattainable in solution. For procurement managers and supply chain heads, this translates to a streamlined workflow where the elimination of solvent handling reduces both raw material costs and the logistical complexity associated with hazardous chemical storage and transport.
Mechanistic Insights into Mechanochemical Alkylation
The core of this innovative process lies in the efficient utilization of mechanical energy to overcome the activation barrier for the nucleophilic substitution reaction. In the solid state, the intimate mixing of benzimidazole and the alkylating agent within the high-energy impact zones of the ball mill creates localized hot spots and defects in the crystal lattice. These mechanical forces facilitate the deprotonation of the benzimidazole nitrogen by the solid base, potassium carbonate, generating a reactive nucleophile in situ. Unlike solution chemistry where diffusion limits the collision frequency of reactants, the continuous fracturing and cold-welding of particles in the ball mill ensure constant exposure of fresh reactive surfaces. This dynamic environment promotes rapid alkylation at both nitrogen positions of the benzimidazole ring, leading to the formation of the disubstituted cationic species with remarkable speed and efficiency.
Furthermore, the mechanistic pathway inherently supports superior impurity control, which is a critical parameter for pharmaceutical intermediate suppliers. The stoichiometric use of potassium carbonate not only acts as the base but also captures the halide leaving group as an insoluble inorganic salt, such as potassium bromide. This byproduct remains in the solid phase and can be easily separated from the organic product through a simple suspension and filtration step using dichloromethane. This elegant separation mechanism avoids the formation of emulsions common in liquid-liquid extractions and eliminates the need for extensive aqueous workups. The resulting crude product is of such high purity that a simple wash with tetrahydrofuran is often sufficient to isolate the final target molecule, thereby minimizing material loss and maximizing the overall mass balance of the process.
How to Synthesize N,N'-disubstituted benzimidazolium derivatives Efficiently
Implementing this mechanochemical protocol requires precise control over milling parameters and reagent ratios to ensure reproducibility and optimal yield. The process begins with the careful weighing of benzimidazole, the specific alkyl or aryl halide substituent, and anhydrous potassium carbonate in a molar ratio of 1:3:1.5. These components are loaded into a stainless steel grinding jar equipped with stainless steel balls, which serve as the grinding media to transmit kinetic energy to the reactants. The simplicity of the setup belies the sophistication of the chemical transformation occurring within, where mechanical force replaces thermal energy to drive the reaction to completion. For detailed operational parameters and safety considerations regarding the specific equipment setup, please refer to the standardized synthesis guide provided below.
- Mix benzimidazole, alkyl or aryl halide substituent, and anhydrous K2CO3 in a molar ratio of 1: 3:1.5 within a steel grinding jar containing stainless steel balls.
- Seal the grinding jar and operate the planetary ball mill at a frequency of 37.5 Hz for 2 hours at room temperature to obtain the crude solid product.
- Suspend the crude solid in dichloromethane, filter to remove inorganic salts like KBr, evaporate the filtrate, and wash the residue with tetrahydrofuran to isolate the pure derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this solvent-free mechanochemical technology offers profound advantages that directly address the pain points of modern chemical supply chains. The elimination of organic solvents removes a major cost center associated with purchasing, storing, and disposing of volatile chemicals, while simultaneously mitigating regulatory risks related to VOC emissions. For procurement managers, this means a more stable cost structure that is less susceptible to fluctuations in solvent markets. Additionally, the dramatic reduction in reaction time from potentially several days to just two hours allows for significantly faster batch turnover, enabling manufacturers to respond more agilely to market demands and reduce inventory holding times. This efficiency gain is crucial for maintaining competitiveness in the fast-paced pharmaceutical and agrochemical sectors.
- Cost Reduction in Manufacturing: The transition to a solvent-free process fundamentally reshapes the cost equation by removing the need for expensive solvent recovery infrastructure and reducing energy consumption associated with heating and refluxing. Without the requirement for large volumes of organic solvents, the facility can operate with smaller reactor footprints and reduced ventilation requirements, leading to substantial capital expenditure savings. Moreover, the simplified workup procedure involving filtration and washing minimizes labor hours and consumable usage, further driving down the cost of goods sold. These cumulative efficiencies allow for a more competitive pricing strategy while maintaining healthy margins, making the production of high-value intermediates more economically sustainable in the long term.
- Enhanced Supply Chain Reliability: By relying on readily available solid reagents and eliminating the dependency on specific solvent grades, the supply chain becomes more resilient to disruptions. The robustness of the mechanochemical method ensures consistent product quality regardless of minor variations in ambient conditions, as the reaction is driven by mechanical energy rather than sensitive thermal equilibria. This reliability translates to predictable lead times and fewer batch failures, which is essential for maintaining uninterrupted supply to downstream customers. The ability to produce previously unreported compounds with high yields also expands the portfolio of available intermediates, providing customers with greater flexibility in their drug discovery and development pipelines.
- Scalability and Environmental Compliance: Scaling mechanochemical processes is increasingly feasible with modern industrial milling equipment, allowing for the transition from gram-scale optimization to multi-kilogram production without the linear increase in solvent waste typical of batch reactors. The inherent green nature of this technology aligns perfectly with stringent environmental regulations and corporate sustainability goals, reducing the carbon footprint of chemical manufacturing. The absence of solvent waste streams simplifies wastewater treatment and lowers the burden on environmental health and safety departments. This compliance advantage not only future-proofs the manufacturing process against tightening regulations but also enhances the brand reputation of companies committed to sustainable chemistry practices.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this mechanochemical synthesis route. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on how this method outperforms traditional solution-based techniques. Understanding these nuances is vital for technical teams evaluating the feasibility of adopting this technology for their specific production needs. The answers highlight the practical implications of solvent-free processing on yield, purity, and operational safety.
Q: What are the primary advantages of mechanochemical synthesis over traditional reflux methods for benzimidazolium salts?
A: The mechanochemical approach eliminates the need for organic solvents and high-temperature reflux, reducing reaction time from up to 50 hours in conventional methods to just 2 hours while maintaining high yields and purity.
Q: Can this solvent-free method be applied to sensitive substrates requiring mild conditions?
A: Yes, the process operates at room temperature without external heating, making it suitable for thermally sensitive substrates and preventing decomposition often seen in prolonged heating scenarios.
Q: How does the purification process differ in this green synthesis route?
A: Purification is simplified to a suspension in dichloromethane followed by filtration to remove inorganic byproducts like potassium bromide, avoiding complex chromatographic separations typically required in solution chemistry.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N,N'-disubstituted benzimidazolium derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the mechanochemical route described in CN111635363A for producing high-performance chemical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes can be successfully translated into robust industrial operations. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. We understand that consistency is key in the pharmaceutical supply chain, and our dedicated technical team works closely with clients to optimize every step of the manufacturing process for maximum efficiency and reliability.
We invite you to collaborate with us to leverage these cutting-edge synthetic technologies for your next project. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments for N,N'-disubstituted benzimidazolium derivatives or other complex intermediates. Let us help you accelerate your development timeline and achieve your commercial goals with our proven expertise in green chemistry and process optimization.
