Scalable Synthetic Route for Apigenin Glycoside Intermediates and Commercial Production
The pharmaceutical industry constantly seeks robust supply chains for bioactive molecules, particularly those with potent therapeutic potential like flavonoid glycosides. Patent CN101712701A introduces a groundbreaking synthetic methodology for producing apigenin-7-O-β-D-glucopyranosyl-4'-O-α-L-rhamnopyranosid, a compound with significant efficacy in treating ischemic diseases. Unlike traditional methods that rely on the arduous extraction from plants such as Ranunculus sieboldii, where yields are abysmally low (approximately 165mg from 17kg of dry material), this invention provides a chemically synthesized pathway. This shift from extraction to synthesis represents a paradigm change for procurement teams, ensuring a reliable pharmaceutical intermediate supplier can meet global demand without being constrained by agricultural harvest cycles or geographical limitations. The patent details a series of novel intermediates, specifically Formula D, which serve as the cornerstone for this efficient production line.
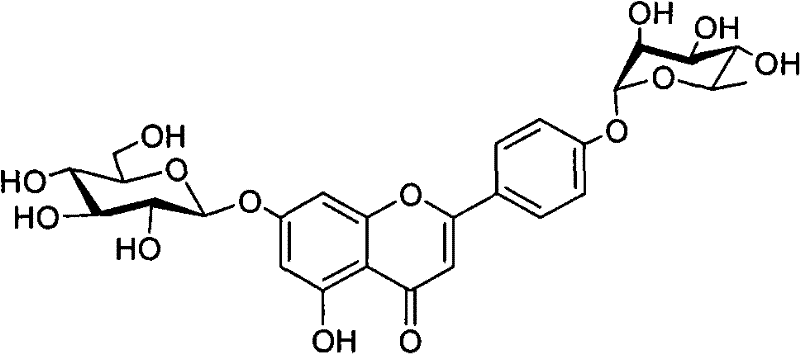
Historically, the reliance on natural extraction for complex flavonoid glycosides has been a major bottleneck for drug development. The conventional approach involves harvesting specific botanical sources, followed by extensive solvent extraction and purification processes that are not only environmentally taxing but also economically inefficient due to the trace amounts of the target compound present in the biomass. This variability in raw material quality often leads to inconsistent batch-to-batch purity, a critical concern for R&D directors managing impurity profiles for regulatory filings. In stark contrast, the novel approach outlined in the patent utilizes a stepwise organic synthesis strategy starting from commercially available apigenin. By employing strategic protecting group manipulations, specifically using acyl and benzyl groups, the synthesis achieves high regioselectivity. This method eliminates the unpredictability of biological sources, offering a deterministic path to high-purity intermediates that can be rigorously controlled and optimized for cost reduction in API manufacturing.
Mechanistic Insights into Mitsunobu Coupling and Selective Deprotection
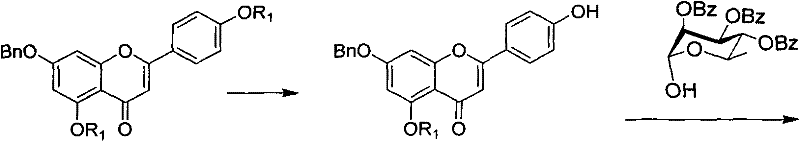
The core of this synthetic innovation lies in the precise control of glycosidic bond formation and protecting group orthogonality. A pivotal step involves the Mitsunobu reaction between the phenolic hydroxyl group of the apigenin derivative (Formula E) and the anomeric hydroxyl of the protected rhamnose (Formula I). This reaction is typically conducted in aprotic solvents like dimethylformamide at temperatures ranging from -20°C to 20°C, utilizing triphenylphosphine coupled with diethyl azodicarboxylate (DEAD) or dibenzyl azodicarboxylate (DBAD). The choice of the R1 protecting group on the apigenin scaffold is critical; experimental data within the patent demonstrates that when R1 is a hexanoyl group (-CO(CH2)4CH3), the intermediate exhibits superior solubility characteristics. This enhanced solubility directly translates to improved reaction kinetics and higher isolated yields during the coupling phase, minimizing the formation of difficult-to-remove side products.
Following the initial coupling, the synthesis proceeds through a catalytic hydrogenation step to remove benzyl protecting groups, followed by a phase-transfer catalyzed glycosylation with protected glucose. The final deprotection is achieved under mild alkaline conditions using sodium methoxide. This sequence ensures that the sensitive glycosidic linkages remain intact while removing the bulky acyl groups. The mechanistic elegance of this route allows for the isolation of stable intermediates like Formula D and Formula B, which can be stockpiled or further processed. For supply chain heads, this modularity means that production can be paused at stable intermediate stages, reducing the risk of total batch loss and allowing for flexible inventory management. The entire pathway avoids the use of heavy metal catalysts that require complex removal steps, thereby simplifying the downstream processing and waste treatment protocols significantly.
How to Synthesize Apigenin Glycoside Intermediates Efficiently
The synthesis of these high-value flavonoid intermediates requires strict adherence to the optimized reaction conditions described in the patent to ensure maximum yield and purity. The process begins with the selective deprotection of fully protected apigenin derivatives, followed by the critical Mitsunobu coupling with rhamnose donors. Subsequent steps involve catalytic hydrogenation to expose the 7-hydroxyl group, which is then glycosylated with a protected glucose bromide donor. The final step involves global deprotection to reveal the active pharmacophore. Each step has been optimized for solvent systems, temperature ranges, and stoichiometric ratios to prevent side reactions such as migration of acyl groups or hydrolysis of the glycosidic bond. Detailed standardized operating procedures for these transformations are essential for maintaining consistency across large-scale batches.
- Selective Deprotection: Convert protected apigenin derivative (Formula F) to Formula E using alkaline reagents like potassium carbonate in mixed solvents.
- Mitsunobu Coupling: React Formula E with tribenzoyloxyrhamnose (Formula I) using triphenylphosphine and azodicarboxylate to form Formula D.
- Hydrogenation and Glycosylation: Remove benzyl groups via catalytic hydrogenation to get Formula C, then couple with bromoglucose (Formula H) to form Formula B.
- Final Deprotection: Treat Formula B with sodium methoxide to remove acyl protecting groups and yield the final target compound (Formula A).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the transition from extraction to this synthetic route offers profound logistical and economic benefits. The primary advantage is the decoupling of supply from agricultural constraints. Plant-based extraction is subject to seasonal variations, climate change impacts, and geopolitical instability in sourcing regions. By adopting this chemical synthesis route, companies can secure a continuous, year-round supply of the intermediate, drastically reducing lead time for high-purity pharmaceutical intermediates. Furthermore, the starting materials, such as apigenin and protected sugars, are commodity chemicals available from multiple global vendors, mitigating the risk of single-source dependency. This diversification of the supply base enhances resilience against market shocks and price volatility.
- Cost Reduction in Manufacturing: The synthetic route utilizes standard, off-the-shelf reagents such as potassium carbonate, triphenylphosphine, and palladium on carbon, avoiding the need for expensive, proprietary catalysts or rare earth metals. The reaction conditions are mild, typically operating between -20°C and 50°C, which reduces energy consumption compared to high-temperature or high-pressure processes. Additionally, the high solubility of the hexanoyl-protected intermediates minimizes solvent usage and improves filtration efficiency, leading to substantial cost savings in utility and waste disposal. The elimination of complex chromatographic purifications at every step, replaced by efficient crystallization or simple column chromatography, further drives down the cost of goods sold.
- Enhanced Supply Chain Reliability: The modular nature of the synthesis allows for the production and storage of stable intermediates like Formula D and Formula B. This capability enables manufacturers to build strategic inventory buffers, ensuring that final API production is not halted by minor disruptions in the supply of early-stage precursors. The robustness of the chemical steps, evidenced by high yields across multiple examples in the patent, guarantees a predictable output volume. This predictability is crucial for long-term capacity planning and meeting the rigorous delivery schedules required by multinational pharmaceutical clients.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing solvents like dichloromethane, ethanol, and ethyl acetate, which are well-understood in industrial settings and have established recovery protocols. The absence of toxic heavy metals simplifies the environmental impact assessment and reduces the burden on wastewater treatment facilities. The high atom economy of the key coupling steps and the ability to recycle solvents contribute to a greener manufacturing footprint. This alignment with environmental, social, and governance (ESG) goals makes the supply chain more attractive to investors and regulatory bodies focused on sustainable chemistry practices.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its implementation. The following questions address common concerns regarding yield optimization, impurity control, and scale-up feasibility. The answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a factual basis for decision-making. These insights help bridge the gap between laboratory research and commercial production realities.
Q: What are the advantages of this synthetic method over plant extraction?
A: Plant extraction yields are extremely low (e.g., 165mg from 17kg of dry herb), making it unviable for industrial scale. This synthetic route offers high yield, consistent purity, and scalability independent of agricultural variables.
Q: Which protecting group strategy provides the best overall yield?
A: Experimental data indicates that using a hexanoyl group (R1 = -CO(CH2)4CH3) provides optimal solubility and reaction efficiency, resulting in the highest total yield compared to acetyl or benzoyl variants.
Q: Is this process suitable for large-scale GMP manufacturing?
A: Yes, the process utilizes standard reagents (e.g., Pd/C, K2CO3) and mild conditions (-20°C to 50°C), avoiding exotic catalysts or extreme pressures, which facilitates easy scale-up and regulatory compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Apigenin Glycoside Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable supply of high-quality intermediates for cardiovascular and antiviral drug development. Our team of expert chemists has extensively analyzed the synthetic routes disclosed in patent CN101712701A and possesses the technical capability to execute this complex multi-step synthesis with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from clinical trials to market launch. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of apigenin glycoside intermediate meets the highest international standards.
We invite you to collaborate with us to optimize this synthesis for your specific needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Whether you need specific COA data for regulatory submissions or detailed route feasibility assessments for process validation, we are committed to supporting your supply chain goals. Contact us today to discuss how we can become your strategic partner in delivering reliable, cost-effective, and high-purity pharmaceutical intermediates.
