Scalable Synthesis Of Apigenin Glycoside Intermediates For Commercial Pharmaceutical Manufacturing
The pharmaceutical industry is constantly seeking robust supply chains for bioactive natural products, particularly those with potent cardiovascular therapeutic potential. Patent CN101712668B discloses a groundbreaking synthetic methodology for producing apigenin-7-O-β-D-glucoside-4'-O-α-L-rhamnoside, a flavonoid compound demonstrating significant efficacy in treating ischemic diseases such as coronary heart disease and cerebral infarction. Unlike traditional extraction methods which suffer from extremely low recovery rates, this patent outlines a chemically defined pathway utilizing a novel intermediate, Formula E. This technical advancement addresses the critical bottleneck of raw material scarcity, enabling the transition from laboratory-scale curiosity to commercial-scale reality. By leveraging selective protection and deprotection strategies, the process ensures high purity and reproducibility, which are paramount for regulatory approval in the global pharmaceutical market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the sourcing of complex flavonoid glycosides like Formula A has relied heavily on botanical extraction, a method fraught with inefficiency and supply chain volatility. The background art highlights a stark reality where isolating mere milligrams of the active compound requires processing kilograms of raw plant biomass, specifically yielding only 165mg from 17kg of dried Ranunculus sieboldii. This abysmal efficiency not only drives up costs exponentially but also introduces significant variability in impurity profiles due to seasonal and geographical differences in plant growth. Furthermore, the reliance on agricultural sources creates a fragile supply chain susceptible to crop failures and environmental regulations, making it impossible to guarantee the consistent tonnage required for clinical trials and subsequent commercial launch. The purification burden is immense, often requiring extensive chromatography that further erodes yield and increases solvent waste, rendering the process economically unviable for large-scale API manufacturing.
The Novel Approach
The synthetic route detailed in the patent represents a paradigm shift towards total synthesis, bypassing the limitations of nature's low abundance. By constructing the molecule from commercially available apigenin (Formula G), the process allows for precise control over every functional group installation. The core innovation lies in the preparation and utilization of the Formula E intermediate, which features a specific acyl protecting group at the 5-position and a benzyl group at the 7-position. This strategic protection scheme facilitates the sequential glycosylation at the 4' and 7 positions with high regioselectivity. 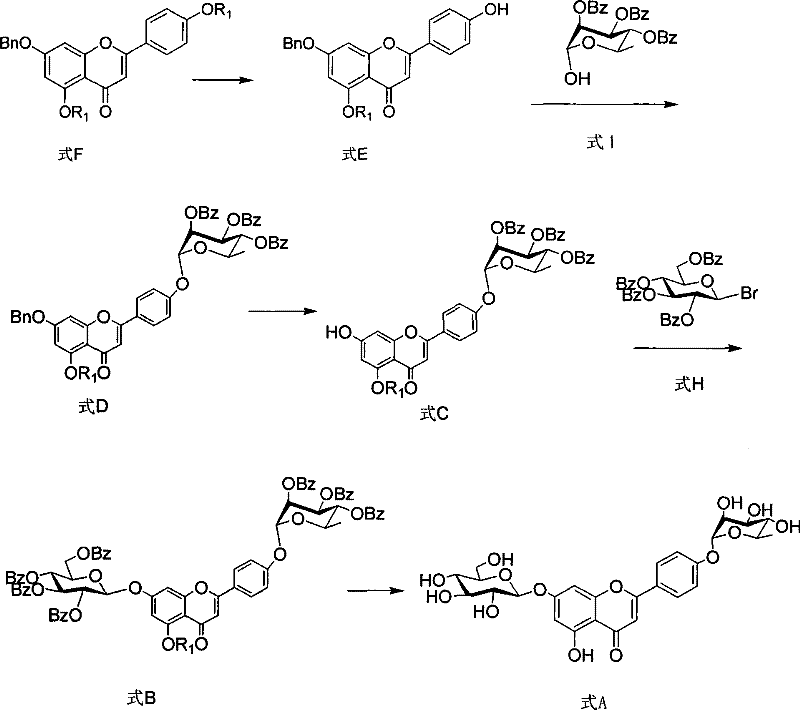 The workflow integrates modern organic transformations including Mitsunobu coupling and phase-transfer catalyzed glycosylation, ensuring that each step proceeds with predictable kinetics. This approach transforms the production of this valuable therapeutic agent into a reliable industrial process, decoupling supply from agricultural constraints and establishing a foundation for cost reduction in pharmaceutical intermediate manufacturing.
The workflow integrates modern organic transformations including Mitsunobu coupling and phase-transfer catalyzed glycosylation, ensuring that each step proceeds with predictable kinetics. This approach transforms the production of this valuable therapeutic agent into a reliable industrial process, decoupling supply from agricultural constraints and establishing a foundation for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Selective Deprotection and Glycosylation
The chemical elegance of this route is anchored in the precise manipulation of hydroxyl reactivity. The conversion of Formula F to the key intermediate Formula E involves a selective dehydroxylation where the 4'-hydroxyl group is liberated while the 5-acyl and 7-benzyl groups remain intact. This is achieved under mild alkaline conditions using reagents like potassium carbonate in a mixed solvent system of dichloromethane and methanol at temperatures ranging from -10°C to 30°C. The choice of the acyl group R1 is mechanistically critical; experimental data indicates that when R1 is a hexanoyl chain (-CO(CH2)4CH3), the intermediate exhibits superior solubility characteristics. This enhanced solubility prevents precipitation during reaction workups and improves mass transfer, directly correlating to the observed yield increase to 90.3% for this specific step. Such optimization minimizes side reactions and simplifies downstream purification, a crucial factor for maintaining high purity specifications in complex molecule synthesis.
Subsequent glycosylation steps employ rigorous stereochemical control mechanisms to ensure biological fidelity. The attachment of the rhamnose moiety utilizes a Mitsunobu reaction between Formula E and 2,3,4-tribenzoyloxyrhamnose (Formula I), mediated by triphenylphosphine and diethyl azodicarboxylate. This inversion-prone mechanism is carefully managed to secure the desired alpha-linkage. Following this, catalytic hydrogenation removes the 7-benzyl protecting group without disturbing the newly formed glycosidic bonds, a testament to the orthogonality of the protecting group strategy. The final glucose unit is installed via a nucleophilic substitution using 2,3,4,6-tetrabenzoyloxyglucose bromide (Formula H) under phase-transfer catalysis. 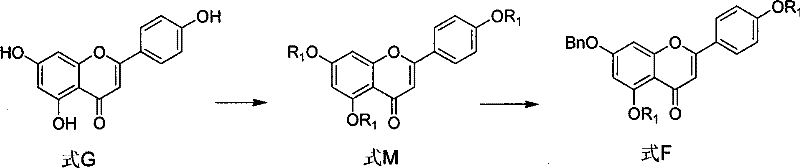 This sequence demonstrates a deep understanding of carbohydrate chemistry, ensuring that the final deprotection step yields the target Formula A with the correct anomeric configurations essential for its ischemic disease treatment activity.
This sequence demonstrates a deep understanding of carbohydrate chemistry, ensuring that the final deprotection step yields the target Formula A with the correct anomeric configurations essential for its ischemic disease treatment activity.
How to Synthesize Apigenin Glycoside Intermediate Efficiently
Executing this synthesis requires strict adherence to the optimized reaction parameters outlined in the patent to maximize yield and purity. The process begins with the preparation of the fully protected precursor Formula F, followed by the critical selective deprotection to generate Formula E. Operators must maintain precise temperature control during the alkaline treatment to prevent over-hydrolysis of the ester moieties. The subsequent glycosylation steps demand anhydrous conditions and careful stoichiometry of coupling reagents to avoid incomplete reactions. Detailed standard operating procedures regarding solvent ratios, such as the 1:1 volume ratio of dichloromethane to methanol, are essential for reproducibility. For the complete step-by-step laboratory protocol including exact reagent quantities and workup procedures, please refer to the standardized guide below.
- Perform selective acylation of apigenin (Formula G) using acyl chlorides and organic bases in aprotic solvents to generate Formula M.
- Execute benzylation of Formula M using benzyl chloride and inorganic bases to produce the protected intermediate Formula F.
- Conduct selective dehydroxylation of Formula F using alkaline reagents in mixed solvents to yield the key intermediate Formula E.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from extraction to this synthetic route offers profound strategic advantages beyond simple availability. The primary value driver is the stabilization of supply continuity; by synthesizing the compound from basic chemical feedstocks, the risk of raw material shortage due to agricultural factors is completely eliminated. This reliability is crucial for long-term product lifecycle management and ensures that clinical development timelines are not jeopardized by material scarcity. Furthermore, the synthetic pathway is designed with scalability in mind, utilizing common organic solvents and reagents that are readily available in bulk quantities globally. This reduces the complexity of sourcing specialized materials and mitigates the risk of single-supplier dependency for exotic plant extracts.
- Cost Reduction in Manufacturing: The economic model of this synthesis is driven by the significant improvement in overall yield compared to extraction. While plant extraction yields are negligible, the synthetic route achieves a total yield of up to 19.2% when the optimal hexanoyl protecting group is used. This drastic increase in material throughput per batch means that fixed costs such as labor, energy, and equipment depreciation are amortized over a much larger quantity of product. Additionally, the use of standard chemical reagents instead of expensive enzymatic or biotechnological processes keeps variable costs low. The simplified purification steps resulting from improved intermediate solubility further reduce solvent consumption and waste disposal costs, contributing to substantial cost savings in API manufacturing without compromising quality.
- Enhanced Supply Chain Reliability: Dependence on botanical sources introduces uncontrollable variables such as weather patterns, pest infestations, and geopolitical trade restrictions on herbal materials. By shifting to a fully synthetic supply chain, manufacturers gain full control over production schedules and inventory levels. The ability to produce the intermediate Formula E and subsequent derivatives on demand allows for a just-in-time manufacturing strategy, reducing the need for massive warehousing of unstable raw plant materials. This agility enhances the resilience of the supply chain against external shocks, ensuring a steady flow of high-purity pharmaceutical intermediates to downstream formulation partners.
- Scalability and Environmental Compliance: The synthetic process is inherently more scalable than extraction, which is limited by the finite biomass available. Chemical reactors can be scaled from liters to cubic meters with predictable outcomes, facilitating the transition from pilot plant to commercial production. Moreover, the process avoids the massive solid waste generation associated with processing tons of plant matter. The use of recyclable organic solvents and the absence of heavy metal catalysts in key steps align with modern green chemistry principles. This reduces the environmental footprint of the manufacturing process, simplifying regulatory compliance regarding waste discharge and supporting corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. These answers are derived directly from the experimental data and embodiments provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these nuances is vital for assessing the feasibility of adopting this route for your specific production needs.
Q: Why is the synthetic route preferred over plant extraction for Formula A?
A: Plant extraction yields are critically low, obtaining only 165mg from 17kg of dry plant material, making it unviable for industrial scale. The synthetic route described in CN101712668B offers a controllable, scalable process with significantly higher total yields and consistent purity profiles essential for regulatory compliance.
Q: What is the optimal protecting group strategy for this synthesis?
A: The patent identifies the hexanoyl group (R1 = -CO(CH2)4CH3) as the optimal protecting group. This specific chain length provides the best balance of solubility and reactivity, resulting in the highest overall yield of 19.2% compared to acetyl or benzoyl variants, thereby streamlining the purification process.
Q: How is stereocontrol achieved during the glycosylation steps?
A: Stereocontrol is managed through the use of specific glycosyl donors like 2,3,4-tribenzoyloxyrhamnose (Formula I) and 2,3,4,6-tetrabenzoyloxyglucose bromide (Formula H). The Mitsunobu reaction conditions and phase-transfer catalyzed nucleophilic substitution ensure the correct alpha and beta linkages required for biological activity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Apigenin Glycoside Intermediate Supplier
The successful commercialization of complex flavonoid therapeutics requires a partner with deep technical expertise in multi-step organic synthesis and process optimization. NINGBO INNO PHARMCHEM stands at the forefront of this capability, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific reaction conditions required for this synthesis, including low-temperature deprotections and pressure hydrogenations, ensuring that every batch meets stringent purity specifications. Our rigorous QC labs utilize advanced analytical techniques to verify the structural integrity and impurity profile of every intermediate, guaranteeing that the final API precursor is fit for purpose in global regulatory filings.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this efficient synthetic route for your cardiovascular drug development programs. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact us today to request specific COA data for our flavonoid intermediates and discuss route feasibility assessments that can accelerate your path to market while optimizing your manufacturing budget.
