Scalable Biocatalytic Production of Key Aprepitant Intermediate (R)-[3,5-Bis(trifluoromethyl)phenyl]ethanol
Introduction to Advanced Biocatalytic Synthesis
The pharmaceutical industry continuously seeks robust and sustainable pathways for synthesizing chiral intermediates, particularly for blockbuster drugs like Aprepitant. Patent CN102102087B introduces a groundbreaking biocatalytic solution utilizing a novel strain, Leifsonia xyli HS0904, specifically engineered for the asymmetric reduction of [3,5-bis(trifluoromethyl)phenyl]ethanone. This technology addresses critical bottlenecks in the production of (R)-[3,5-bis(trifluoromethyl)phenyl]ethanol, a pivotal chiral building block. By leveraging whole-cell biocatalysis, this method achieves superior stereoselectivity and substrate tolerance compared to historical benchmarks. For R&D directors and procurement specialists, this represents a shift towards greener, more cost-effective manufacturing paradigms that align with modern regulatory standards. The strain, deposited under CCTCC NO: M2010241, offers a reliable alternative to transition metal-catalyzed processes, ensuring high optical purity without the burden of heavy metal residues.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of (R)-[3,5-bis(trifluoromethyl)phenyl]ethanol has relied heavily on chemical asymmetric hydrogenation or enzymatic kinetic resolution, both of which present significant operational challenges. Chemical routes often necessitate the use of precious metal catalysts such as rhodium or ruthenium complexes, which are not only prohibitively expensive but also introduce severe environmental liabilities due to heavy metal waste streams. Alternatively, enzymatic kinetic resolution strategies, while effective, suffer from an inherent theoretical yield limitation of 50% unless coupled with dynamic processes. As illustrated in prior art literature referenced within the patent background, lipase-catalyzed transesterification using agents like Candida antarctica lipase-B (CAL-B) requires multiple steps including acylation and subsequent hydrolysis to recover the desired enantiomer.
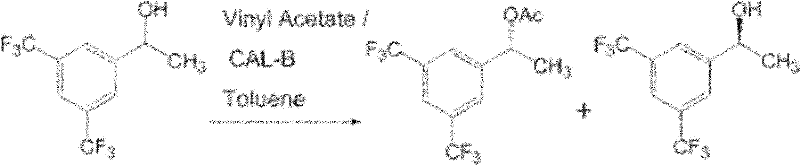
Furthermore, these conventional biocatalytic methods often struggle with low substrate loading capacities. For instance, earlier reports indicate that when substrate concentrations exceed modest levels, such as 50mmol/L, the conversion rates drop precipitously, rendering the process economically unviable for industrial applications. The necessity for organic solvents in some resolution protocols further complicates downstream processing and increases the overall carbon footprint of the manufacturing cycle. These cumulative inefficiencies drive up the cost of goods sold (COGS) and create supply chain vulnerabilities for manufacturers of NK-1 receptor antagonists.
The Novel Approach
In stark contrast, the novel approach detailed in CN102102087B utilizes the newly isolated Leifsonia xyli HS0904 strain to perform direct asymmetric reduction, bypassing the yield ceilings associated with kinetic resolution. This whole-cell biocatalyst demonstrates remarkable robustness, maintaining high catalytic activity even at substrate concentrations as high as 200mmol/L, which is a substantial improvement over the 5mmol/L to 50mmol/L limits observed in previous microbial studies. The process operates under mild physiological conditions, typically around 30°C and neutral pH, eliminating the need for high-pressure hydrogenation equipment or cryogenic temperatures. By employing a co-substrate system, such as isopropanol or glucose, the system efficiently regenerates necessary cofactors in situ, sustaining the redox balance required for continuous reduction. This streamlined single-step transformation significantly reduces unit operations, thereby enhancing the overall process mass intensity (PMI) and offering a clear pathway for cost reduction in API manufacturing.
Mechanistic Insights into Leifsonia xyli-Mediated Asymmetric Reduction
The core of this technological advancement lies in the specific enzymatic machinery within the Leifsonia xyli HS0904 cells that facilitates highly stereoselective carbonyl reduction. The biocatalyst likely employs NADPH-dependent oxidoreductases that exhibit strict geometric recognition of the ketone substrate, preferentially delivering a hydride ion to the re-face of the carbonyl group to generate the (R)-configured alcohol. This intrinsic selectivity is encoded in the strain's genome, as evidenced by the 16S rDNA sequence analysis showing 99% homology with Leifsonia xyli. The cellular environment provides a protective matrix for these enzymes, enhancing their stability against the potentially inhibitory effects of the fluorinated substrate. Unlike isolated enzyme systems that may require expensive external cofactor addition, the whole-cell format leverages the microbe's endogenous metabolic pathways to recycle cofactors, ensuring sustained catalytic turnover over extended reaction periods.
Impurity control is another critical aspect where this biological mechanism excels over chemical counterparts. The high enantioselectivity (ee > 99.9%) ensures that the formation of the undesired (S)-enantiomer is virtually suppressed, simplifying the purification landscape. In chemical catalysis, trace metal leaching or non-selective background reactions can lead to complex impurity profiles that require rigorous chromatographic separation. However, the biocatalytic route produces a cleaner crude profile, primarily consisting of the target alcohol and unreacted ketone, which can be easily separated via standard extraction or crystallization techniques. This purity advantage is paramount for pharmaceutical intermediates, where strict specifications regarding chiral purity and residual solvents must be met to satisfy global regulatory bodies like the FDA and EMA.
How to Synthesize (R)-[3,5-Bis(trifluoromethyl)phenyl]ethanol Efficiently
Implementing this biocatalytic route requires precise control over fermentation and bioconversion parameters to maximize yield and productivity. The patent outlines a systematic approach starting from strain activation to final product isolation, emphasizing the importance of optimized media composition and reaction conditions. Operators must ensure that the wet thallus cells are harvested at the peak of their enzymatic activity, typically after 48 hours of cultivation in a glucose-rich medium. The subsequent bioconversion step relies on maintaining an optimal pH of 8.0 and a temperature of 30°C to facilitate rapid kinetics. Detailed standardized synthetic steps see the guide below.
- Cultivate Leifsonia xyli HS0904 in optimized fermentation medium containing glucose and yeast extract at 30°C for 48 hours to obtain wet thallus cells.
- Suspend the wet cells in phosphate buffer (pH 8.0) and add the substrate [3,5-bis(trifluoromethyl)phenyl]ethanone along with a co-substrate like isopropanol for cofactor regeneration.
- Maintain the bioconversion reaction at 30°C with shaking for 24 to 48 hours, followed by extraction with ethyl acetate to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Leifsonia xyli-based technology translates into tangible strategic benefits beyond mere technical feasibility. The elimination of precious metal catalysts removes a significant variable cost component and mitigates the risk associated with the volatility of rhodium and ruthenium markets. Furthermore, the ability to run reactions at high substrate concentrations directly impacts facility throughput, allowing manufacturers to produce larger batches in existing reactor volumes without capital expenditure on new equipment. This efficiency gain is crucial for meeting the growing global demand for anti-emetic therapies while maintaining competitive pricing structures.
- Cost Reduction in Manufacturing: The transition from chemical hydrogenation to biocatalysis fundamentally alters the cost structure by removing the need for expensive transition metal ligands and high-pressure infrastructure. Since the process utilizes renewable biological catalysts that can be propagated via fermentation, the recurring cost of the catalytic agent is drastically reduced compared to single-use chemical catalysts. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, contributing to lower utility costs per kilogram of product. The simplified downstream processing, resulting from high selectivity and fewer by-products, further decreases solvent usage and waste disposal fees, driving substantial overall cost savings.
- Enhanced Supply Chain Reliability: Relying on a fermentable microbial strain ensures a sustainable and scalable source of catalytic activity that is not subject to the geopolitical supply constraints often affecting rare earth metals. The robustness of the Leifsonia xyli HS0904 strain allows for consistent batch-to-batch performance, reducing the risk of production delays caused by catalyst deactivation or variability. High substrate tolerance means that raw material logistics are streamlined, as fewer batches are needed to achieve the same output volume, thereby reducing the frequency of raw material deliveries and inventory holding costs. This reliability is essential for securing long-term supply agreements with major pharmaceutical partners.
- Scalability and Environmental Compliance: The aqueous nature of the bioconversion system aligns perfectly with green chemistry principles, significantly reducing the generation of hazardous organic waste streams associated with traditional organic synthesis. Scaling this process from laboratory to commercial production is facilitated by the use of standard fermentation and stirred-tank reactor technologies familiar to most CDMOs. The absence of heavy metals simplifies the environmental permitting process and reduces the burden on wastewater treatment facilities. Consequently, manufacturers can achieve commercial scale-up of complex pharmaceutical intermediates with a smaller environmental footprint, enhancing corporate sustainability profiles and ensuring compliance with increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biocatalytic platform. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the fit of this technology within their existing manufacturing portfolios.
Q: What are the advantages of using Leifsonia xyli HS0904 over chemical catalysts?
A: Unlike traditional chemical methods requiring expensive rhodium or ruthenium catalysts, this biocatalytic route operates under mild conditions (30°C, neutral pH) and eliminates heavy metal contamination risks, ensuring higher environmental compliance and lower purification costs.
Q: What optical purity and yield can be achieved with this strain?
A: The patent data demonstrates that at a high substrate concentration of 200mmol/L, the process achieves an exceptional enantiomeric excess (ee) of 99.9% and a productive rate of 82.9%, significantly outperforming many previously reported microbial strains.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the strain exhibits high substrate tolerance (up to 200mmol/L) and robust catalytic efficiency, which are critical parameters for reducing reactor volume and processing time in commercial-scale production of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-[3,5-Bis(trifluoromethyl)phenyl]ethanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the Leifsonia xyli HS0904 technology in securing the supply chain for critical oncology supportive care drugs. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to industrial manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the critical chiral purity metrics required for this intermediate. We are committed to delivering high-quality materials that meet the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with us to leverage this advanced biocatalytic route for your upcoming projects. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this green chemistry approach can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective supply of this vital chiral building block.
